-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
Consumption of Meals Prepared at Home and Risk of Type 2 Diabetes: An Analysis of Two Prospective Cohort Studies
With the aim of informing healthy eating behaviors, Qi Sun and colleagues investigate associations between the proportion of meals consumed by US health professionals that were prepared at home and incidence of type 2 diabetes.
Published in the journal: Consumption of Meals Prepared at Home and Risk of Type 2 Diabetes: An Analysis of Two Prospective Cohort Studies. PLoS Med 13(7): e32767. doi:10.1371/journal.pmed.1002052
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002052Summary
With the aim of informing healthy eating behaviors, Qi Sun and colleagues investigate associations between the proportion of meals consumed by US health professionals that were prepared at home and incidence of type 2 diabetes.
Introduction
Dining out has become increasingly popular in many countries [1–5]. In the US, percentages of calorie intake attributed to meals prepared out of home (MPOH) increased from <10% in 1965–1966 to >31% in 2005–2008, and this has been accompanied by a significantly reduced time spent on cooking [2,3,6]. According to recent national survey data, 35%, 28%, and 61% of American adults ate in fast-food restaurants, full-service restaurants, and nonrestaurant sources of MPOH daily [7], with more than 10% of their energy intake coming from fast foods regardless of income levels [8].
MPOH are usually high in energy and fat but low in micronutrients, such as calcium, vitamin C, and iron [9]. One study reported that participants consuming MPOH had lower serum concentrations of vitamins D, E, C, B-6, B-12, folate, and carotenoids [10]. This less-optimal nutritional profile has raised public health concerns regarding the impact of eating out on long-term well-being, especially in relation to obesity and related chronic diseases [2,11]. In support of this, existing studies have reported a positive association between MPOH, especially fast-food intake, and weight gain among children and young adults [12], although data for diabetes are relatively sparse [13–16].
More frequent consumption of MPOH is often at the cost of consuming fewer meals prepared at home (MPAH) [2,17]. In contrast to accumulating evidence regarding potential adverse effects of MPOH or fast foods, few studies have explicitly evaluated consuming MPAH in relation to health consequences. Previously, eating dinner cooked at home has been associated with lower intakes of fat and sugar [17], but whether eating MPAH is associated with obesity and obesity-related diseases remains largely unknown. To narrow the knowledge gap, we investigated the association between frequencies of consuming midday and evening MPAH and the risk of type 2 diabetes (T2D) in middle-aged and older US men and women.
Methods
Ethics Statement
The study protocol was approved by the institutional review boards of the Brigham and Women's Hospital and the Harvard T. H. Chan School of Public Health. Completion of the self-administered questionnaire was considered to imply informed consent.
Study Populations
The current investigation was based on two prospective cohort studies: the Nurses’ Health Study (NHS), which includes 121,700 female registered nurses aged 30–55 y who were enrolled in 1976 [18], and the Health Professionals Follow-up Study (HPFS), which consists of 51,529 male health professionals aged 40–75 y who were enrolled in 1986 [19]. Participants in both studies have been followed through mailed biennial questionnaires to collect and update information on lifestyles, health-related behaviors, and medical histories. Active follow-up rates exceed 90% in both cohorts.
In the current investigation, the study baseline was 1986 when information on MPAH was collected in both cohorts. Among the 75,521 participants in NHS and 51,529 in HPFS with completed baseline food frequency questionnaire (FFQ), we excluded those who reported a diagnosis of diabetes, cardiovascular diseases, or cancer at baseline (n = 10,828 in NHS, and n = 6,933 in HPFS); had daily energy intake out of a normal range (<500 or >3,500 kcal/d for NHS and <800 or >4,200 kcal/d for HPFS) or missing data of baseline MPAH (n = 5,889 in NHS, and n = 1,507 in HPFS); had a missing diagnosis date of T2D (n = 262 in HPFS); completed the baseline questionnaire only or with missing age at baseline (n = 753 in NHS, and n = 1,151 in HPFS), leaving 58,051 women and 41,676 men for the analyses (S1 Fig). In both NHS and HPFS, the distribution of demographic, socioeconomic, anthropometric, and lifestyle factors were similar between participants who had missing MPAH data and those included in the final analysis.
Ascertainment of Diet and Frequencies of MPAH
Since 1986 and every 4 y thereafter, a validated 138-item FFQ was sent to participants in both cohorts to assess and update their habitual diet during the past year [20]. We calculated cumulative averages of diet based on valid assessments from baseline to the end of follow-up and stopped updating dietary information if participants reported a diagnosis of diabetes, cardiovascular disease, or cancer [20]. Nutrient intakes were adjusted for total energy intake using the residual method [21]. The 2010 Alternative Health Eating Index (AHEI) was calculated as described previously with alcohol consumption excluded [20].
In 1986, participants were asked the following two questions: “How often your midday meals were prepared at home” and “How often your evening meals were prepared at home,” with five prespecified response categories in the NHS (never, 1–2, 3–4, 5–6, and 7 times/week) and four categories in HPFS (never, 1–2, 3–4, and 5–7 times/week). We summed midpoint values of midday and evening MPAH in each category to generate the overall MPAH frequency. To ensure an adequate number of T2D cases in each comparison group while maintaining a meaningful variation, we categorized overall MPAH frequency into four groups (0–6, 7–8, 9–10, or 11–14 times/week) and frequency of consuming midday or evening MPAH into three groups (0–2, 3–4, or 5–7/week). In the 2002 questionnaire of NHS and the 2004 and 2006 questionnaires of HPFS, the frequency of eating MPAH (either lunch or dinner) was assessed again, and response categories were changed to the following: almost none, 25%, 50%, 75%, and almost all. For the analysis of overall MPAH, we matched the same categories to the 2002–2006 MPAH question as follows: “almost none” and “25%” were assigned to 0–6 MPAH, “50%” to 7–8 MPAH, “75%” to 9–10 MPAH, and “almost all” to 11–14 MPAH. We used this updated MPAH frequency to account for potential changes in diet behavior and food environment during the long follow-up. The analysis for midday and evening MPAH used baseline MPAH frequency only.
Ascertainment of Incident T2D
In both cohorts, participants who reported a diagnosis of diabetes were mailed a supplementary questionnaire regarding symptoms, diagnostic tests, and hypoglycemic therapy. The diagnosis of T2D was considered confirmed if at least one of the following was reported on the supplementary questionnaire according to the National Diabetes Data Group criteria [22]: (1) one or more classic symptoms (excessive thirst, polyuria or frequent urination, weight loss, and hunger) plus fasting plasma glucose ≥7.8 mmol/l or random plasma glucose levels ≥11.1 mmol/l; (2) ≥2 elevated plasma glucose concentrations on different occasions (fasting glucose ≥7.8 mmol/l, random plasma glucose ≥11.1 mmol/l, and/or plasma glucose ≥11.1 mmol/l after ≥2 h shown by oral glucose tolerance testing) in the absence of symptoms; or (3) treatment with hypoglycemic medication (insulin or oral hypoglycemic agent). The diagnostic criteria changed in June 1998, and a fasting plasma glucose of 7.0 mmol/l was considered the threshold for the diagnosis of diabetes instead of 7.8 mmol/l according to the American Diabetes Association criteria [23]. In validation studies, 61 of 62 self-reported cases of T2D in the NHS who were confirmed by the supplementary questionnaire were reconfirmed after an endocrinologist reviewed the medical records [24]. In the HPFS, 57 of 59 self-reported diabetes cases (97%) were reconfirmed by medical record review [25].
Assessment of Covariables
Information on anthropometric, demographic, socioeconomic, and lifestyle factors, including body weight, marital status, employment status, number of children, cigarette smoking, physical activity, family history of diabetes, multivitamin use, menopausal status, and hormone use, was updated in follow-up questionnaires. Physical activity was estimated by multiplying the energy expenditure in metabolic equivalent tasks (METs) measured in hours per week by hours spent on the activity, and the values of all activities were summed to derive total physical activity. Body mass index (BMI) was calculated as self-reported weight in kilograms divided by height in meters squared.
Statistical Analysis
To minimize sample reduction because of missing covariables, we used indicator variables for missing categorical variables (cigarette smoking, marital status, employment status, number of children, menopausal status, and postmenopausal hormone use) at baseline and follow-up. The overall percentages of missing values during follow-up ranged from 0.5% (for cigarette smoking) to 7.4% (for employment status) in the NHS, and from 0.1% (for physical activity) to 23.0% (for employment status) in the HPFS. For BMI and physical activity, missing data during follow-up were replaced with the last valid values, and dummy variables were created for missing baseline data when making categories. The overall percentages of missing data for BMI and physical activity were 0.3% and 6.1% in the NHS and 0.3% and 0.1% in the HPFS, respectively. Excluding employment status as a covariable or restricting analysis among participants with valid data of employment status did not change the results.
We calculated Spearman’s rank correlation coefficients between midday and evening MPAH and the correlation coefficients of MPAH frequency assessed at baseline and during follow-up. Person-years were calculated from the return of baseline FFQ to the diagnosis of T2D, last return of a valid follow-up questionnaire, death, or end of follow-up (2012 in NHS and 2010 in HPFS), whichever came first. Cox proportional hazards models were used to calculate hazard ratios (HRs) and 95% confidence intervals (95% CIs) for the association between frequencies of consuming MPAH and T2D risk. To account for confounding of age and time, the Cox regression analysis was stratified by age in months and calendar year. Multivariable models were adjusted for ethnicity (Caucasian, African American, Hispanic, or Asian), marital status (married, not married, or missing), employment status (full-time, part-time, retired, or missing), number of children (none, 1–2, 3–4, 5 or more, or missing), smoking status (never smoked, past smoker, currently smokes 1–14 cigarettes/d, currently smokes 15–24 cigarettes/d, currently smokes ≥25 cigarettes/d, or missing), alcohol intake (gram/d: 0, 0.1–4.9, 5.0–14.9, or >15.0 in women; 0, 0.1–4.9, 5.0–29.9, or >30.0 in men; or missing), multivitamin use (yes or no), family history of diabetes (yes or no), menopause status and postmenopausal hormones use (women only: premenopause, never, former, or current hormone use, or missing), physical activity (METs/week: 0–2.9, 3–8.9, 9–17.9, 18–26.9, ≥27.0, or missing), and total energy intake (kcal/d). We adjusted for employment status, number of children, and marital status in both cohorts to control for potential confounding by socioeconomic status. In a secondary analysis of NHS data, we further adjusted for the highest degree acquired (registered nurse, bachelor’s degree, master’s degree and above, or missing), the education attainment of participant’s husband (some high school or below, high school graduate, college graduate, graduate school, or missing), participant’s perception on her standing in US society (top 20%, 30%, 40%, 50%, >50%, or missing), and participant’s perception on her standing in local community (top 20%, 30%, 40%, 50%, >50%, or missing) to explore whether the associations of interest are independent of these socioeconomic variables. We mutually adjusted for midday MPAH and evening MPAH when modeling their associations with T2D to control for potential confounding by each other. The linear trend was tested by modeling the median values for frequencies of MPAH categories as a continuous variable. HRs from the two cohorts were pooled using a fixed effects model, and the Cochrane Q statistic and the I2 statistic were used to examine the heterogeneity of associations between the two cohorts.
We examined the association between MPAH and weight gain from baseline to 1994 when the mean age of the study participants reached 60 y [26], because differential body weight loss at an older age may obscure true associations of interest [27]. Differences in weight changes across the MPAH groups were estimated using a general linear model, after excluding participants with missing body weight at baseline or with only baseline weight available till 1994 (n = 4,930 in NHS, and n = 2,436 in HPFS). Among nonobese participants (defined by BMI < 30 kg/m2) at baseline (n = 49,355 in NHS, and n = 38,511 in HPFS), we additionally estimated the risk of developing obesity. Multivariable models were adjusted for the same covariables considered in the T2D analysis and further adjusted for baseline BMI for weight change.
Based on existing evidence suggesting that the frequency of eating out may influence food choices and weight change, which are also risk factors for T2D [9,12], we further evaluated whether BMI, overall dietary quality (as reflected by AHEI), and individual dietary factors (carbonated beverages, sodium, total fruits, total vegetables, red meats, processed meats, total dairy products, coffee, French fries, whole grains, the polyunsaturated fatty acids to saturated fatty acids ratio, and trans fat; in quintiles) during follow-up may explain the associations between MPAH and T2D. We estimated the proportion of associations between MPAH and diabetes that was attributable to these factors using a SAS macro developed by Spiegelman et al. (http://www.hsph.harvard.edu/donna-spiegelman/software/mediate/) [28]. We also adjusted for frequencies of consuming fried food away from home to explore whether the association between MPAH and T2D is independent of the energy-dense, obesogenic MPOH.
To test the robustness of associations between MPAH and T2D, we censored participants when they retired or divorced/separated from their partner after baseline survey, because these changes may possibly alter cooking frequency and dining behaviors, thus introducing extraneous variability into the associations of interest. To further exclude the possibility that participants with a very high risk of T2D or prediabetes may reduce their frequency of eating out in an effort to pursue a healthier diet, we excluded participants who reported incident T2D in the first 4 y of follow-up. We also conducted stratified analyses by age (<65 y, ≥65 y), BMI (<30 kg/m2, ≥30 kg/m2), physical activity (<18 METs/week, ≥18 METs/week), and smoking status (currently smoking or not) to determine potential modifying effects of these factors. Time-varying variables were used in stratified analyses, and the p-values for the product terms between median frequency of MAPH categories and stratification variables were used to evaluate the significance of interactions.
All statistical analyses were conducted using SAS 9.4 (SAS Institute, Cary, North Carolina), and p-values were two-sided with a significance level of 0.05.
Results
The baseline characteristics of the study participants are shown in Table 1. The mean age of the study populations was 52.5 y (women: 52.0 y; men: 53.0 y). In both cohorts, those reporting more frequent consumption of MPAH were older, with more children, and more likely to be married, retired, and nonsmokers. Physical activity was higher in women eating more MPAH, but the opposite was observed in men. Regarding dietary factors, men and women eating more MPAH had higher intakes of total calories, fruits, vegetables, red meats, dairy products, and whole grains and lower intakes of carbonated beverages and coffee. Women with different MPAH frequencies had similar intake of fried foods prepared out of home or at home, whereas men eating more MPAH had lower consumption of fried food cooked away from home and higher intake of fried food prepared at home. The distribution of participants’ characteristics was similar when the study populations were categorized according to either midday or evening MPAH (S1 and S2 Tables).
Tab. 1. Baseline characteristics in the NHS and the HPFS (1986) according to frequencies of consuming MPAH. 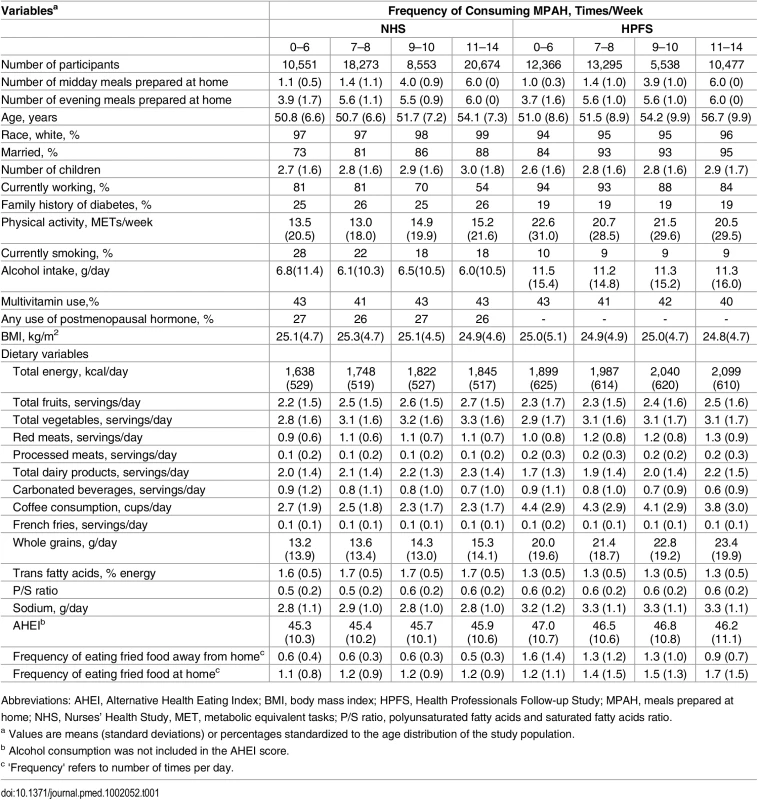
Abbreviations: AHEI, Alternative Health Eating Index; BMI, body mass index; HPFS, Health Professionals Follow-up Study; MPAH, meals prepared at home; NHS, Nurses’ Health Study, MET, metabolic equivalent tasks; P/S ratio, polyunsaturated fatty acids and saturated fatty acids ratio. Spearman’s rank correlation coefficients between frequencies of midday and evening MPAH were 0.19 in women and 0.26 in men (p < 0.001 for both). The correlations of MPAH frequencies at baseline and during follow-up were 0.22 (p < 0.001) between the 1986 and 2002 assessments for NHS, 0.34 (p < 0.001) between the 1986 and 2004 assessments, or 0.33 (p < 0.001) between the 1986 and 2006 assessments in HPFS. The correlation coefficient between the 2004 and 2006 MPAH assessments in HPFS was 0.69 (p < 0.001), suggesting that the frequency of eating MPAH was stable within a shorter period of time.
During 2.1 million person-years of follow-up (mean follow-up duration was 21 y), 9,356 cases of incident diabetes were documented. Associations between the frequency of consuming MPAH and T2D risk are shown in Table 2. In the age-adjusted model, increased consumption of MPAH was significantly associated with a lower risk of T2D. Findings did not materially change after further adjustment for other demographic and socioeconomic factors (model 2) or lifestyle factors (model 3). In model 3, pooled HRs (95% CIs) of T2D were 1 (reference), 0.93 (0.88–0.99), 0.96 (0.90–1.03), and 0.86 (0.81–0.91) for participants consuming 0–6, 7–8, 9–10, and 11–14 MPAH per week (p-trend < 0.001), respectively. As shown in S3 Table, the association between overall MPAH and diabetes risk remained when only baseline MPAH assessments were used for the analysis. Both midday and evening MPAH frequencies were associated with a lower risk of T2D, before or after adjustment for confounding factors. When midday and evening MPAH were mutually adjusted (model 4), participants eating 5–7 midday MPAH had a 9% (95% CI: 4%–13%; p-trend < 0.001) lower risk of T2D compared to the 0–2 midday MPAH group. For evening meals, people with 5–7 MPAH had a 15% (95% CI: 6%–22%; p-trend < 0.001) lower risk of T2D than those with 0–2 MPAH.
Tab. 2. Hazard ratios (95% confidence intervals) of type 2 diabetes according to frequencies of meals prepared at home in the Nurses’ Health Study (1986–2010) and the Health Professionals Follow-up Study (1986–2010). 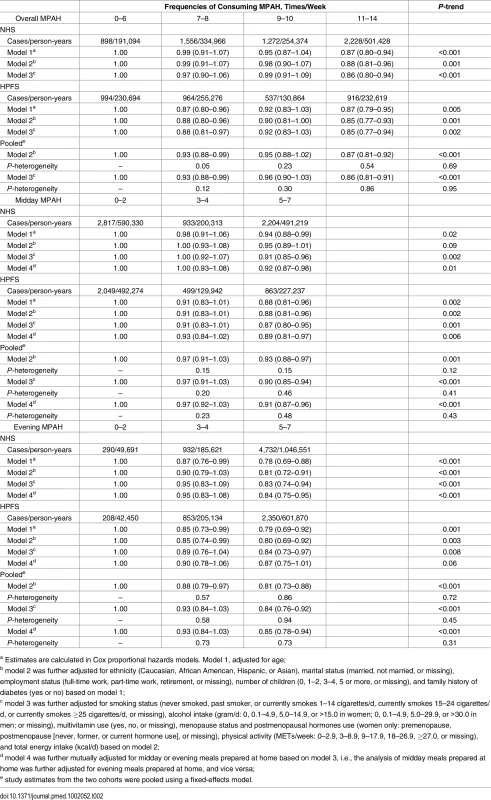
a Estimates are calculated in Cox proportional hazards models. Model 1, adjusted for age; During the follow-up period from 1986 to 1994, more frequent consumption of MPAH was associated with slower weight gain and a lower risk of developing obesity (Table 3). For women, significantly less weight gain (standard error) was observed only when extreme MPAH groups were compared (3.36 [0.06] kg for 0–6 MPAH versus 3.02 [0.05] kg for 11–14 MPAH; p < 0.001). For men, weight changes (standard error) were 3.85 (0.09, 0–6 MPAH) kg, 3.32 (0.08, 7–8 MPAH) kg, 3.34 (0.13, 9–10 MPAH) kg, and 2.62 (0.10, 11–14 MPAH) kg, and all groups with >6 MPAH had significantly less weight gain than the 0–6 MPAH group did (all p < 0.01 for between-group comparisons). Least squared mean differences in weight changes comparing the 11–14 with the 0–6 groups were −0.34 (95% CIs: −0.15 to −0.53; p < 0.001) kg in women and −1.23 (95% CIs: −0.92 to −1.54; p < 0.001) kg in men. Compared with the 0–6 MPAH group, pooled HR (95% CI) of developing obesity was 0.86 (0.82–0.91) for participants consuming 11–14 MPAH. Women with 5–7 midday MPAH had marginally less weight gain than those who ate 0–2 midday MPAH (p-trend = 0.06), whereas the same comparison reached significance in men (p-trend <0.01). For evening meals, men and women eating 5–7 evening MPAH gained less weight in comparison with the 0–2 evening MPAH groups (p-trend < 0.001). Consistently, participants eating 5–7 midday MPAH had a 7% lower risk of developing obesity (p < 0.001), whereas participants eating 5–7 evening MPAH had a 24% (p < 0.001) lower risk, compared with the corresponding 0–2 MPAH groups.
Tab. 3. Weight changes and risk of obesity according to frequencies of meals prepared at home, based on the NHS and the HPFS (1986–1994). 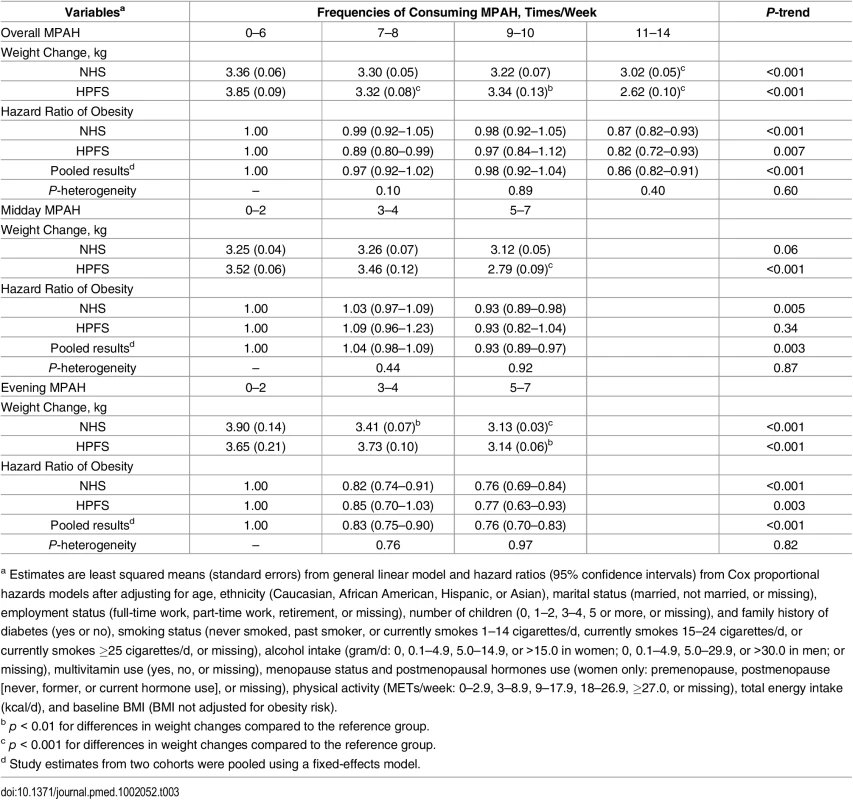
a Estimates are least squared means (standard errors) from general linear model and hazard ratios (95% confidence intervals) from Cox proportional hazards models after adjusting for age, ethnicity (Caucasian, African American, Hispanic, or Asian), marital status (married, not married, or missing), employment status (full-time work, part-time work, retirement, or missing), number of children (0, 1–2, 3–4, 5 or more, or missing), and family history of diabetes (yes or no), smoking status (never smoked, past smoker, or currently smokes 1–14 cigarettes/d, currently smokes 15–24 cigarettes/d, or currently smokes ≥25 cigarettes/d, or missing), alcohol intake (gram/d: 0, 0.1–4.9, 5.0–14.9, or >15.0 in women; 0, 0.1–4.9, 5.0–29.9, or >30.0 in men; or missing), multivitamin use (yes, no, or missing), menopause status and postmenopausal hormones use (women only: premenopause, postmenopause [never, former, or current hormone use], or missing), physical activity (METs/week: 0–2.9, 3–8.9, 9–17.9, 18–26.9, ≥27.0, or missing), total energy intake (kcal/d), and baseline BMI (BMI not adjusted for obesity risk). Further controlling for time-varying BMI attenuated the inverse association between MPAH and T2D (Table 4). We estimated that 57% (95% CI: 29%–81%; p < 0.001) of the association between MPAH and T2D was explained by BMI in women, and 24% (95% CI: 10%–48%; p < 0.001) was explained in men. The corresponding mediation effects were 62% (95% CI: 23%–90%; p < 0.001; women) for midday MPAH and 33% (95% CI: 19%–51%; p < 0.001; women) and 47% (95% CI: 10%–88%; p < 0.001; men) for evening MPAH. After further adjusting for dietary factors, the association between consuming MPAH and diabetes was attenuated when carbonated beverage intake was controlled for (Table 4). In women, carbonated beverage consumption was estimated to explain 22% (p < 0.001, for evening MPAH only) to 32% (p < 0.001, for both MPAH) of the association between MPAH and diabetes risk, whereas the values ranged from 23% (p < 0.001, for midday MPAH) to 41% (p < 0.001, for dinner) in men. Adjusting for fried food prepared away from home slightly attenuated the association for evening MPAH and overall MPAH. Other dietary factors did not explain the association between MPAH and T2D (Table 4 and S4 Table).
Tab. 4. Hazard ratios (95% confidence intervals) of type 2 diabetes according to frequencies of consuming meals prepared at home by further adjusting for BMI and dietary factors. 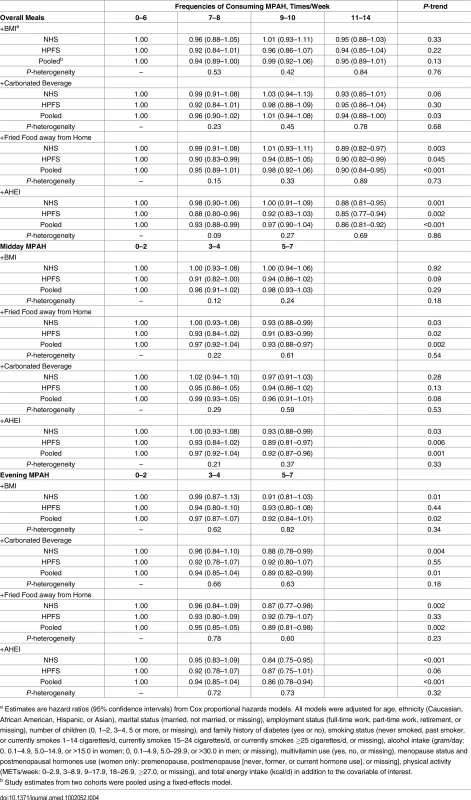
a Estimates are hazard ratios (95% confidence intervals) from Cox proportional hazards models. All models were adjusted for age, ethnicity (Caucasian, African American, Hispanic, or Asian), marital status (married, not married, or missing), employment status (full-time work, part-time work, retirement, or missing), number of children (0, 1–2, 3–4, 5 or more, or missing), and family history of diabetes (yes or no), smoking status (never smoked, past smoker, or currently smokes 1–14 cigarettes/d, currently smokes 15–24 cigarettes/d, or currently smokes ≥25 cigarettes/d, or missing), alcohol intake (gram/day: 0, 0.1–4.9, 5.0–14.9, or >15.0 in women; 0, 0.1–4.9, 5.0–29.9, or >30.0 in men; or missing), multivitamin use (yes, no, or missing), menopause status and postmenopausal hormones use (women only: premenopause, postmenopause [never, former, or current hormone use], or missing], physical activity (METs/week: 0–2.9, 3–8.9, 9–17.9, 18–26.9, ≥27.0, or missing), and total energy intake (kcal/d) in addition to the covariable of interest. We did not observe significant interactions of MPAH with age, BMI, physical activity, or smoking status on T2D risk (S5 Table). In sensitivity analyses, the associations between MPAH and T2D remained after participants who retired or divorced/separated were censored or when patients with incident T2D diagnosed in the first 4 y were excluded (S6 Table). In the NHS, further adjusting for the highest degree attained, the education attainment of the participant’s husband, the participant’s perception on her standing in US society, or the participant’s perception on her standing in the local community did not materially change the association between MPAH and T2D (S7 Table).
Discussion
In two large prospective cohort studies of US men and women, we found that more frequent consumption of MPAH was inversely associated with T2D risk during 24–26 y of follow-up. The association between eating MPAH and T2D risk was independent of established and potential demographic, socioeconomic, lifestyle, and dietary risk factors of diabetes and was partially explained by less weight gain among participants who consumed MPAH more frequently. When analyzed separately, both midday and evening MPAH frequencies were associated with lower risks of obesity and T2D, and the associations for evening MPAH tended to be stronger than those for midday MPAH. Our study is among the first to explicitly examine MPAH in relation to T2D risk, and thus enables a more comprehensive understanding regarding the health impact of the current trend in changing from eating homemade meals to dining out. These findings provide further evidence to support reducing eating out frequency and increasing consumption of homemade meals for the prevention of excess weight gain and diabetes [29].
Because having more midday and evening MPAH is generally accompanied by less consumption of MPOH, our findings are in line with previous studies that linked meals cooked away from home, especially fast foods, with an increased risk of diabetes. For example, fast-food consumption has been associated with a higher risk of gestational diabetes among 3,048 Spanish women [13]. In the Coronary Artery Risk Development in Young Adults (CARDIA) study, consuming fast foods was associated with increased insulin resistance after 10–13 y of follow-up [15]. However, dining out in sit-down restaurants or cafeterias was not associated with insulin resistance [30]. Higher consumption of fried chicken, fried fish, and Chinese food in sit-down and fast-food restaurants was positively associated with a 10-y diabetes risk in the Black Women’s Health Study (BWHS), whereas eating fried chicken and fried fish prepared at home was not [14]. In the NHS and HPFS, consumption of fried foods cooked away from home, but not those made at home, has also been associated with a higher risk of T2D and coronary heart disease [16]. Fried foods prepared away from home and carbonated beverages are often consumed at fast-food restaurants [16,31], and adjusting for these foods/beverages partly attenuated the associations between MPAH and T2D in the current study. Therefore, participants eating more MPAH may benefit from lower fast-food consumption but also from consuming MPAH as a healthy dining option per se.
We also found that participants consuming more MPAH had less weight gain and a lower risk of developing obesity. Similarly, previous studies focusing on MPOH demonstrated that frequent consumption of foods in sit-down and fast-food restaurants was associated with excess weight gain as a result of prolonged positive energy balance [14,15,30–34]. For instance, each serving/week of fast-food consumption at baseline was associated with 0.32–0.74 kg more weight gain over 15 y in the CARDIA study [15,30,32]. More importantly, we found that BMI during follow-up partly explained the association between MPAH and T2D. In BWHS, consuming more MPOH was associated with a higher risk of developing obesity among initially lean participants [31], and adjusting for baseline BMI markedly attenuated the association between MPOH and T2D risk [14]. Clearly, future studies are needed to explicitly investigate the association of consuming MPAH/MPOH with other obesity-related diseases and potential intermediate role of weight gain.
Interestingly, both midday and evening MPAH were associated with risks of obesity and T2D in our study, and the associations were stronger for evening MPAH than for midday MPAH. One possible reason is the higher contribution of evening meals to total energy and nutrient intake for Americans [35]. Over the past four decades, daily energy from lunch was typically between 23.3% and 26.8% among US adults, and the range for dinner was 36.2%–38.8% [35], which may lead to a greater health impact of dinner than lunch.
The strengths of our investigation include large sample sizes, long follow-up durations, and repeated measurements of lifestyle and dietary factors in the analysis. However, our research is subject to a few limitations. First, although our questions regarding MPAH explicitly inquired about the location of meal preparation instead of consumption, we cannot further distinguish the processing extent or nutritional profile of ingredients used for MPAH. Homemade meals could be based on either convenience foods or raw/fresh ingredients, and evidence suggests that ready-to-eat convenience foods purchased from grocery stores and supermarkets have poorer nutritional profiles than raw, unprocessed foods [36]. If these foods were considered as MPAH, the expected association between MPAH and T2D could have been weakened. However, it has been shown that older participants consider that preparing meals from scratch is an essential component of cooking more than young people do [37]. Based on this evidence, in our middle-aged and older NHS and HPFS participants, MPAH is more likely to be based on raw, unprocessed materials. Varying but unclear definitions of MPAH/MPOH in existing literature have been a major challenge for interpreting related findings [35]. In this regard, future studies need to carefully assess locations of meal preparation/consumption, ingredients/foods used on each occasion, serving size and eating frequency, extent of food preparation/cooking, and these factors’ interrelationships to facilitate further understanding of how MPAH/MPOH influences human health [5,12]. Second, our participants are exclusively health professionals and mainly Caucasians, limiting generalizability of our findings to populations with similar ethnic and socioeconomic characteristics. On the other hand, the homogeneous socioeconomic status of our participants also reduces the residual confounding by socioeconomic factors. Third, measurement errors in self-reported assessments are inevitable. However, validation studies in the two cohorts demonstrated reasonable validity of self-reported assessments of diet, body weight, and diabetes incidence. For example, the correlation coefficient between self-reported and measured body weight was 0.97 [36]. Fourth, we cannot exclude the role of residual or unmeasured confounding. The adjustment of employment status, marriage status, and number of children may not fully account for differences in socioeconomic status. Fifth, we observed higher energy intake among participants with more MPAH. It is likely that participants who prepared their own meals may be more conscious about the food that they ate compared with those who consumed their food more passively, leading to underestimated food and total energy intake by participants with fewer MPAH. Therefore, our investigation was not able to determine the role of total energy intake in the associations of MPAH consumption and the risks of obesity and T2D, and findings regarding food intakes across MPAH groups need to be interpreted with caution. Lastly, because of the observational design of the two cohorts, we were unable to establish a causal relationship between consuming MPAH and T2D. Our findings warrant replication in intervention settings as well as observational studies in other populations with various demographic and socioeconomic characteristics.
In conclusion, frequent consumption of MPAH is inversely associated with T2D risk, which is partly attributed to less weight gain among individuals who consumed more MPAH. From a public health prospective, it is critical to facilitate people’s access to healthful, affordable foods and cultivate their confidence and skills for food preparation to encourage the practice of cooking meals at home [17]. Meanwhile, as needs for quick and convenient meals remain [9,37], actions are needed to improve the diet quality of MPOH, including fast-food chain restaurants, full-service restaurants, convenience stores, vending machines, and workplace and school cafeterias, so that the public’s need for convenience meals can be met while the diet quality of Americans is improved.
Supporting Information
Zdroje
1. Smith LP, Ng SW, Popkin BM (2013) Trends in US home food preparation and consumption: analysis of national nutrition surveys and time use studies from 1965–1966 to 2007–2008. Nutr J 12 : 45. doi: 10.1186/1475-2891-12-45 23577692
2. Lin B-H, and Joanne Guthrie (December 2012) Nutritional Quality of Food Prepared at Home and Away From Home, 1977–2008. EIB-105, US Department of Agriculture, Economic Research Service.
3. Kant AK, Graubard BI (2004) Eating out in America, 1987–2000: trends and nutritional correlates. Prev Med 38 : 243–249. 14715218
4. Orfanos P, Naska A, Trichopoulos D, Slimani N, Ferrari P, et al. (2007) Eating out of home and its correlates in 10 European countries. The European Prospective Investigation into Cancer and Nutrition (EPIC) study. Public Health Nutr 10 : 1515–1525. 17582244
5. Bezerra IN, Sichieri R (2009) Eating out of home and obesity: a Brazilian nationwide survey. Public Health Nutr 12 : 2037–2043. doi: 10.1017/S1368980009005710 19433008
6. Zick CD, Stevens RB (2010) Trends in Americans' food-related time use: 1975–2006. Public Health Nutr 13 : 1064–1072. doi: 10.1017/S1368980009992138 19943999
7. Nguyen BT, Powell LM (2014) The impact of restaurant consumption among US adults: effects on energy and nutrient intakes. Public Health Nutr 17 : 2445–2452. doi: 10.1017/S1368980014001153 25076113
8. Fryer CD, Ervin RB (2013) Caloric intake from fast food among adults: United States, 2007–2010. NCHS Data Brief: 1–8.
9. Lachat C, Nago E, Verstraeten R, Roberfroid D, Van Camp J, et al. (2012) Eating out of home and its association with dietary intake: a systematic review of the evidence. Obes Rev 13 : 329–346. doi: 10.1111/j.1467-789X.2011.00953.x 22106948
10. Kant AK, Whitley MI, Graubard BI (2015) Away from home meals: associations with biomarkers of chronic disease and dietary intake in American adults, NHANES 2005–2010. Int J Obes (Lond) 39 : 820–827.
11. Lachat CK, Huybregts LF, Roberfroid DA, Van Camp J, Remaut-De Winter AM, et al. (2009) Nutritional profile of foods offered and consumed in a Belgian university canteen. Public Health Nutr 12 : 122–128. doi: 10.1017/S1368980008002048 18416870
12. US Department of Agriculture, Nutrition Evidence Library. What is the relationship between eating out and/or take away meals and body weight in children and adults? 2015. http://www.nel.gov/evidence.cfm?evidence_summary_id=250509
13. Dominguez LJ, Martinez-Gonzalez MA, Basterra-Gortari FJ, Gea A, Barbagallo M, et al. (2014) Fast food consumption and gestational diabetes incidence in the SUN project. PLoS ONE 9: e106627. doi: 10.1371/journal.pone.0106627 25215961
14. Krishnan S, Coogan PF, Boggs DA, Rosenberg L, Palmer JR (2010) Consumption of restaurant foods and incidence of type 2 diabetes in African American women. Am J Clin Nutr 91 : 465–471. doi: 10.3945/ajcn.2009.28682 20016014
15. Pereira MA, Kartashov AI, Ebbeling CB, Van Horn L, Slattery ML, et al. (2005) Fast-food habits, weight gain, and insulin resistance (the CARDIA study): 15-year prospective analysis. Lancet 365 : 36–42. 15639678
16. Cahill LE, Pan A, Chiuve SE, Sun Q, Willett WC, et al. (2014) Fried-food consumption and risk of type 2 diabetes and coronary artery disease: a prospective study in 2 cohorts of US women and men. Am J Clin Nutr 100 : 667–675. doi: 10.3945/ajcn.114.084129 24944061
17. Wolfson JA, Bleich SN (2015) Is cooking at home associated with better diet quality or weight-loss intention? Public Health Nutr 18 : 1397–1406. doi: 10.1017/S1368980014001943 25399031
18. Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, et al. (2001) Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med 345 : 790–797. 11556298
19. van Dam RM, Willett WC, Rimm EB, Stampfer MJ, Hu FB (2002) Dietary fat and meat intake in relation to risk of type 2 diabetes in men. Diabetes Care 25 : 417–424. 11874924
20. Chiuve SE, Fung TT, Rimm EB, Hu FB, McCullough ML, et al. (2012) Alternative dietary indices both strongly predict risk of chronic disease. J Nutr 142 : 1009–1018. doi: 10.3945/jn.111.157222 22513989
21. Willett WC, Howe GR, Kushi LH (1997) Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr 65 : 1220S–1228S; discussion 1229S-1231S. 9094926
22. National Diabetes Data Group (1979) Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. National Diabetes Data Group. Diabetes 28 : 1039–1057. 510803
23. The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (1997) Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 20 : 1183–1197. 9203460
24. Manson JE, Rimm EB, Stampfer MJ, Colditz GA, Willett WC, et al. (1991) Physical activity and incidence of non-insulin-dependent diabetes mellitus in women. Lancet 338 : 774–778. 1681160
25. Hu FB, Leitzmann MF, Stampfer MJ, Colditz GA, Willett WC, et al. (2001) Physical activity and television watching in relation to risk for type 2 diabetes mellitus in men. Arch Intern Med 161 : 1542–1548. 11427103
26. Field AE, Willett WC, Lissner L, Colditz GA (2007) Dietary fat and weight gain among women in the Nurses' Health Study. Obesity (Silver Spring) 15 : 967–976.
27. Kuk JL, Saunders TJ, Davidson LE, Ross R (2009) Age-related changes in total and regional fat distribution. Ageing Res Rev 8 : 339–348. doi: 10.1016/j.arr.2009.06.001 19576300
28. Lin DY, Fleming TR, De Gruttola V (1997) Estimating the proportion of treatment effect explained by a surrogate marker. Stat Med 16 : 1515–1527. 9249922
29. Committee DGA (2015) Scientific Report of the 2015 Dietary Guidelines Advisory Committee. Washington (DC): USDA and US Department of Health and Human Services.
30. Duffey KJ, Gordon-Larsen P, Steffen LM, Jacobs DR Jr., Popkin BM (2009) Regular consumption from fast food establishments relative to other restaurants is differentially associated with metabolic outcomes in young adults. J Nutr 139 : 2113–2118. doi: 10.3945/jn.109.109520 19776183
31. Boggs DA, Rosenberg L, Coogan PF, Makambi KH, Adams-Campbell LL, et al. (2013) Restaurant foods, sugar-sweetened soft drinks, and obesity risk among young African American women. Ethn Dis 23 : 445–451. 24392607
32. Duffey KJ, Gordon-Larsen P, Jacobs DR Jr., Williams OD, Popkin BM (2007) Differential associations of fast food and restaurant food consumption with 3-y change in body mass index: the Coronary Artery Risk Development in Young Adults Study. Am J Clin Nutr 85 : 201–208. 17209197
33. French SA, Harnack L, Jeffery RW (2000) Fast food restaurant use among women in the Pound of Prevention study: dietary, behavioral and demographic correlates. Int J Obes Relat Metab Disord 24 : 1353–1359. 11093299
34. Bes-Rastrollo M, Basterra-Gortari FJ, Sanchez-Villegas A, Marti A, Martinez JA, et al. (2010) A prospective study of eating away-from-home meals and weight gain in a Mediterranean population: the SUN (Seguimiento Universidad de Navarra) cohort. Public Health Nutr 13 : 1356–1363. doi: 10.1017/S1368980009992783 19954575
35. Bezerra IN, Curioni C, Sichieri R (2012) Association between eating out of home and body weight. Nutr Rev 70 : 65–79. doi: 10.1111/j.1753-4887.2011.00459.x 22300594
36. Rimm EB, Stampfer MJ, Colditz GA, Chute CG, Litin LB, et al. (1990) Validity of self-reported waist and hip circumferences in men and women. Epidemiology 1 : 466–473. 2090285
37. Blanck HM, Yaroch AL, Atienza AA, Yi SL, Zhang J, et al. (2009) Factors influencing lunchtime food choices among working Americans. Health Educ Behav 36 : 289–301. 17602103
Štítky
Interné lekárstvo
Článok vyšiel v časopisePLOS Medicine
Najčítanejšie tento týždeň
2016 Číslo 7- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Statinová intolerance
- Genetický podklad a screening familiární hypercholesterolémie
- Metabolit živočišné stravy produkovaný střevní mikroflórou zvyšuje riziko závažných kardiovaskulárních příhod
- DESATORO PRE PRAX: Aktuálne odporúčanie ESPEN pre nutričný manažment u pacientov s COVID-19
-
Všetky články tohto čísla
- The Clinical and Public Health Challenges of Diabetes Prevention: A Search for Sustainable Solutions
- Screening for Dysglycemia: Connecting Supply and Demand to Slow Growth in Diabetes Incidence
- Diabetes: A Cinderella Subject We Can’t Afford to Ignore
- Prescribing Exercise and Lifestyle Training for High Risk Women in Pregnancy and Early Post-partum—Is It Worth It?
- Population Approaches to Prevention of Type 2 Diabetes
- First-Year Evaluation of Mexico’s Tax on Nonessential Energy-Dense Foods: An Observational Study
- Effects of Saturated Fat, Polyunsaturated Fat, Monounsaturated Fat, and Carbohydrate on Glucose-Insulin Homeostasis: A Systematic Review and Meta-analysis of Randomised Controlled Feeding Trials
- Leveraging Genetics to Advance Type 2 Diabetes Prevention
- Cardiometabolic Risk Factor Changes Observed in Diabetes Prevention Programs in US Settings: A Systematic Review and Meta-analysis
- Risks and Population Burden of Cardiovascular Diseases Associated with Diabetes in China: A Prospective Study of 0.5 Million Adults
- Dietary Diversity, Diet Cost, and Incidence of Type 2 Diabetes in the United Kingdom: A Prospective Cohort Study
- Detecting Dysglycemia Using the 2015 United States Preventive Services Task Force Screening Criteria: A Cohort Analysis of Community Health Center Patients
- Exercise Training and Weight Gain in Obese Pregnant Women: A Randomized Controlled Trial (ETIP Trial)
- Associations between Recreational and Commuter Cycling, Changes in Cycling, and Type 2 Diabetes Risk: A Cohort Study of Danish Men and Women
- Association of Plasma Phospholipid n-3 and n-6 Polyunsaturated Fatty Acids with Type 2 Diabetes: The EPIC-InterAct Case-Cohort Study
- Engagement, Retention, and Progression to Type 2 Diabetes: A Retrospective Analysis of the Cluster-Randomised "Let's Prevent Diabetes" Trial
- Obesity and Life Expectancy with and without Diabetes in Adults Aged 55 Years and Older in the Netherlands: A Prospective Cohort Study
- Supported Telemonitoring and Glycemic Control in People with Type 2 Diabetes: The Telescot Diabetes Pragmatic Multicenter Randomized Controlled Trial
- Mothers after Gestational Diabetes in Australia (MAGDA): A Randomised Controlled Trial of a Postnatal Diabetes Prevention Program
- Cycling and Diabetes Prevention: Practice-Based Evidence for Public Health Action
- Consumption of Meals Prepared at Home and Risk of Type 2 Diabetes: An Analysis of Two Prospective Cohort Studies
- PLOS Medicine
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Mothers after Gestational Diabetes in Australia (MAGDA): A Randomised Controlled Trial of a Postnatal Diabetes Prevention Program
- Consumption of Meals Prepared at Home and Risk of Type 2 Diabetes: An Analysis of Two Prospective Cohort Studies
- Obesity and Life Expectancy with and without Diabetes in Adults Aged 55 Years and Older in the Netherlands: A Prospective Cohort Study
- Effects of Saturated Fat, Polyunsaturated Fat, Monounsaturated Fat, and Carbohydrate on Glucose-Insulin Homeostasis: A Systematic Review and Meta-analysis of Randomised Controlled Feeding Trials
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



