-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
Herpes Simplex Virus Reorganizes the Cellular DNA Repair and Protein Quality Control Machinery
article has not abstract
Published in the journal: Herpes Simplex Virus Reorganizes the Cellular DNA Repair and Protein Quality Control Machinery. PLoS Pathog 6(11): e32767. doi:10.1371/journal.ppat.1001105
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1001105Summary
article has not abstract
When a virus infects a cell, it must contend with a hostile environment and host machinery that is intrinsically antiviral. One of the hallmarks of herpes simplex virus (HSV) infection is the dramatic reorganization of the infected cell nucleus leading to the formation of large globular replication compartments in which gene expression, DNA replication, and encapsidation occur ([1], [2] and references therein) (see Figure 1). During infection, cellular factors that are beneficial to the virus are hijacked while other factors and pathways are degraded or inactivated. Two of the cellular homeostatic pathways affected by HSV-1 infection are the protein quality control (PQC) and DNA damage response pathways. These events are orchestrated by several viral proteins including the immediate early proteins ICP4, ICP27, ICP0, and ICP22 that allow the virus to create an environment conducive to infection and counteract the cell's intrinsic capacity to inhibit viral infection [3]–[6]. ICP4 and ICP27 play essential roles in stimulation of robust viral gene expression [3], [4]. The immediate early protein ICP0 activates viral and cellular gene expression and functions as an E3 ubiquitin ligase by degrading several cellular proteins [5], [7]. One target of ICP0 is PML, a major component of nuclear foci called ND10 that play a repressive role in viral gene expression. ICP0 interferes with several intrinsic host defense mechanisms including host interferon responses [5], [7], [8], thereby playing a major role in the establishment of a permissive viral infection. ICP22 is required for efficient growth and expression of late viral genes in some but not all cultured cells [6]. ICP22 also plays a role in the post-translational modification of the cellular RNA polymerase II ([9] and references therein). As described below, these immediate early proteins play important roles in remodeling the infected cell nucleus, hijacking host PQC machinery, and manipulating cellular DNA damage responses.
Fig. 1. Hsc70 is detected in virus-induced chaperone-enriched (VICE) domains that form adjacent to replication compartments in HSV-1-infected cells. 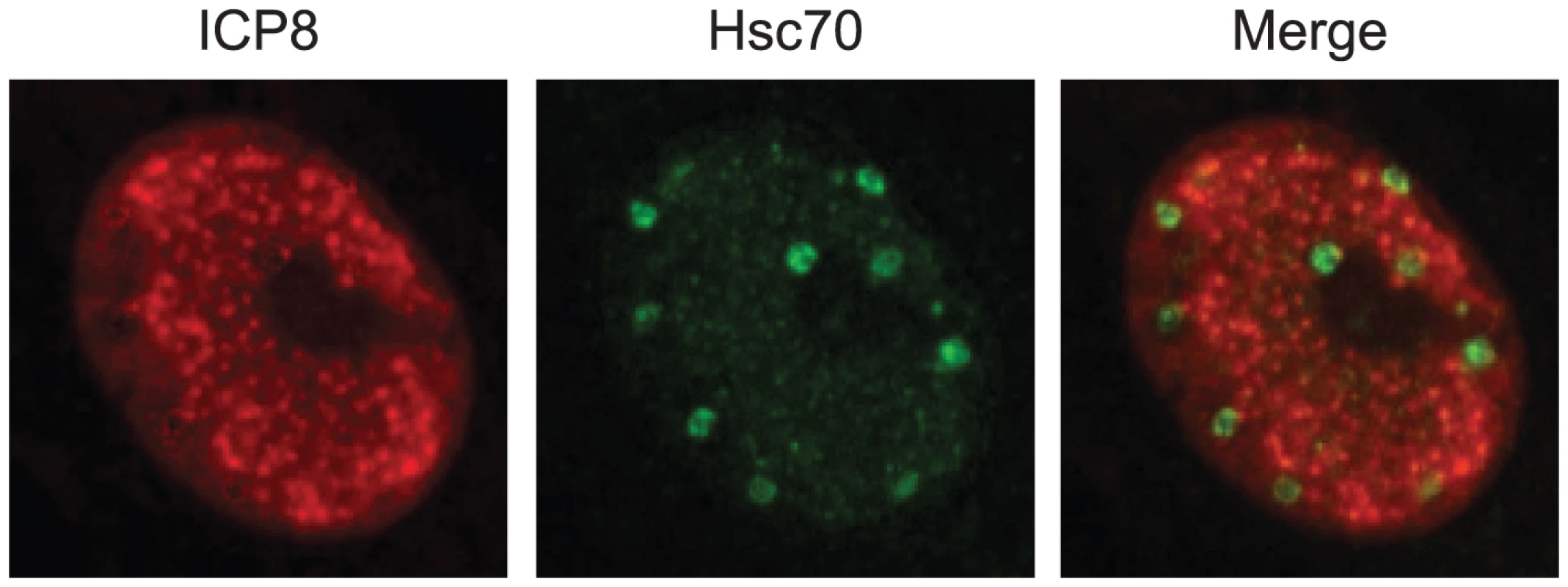
Vero cells were infected with HSV-1 strain KOS at an MOI of 10 for 6 hours. Infected cells were prepared for immunofluorescence imaging of ICP8 in red (left), Hsc70 in green (center), and a merged image (right) as described in [24]. HSV-1 Infection Results in Remodeling of the Infected Cell Nucleus
The formation of replication compartments follows an ordered assembly process, resulting in a drastic remodeling of the nucleus [1], [2], [10]–[13]. Following entry of viral genomes into the nucleus, nucleoprotein complexes containing viral immediate early proteins ICP4 and ICP27 can be detected on viral genomes [14]. PML and ND10 components are then recruited from ND10 to viral genomes at a position adjacent to the ICP4 foci [14]. The HSV immediate early protein ICP0 counteracts the silencing effects by degrading PML and other ND10 proteins in these newly formed ND10-like foci [15]. Replication proteins including the HSV single-strand DNA binding protein ICP8 are recruited into prereplicative sites that form at a position adjacent to ICP4 nucleoprotein complexes [11], [12]; efficient prereplicative site formation is dependent on the ability of ICP4 to oligomerize on DNA [2]. Prereplicative sites containing replication proteins eventually coalesce into mature replication compartments [16]. In addition to all seven replication proteins, several cellular proteins are also recruited to replication compartments (discussed below). As replication compartments grow and eventually occupy most of the volume of the nucleus, host chromatin is marginalized to the periphery of the nucleus [17], [18]. Other alterations caused by HSV infection include modifications in the nucleolus and disruption of the nuclear lamina [18]–[20].
HSV-1 Hijacks the Host Protein Quality Control Machinery
Remodeling of the nucleus also involves the reorganization of the cellular PQC machinery, including cellular chaperone proteins (Hsp27, Hsp40, Hsc70, Hsp70, and Hsp90) and components of the 20S proteosome [21], [22]. Within the first 2 hours of infection, Hsc70 and other components of the PQC machinery are recruited into nuclear foci called virus-induced chaperone-enriched (VICE) domains that form adjacent to replication compartments (Figure 1). Viral immediate early proteins have been implicated in the reorganization of PQC machinery into VICE domains; ICP0 and ICP27 are required for robust viral gene expression, and their roles in VICE domain formation may be indirect [23], [24]. On the other hand, ICP22 localizes to VICE domains [25] and appears to be directly required for Hsc70 reorganization [26]. VICE domains form after ND10-like foci are disrupted at a position adjacent to the ICP4 nucleoprotein complexes and prereplicative foci [2]. The precise roles of Hsc70 and VICE domain formation in HSV infection are still a mystery, although the ATPase activity of Hsc70 is essential for efficient immediate early gene expression and replication-compartment formation [23] (C. Livingston and S. K. Weller, unpublished data). We have suggested that Hsc70 may participate in protein remodeling, nuclear transport of viral proteins, and assembly of multi-protein complexes [24]. VICE domains may function as storage sites for chaperones that can rapidly exchange with other sites in the nucleus to achieve protein remodeling. Sequestration of misfolded proteins in VICE domains may also be cytoprotective, and in this light it is of interest to note that they are reminiscent of intranuclear inclusion bodies that form in the presence of excess misfolded protein in some diseases. In cells from patients afflicted with Huntington's disease or spinal cerebellar ataxia, mutant huntingtin and ataxin-1 protein are sequestered, thereby avoiding cytotoxic events that could lead to the induction of apoptosis [27], [28]. Taken together, these results suggest that HSV has evolved to utilize the beneficial aspects of PQC machinery and avoid potentially negative pro-apoptotic consequences related to the presence of misfolded proteins [2], [21], [24].
HSV Also Manipulates the Cellular DNA Damage Response Machinery
Herpesviruses have also evolved a complex relationship with host DNA damage response pathways (reviewed in [29]–[31]). In response to DNA damage or replication stress, mammalian cells activate signal transduction pathways leading to repair, activation of cell cycle checkpoints, gene silencing or, if the damage is irreparable, induction of apoptosis. DNA damage is generally signaled by the activation of one or more damage-sensing PI3 kinase-like kinases: ATM, ATR, or DNA-PK. Interestingly, in cells infected with HSV-1, some aspects of the damage signaling pathways are activated and others are sabotaged. For instance, DNA-PK is degraded by ICP0, which may inactivate the nonhomologous end-joining pathway at least in some cell types [32]. Interestingly, ATR-mediated phosphorylation of downstream targets RPA and Chk1 is also suppressed [33]–[35]. Furthermore, although some ATM targets are phosphorylated during infection [33], [36]–[38], damage foci that would normally form after DNA damage are prevented due to the ability of ICP0 to degrade cellular histone ubiquitin ligases RNF8 and RNF168 [39]. The observation that ICP0 is responsible for inactivating some aspects of the host damage signaling machinery is consistent with the notion that these pathways may be intrinsically antiviral, resulting in genome silencing, induction of cell cycle checkpoints, or apoptosis. Despite the inactivation of some components of the DNA damage sensing pathways, several of the proteins involved in these pathways are recruited to replication compartments, possibly to aid in viral DNA replication (see below).
The Formation of Larger-than-Unit-Length Concatemers May Involve Recombination-Dependent Replication Using Viral and Host Proteins
Production of HSV concatemeric DNA is an essential step for the generation of progeny virus, as the packaging machinery must recognize longer-than-unit-length concatemers during encapsidation. Although it has been proposed that the viral genome circularizes and rolling circle replication leads to the formation of concatemers, several lines of evidence suggest that HSV DNA replication is more complex and may involve recombination-dependent replication reminiscent of bacteriophages lambda and T4 (reviewed in [30]). For instance, simple rolling circle replication does not explain the observation that genomic inversions occur as soon as viral DNA synthesis can be detected [40]–[42]. We suggest that HSV has evolved a novel mechanism of DNA replication utilizing viral and cellular proteins.
HSV-1 encodes an enzyme complex reminiscent of the two-subunit Red α/β recombinase encoded by bacteriophage lambda. The viral 5′ to 3′ alkaline exonuclease (UL12) and the single-strand binding protein (ICP8) interact with each other, are recruited to replication compartments, and are essential for efficient virus production ([43] and references within; [N. Balasubramanian and S. K. Weller, unpublished results). UL12 and ICP8 together mediate strand exchange in vitro, suggesting a role as a two-component recombinase reminiscent of the lambda Red α/β recombination system [43]. Alternatively, it is possible that UL12 and ICP8 function to regulate recombination or to process replication intermediates into a form that can be packaged into infectious virus. A role for cellular proteins in viral DNA replication has been suggested by the observation that several cellular factors involved in homologous repair and recombination (HRR) including Mre11, Rad50, Nbs1, and RAD51 are recruited to viral prereplicative sites and replication compartments [33], [36]–[38]. These and other HRR proteins appear to be important for efficient virus production, as mutant or knock down ATM, Mre11, Nbs1, and WRN cell lines exhibit significant defects in virus production [37], [38]. Interestingly, UL12 has recently been shown to interact specifically with the DNA damage sensing MRN complex [44], and Taylor and Knipe have reported that ICP8 interacts either directly or indirectly with 19 cellular proteins involved in DNA replication, repair or recombination [38]. Taken together, these data suggest that viral and cellular proteins collaborate to produce concatemers that can be encapsidated into infectious virions; however, direct proof of this model will require additional experimentation.
Conclusion: In Order to Create an Environment Conducive to Lytic Infection, HSV-1 Manipulates Several Cellular Homeostatic Pathways
During the earliest stages of viral infection, HSV transforms the cellular environment from one that is hostile to virus infection to one that supports virus growth. Work in this area has only scratched the surface in terms of defining the mechanisms by which ICP0 and other viral proteins manipulate cellular pathways and create environments that promote lytic infection. Elucidating how cellular homeostatic pathways limit viral infections will be important to our understanding of the delicate balance between lytic and latent infection and to aid in the development of new antiviral therapies.
Zdroje
1. QuinlanMP
ChenLB
KnipeDM
1984 The intranuclear location of a herpes simplex virus DNA-binding protein is determined by the status of viral DNA replication. Cell 36 857 868
2. LivingstonCM
DeLucaNA
WilkinsonDE
WellerSK
2008 Oligomerization of ICP4 and Rearrangement of Heat Shock Proteins May Be Important for Herpes Simplex Virus Type 1 Prereplicative Site Formation. J Virol 82 6324 6336
3. KuddusRH
DeLucaNA
2007 DNA-Dependent Oligomerization of Herpes Simplex Virus Type 1 Regulatory Protein ICP4. J Virol 81 9230 9237
4. Sandri-GoldinRM
2008 The many roles of the regulatory protein ICP27 during herpes simplex virus infection. Front Biosci 13 5241 5256
5. HagglundR
RoizmanB
2004 Role of ICP0 in the strategy of conquest of the host cell by herpes simplex virus 1. J Virol 78 2169 2178
6. SearsAE
HalliburtonIW
MeignierB
SilverS
RoizmanB
1985 Herpes simplex virus 1 mutant deleted in the alpha 22 gene: growth and gene expression in permissive and restrictive cells and establishment of latency in mice. J Virol 55 338 346
7. EverettRD
2000 ICP0, a regulator of herpes simplex virus during lytic and latent infection. BioEssays 22 761 770
8. PaladinoP
MossmanKL
2009 Mechanisms employed by herpes simplex virus 1 to inhibit the interferon response. J Interferon Cytokine Res 29 599 607
9. FraserKA
RiceSA
2007 Herpes simplex virus immediate-early protein ICP22 triggers loss of serine 2-phosphorylated RNA polymerase II. J Virol 81 5091 5101
10. de Bruyn KopsA
KnipeDM
1994 Preexisting nuclear architecture defines the intranuclear location of herpesvirus DNA replication structures. J Virol 68 3512 3526
11. LiptakL
UprichardS
KnipeD
1996 Functional order of assembly of herpes simplex virus DNA replication proteins into prereplicative site structures. J Virol 70 1759 1767
12. LukonisC
WellerS
1997 Formation of herpes simplex virus type 1 replication compartments by transfection: requirements and localization to nuclear domain 10. J Virol 71 2390 2399
13. BurkhamJ
CoenDM
WellerSK
1998 ND10 Protein PML Is Recruited to Herpes Simplex Virus Type 1 Prereplicative Sites and Replication Compartments in the Presence of Viral DNA Polymerase. J Virol 72 10100 10107
14. EverettRD
MurrayJ
2005 ND10 components relocate to sites associated with herpes simplex virus type 1 nucleoprotein complexes during virus infection. J Virol 79 5078 5089
15. EverettRD
FreemontP
SaitohH
DassoM
OrrA
1998 The disruption of ND10 during herpes simplex virus infection correlates with the Vmw110 - and proteasome-dependent loss of several PML isoforms. J Virol 72 6581 6591
16. TaylorTJ
McNameeEE
DayC
KnipeDM
2003 Herpes simplex virus replication compartments can form by coalescence of smaller compartments. Virology 309 232 247
17. MonierK
ArmasJC
EtteldorfS
GhazalP
SullivanKF
2000 Annexation of the interchromosomal space during viral infection. Nat Cell Biol 2 661 665
18. Simpson-HolleyM
ColgroveRC
NalepaG
HarperJW
KnipeDM
2005 Identification and functional evaluation of cellular and viral factors involved in the alteration of nuclear architecture during herpes simplex virus 1 infection. J Virol 79 12840 12851
19. BesseS
Puvion-DutilleulF
1996 Intranuclear retention of ribosomal RNAs in response to herpes simplex virus type 1 infection. J Cell Sci 109 Pt 1 119 129
20. ScottES
O'HareP
2001 Fate of the inner nuclear membrane protein lamin B receptor and nuclear lamins in herpes simplex virus type 1 infection. J Virol 75 8818 8830
21. BurchAD
WellerSK
2004 Nuclear sequestration of cellular chaperone and proteasomal machinery during herpes simplex virus type 1 infection. J Virol 78 7175 7185
22. MathewSS
Della SelvaMP
BurchAD
2009 Modification and reorganization of the cytoprotective cellular chaperone Hsp27 during herpes simplex virus type 1 infection. J Virol 83 9304 9312
23. LiL
JohnsonLA
Dai-JuJQ
Sandri-GoldinRM
2008 Hsc70 focus formation at the periphery of HSV-1 transcription sites requires ICP27. PLoS ONE 3 e1491 doi:10.1371/journal.pone.0001491
24. LivingstonCM
IfrimMF
CowanAE
WellerSK
2009 Virus-induced chaperone-enriched (VICE) domains function as nuclear protein quality control centers during HSV-1 infection. PLoS Pathog 5 e1000619 doi:10.1371/journal.ppat.1000619
25. JahediS
MarkovitzNS
FilatovF
RoizmanB
1999 Colocalization of the herpes simplex virus 1 UL4 protein with infected cell protein 22 in small, dense nuclear structures formed prior to onset of DNA synthesis. J Virol 73 5132 5138
26. BastianTW
LivingstonCM
WellerSK
RiceSA
2010 Herpes simplex virus type 1 immediate-early protein ICP22 is required for VICE domain formation during productive viral infection. J Virol 84 2384 2394
27. TanakaM
KimYM
LeeG
JunnE
IwatsuboT
2004 Aggresomes formed by alpha-synuclein and synphilin-1 are cytoprotective. J Biol Chem 279 4625 4631
28. TaylorJP
TanakaF
RobitschekJ
SandovalCM
TayeA
2003 Aggresomes protect cells by enhancing the degradation of toxic polyglutamine-containing protein. Hum Mol Genet 12 749 757
29. EverettRD
2006 Interactions between DNA viruses, ND10 and the DNA damage response. Cellular Microbiol 8 365 374
30. WilkinsonD
WellerS
2003 The Role of DNA Recombination in Herpes Simplex Virus DNA Replication. IUBMB Life 55 451 458
31. LilleyCE
ChaurushiyaMS
WeitzmanMD
2009 Chromatin at the intersection of viral infection and DNA damage. Biochim Biophys Acta 1799 319 327
32. ParkinsonJ
Lees-MillerSP
EverettRD
1999 Herpes simplex virus type 1 immediate-early protein vmw110 induces the proteasome-dependent degradation of the catalytic subunit of DNA-dependent protein kinase. J Virol 73 650 657
33. ShirataN
KudohA
DaikokuT
TatsumiY
FujitaM
2005 Activation of ataxia telangiectasia-mutated DNA damage checkpoint signal transduction elicited by herpes simplex virus infection. J Biol Chem 280 30336 30341
34. WilkinsonDE
WellerSK
2006 Herpes simplex virus type I disrupts the ATR-dependent DNA-damage response during lytic infection. J Cell Sci 119 2695 2703
35. MohniKN
LivingstonCM
CortezD
WellerSK
2010 ATR and ATRIP are recruited to Herpes Simplex Virus type 1 replication compartments even though ATR signaling is disabled. J Virol E-pub ahead of print 22 September 2010
36. WilkinsonDE
WellerSK
2004 Recruitment of cellular recombination and repair proteins to sites of herpes simplex virus type 1 DNA replication is dependent on the composition of viral proteins within prereplicative sites and correlates with the induction of the DNA damage response. J Virol 78 4783 4796
37. LilleyCE
CarsonCT
MuotriAR
GageFH
WeitzmanMD
2005 DNA repair proteins affect the lifecycle of herpes simplex virus 1. Proc Natl Acad Sci U S A 102 5844 5849
38. TaylorTJ
KnipeDM
2004 Proteomics of herpes simplex virus replication compartments: association of cellular dna replication, repair, recombination, and chromatin remodeling proteins with ICP8. J Virol 78 5856 5866
39. LilleyCE
ChaurushiyaMS
BoutellC
LandryS
SuhJ
2010 A viral E3 ligase targets RNF8 and RNF168 to control histone ubiquitination and DNA damage responses. EMBO J 29 943 9555
40. LambertiC
WellerSK
1996 The herpes simplex virus type 1 UL6 protein is essential for cleavage and packaging but not for genomic inversion. Virology 226 403 407
41. SeveriniA
ScrabaDG
TyrrellDL
1996 Branched structures in the intracellular DNA of herpes simplex virus type 1. J Virol 70 3169 3175
42. ZhangX
EfstathiouS
SimmonsA
1994 Identification of novel herpes simplex virus replicative intermediates by field inversion gel electrophoresis: implications for viral DNA amplification strategies. Virology 202 530 539
43. ReuvenNB
WillcoxS
GriffithJD
WellerSK
2004 Catalysis of strand exchange by the HSV-1 UL12 and ICP8 proteins: potent ICP8 recombinase activity is revealed upon resection of dsDNA substrate by nuclease. J Mol Biol 342 57 71
44. BalasubramanianN
BaiP
BuchekG
KorzaG
WellerSK
2010 Physical interaction between the herpes simplex virus type 1 exonuclease, UL12, and the DNA double strand break sensing MRN complex. J Virol E-pub ahead of print 13 October 2010
Štítky
Hygiena a epidemiológia Infekčné lekárstvo Laboratórium
Článek Tick Histamine Release Factor Is Critical for Engorgement and Transmission of the Lyme Disease AgentČlánek TGF-b2 Induction Regulates Invasiveness of -Transformed Leukocytes and Disease SusceptibilityČlánek The Origin of Intraspecific Variation of Virulence in an Eukaryotic Immune Suppressive Parasite
Článok vyšiel v časopisePLOS Pathogens
Najčítanejšie tento týždeň
2010 Číslo 11- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Očkování proti virové hemoragické horečce Ebola experimentální vakcínou rVSVDG-ZEBOV-GP
- Koronavirus hýbe světem: Víte jak se chránit a jak postupovat v případě podezření?
-
Všetky články tohto čísla
- Patients with Discordant Responses to Antiretroviral Therapy Have Impaired Killing of HIV-Infected T Cells
- A Molecular Mechanism for Eflornithine Resistance in African Trypanosomes
- Tyrosine Sulfation of the Amino Terminus of PSGL-1 Is Critical for Enterovirus 71 Infection
- Autoimmunity as a Predisposition for Infectious Diseases
- The Subtilisin-Like Protease AprV2 Is Required for Virulence and Uses a Novel Disulphide-Tethered Exosite to Bind Substrates
- Structural Analysis of HIV-1 Maturation Using Cryo-Electron Tomography
- Tick Histamine Release Factor Is Critical for Engorgement and Transmission of the Lyme Disease Agent
- Interferon-Inducible CXC Chemokines Directly Contribute to Host Defense against Inhalational Anthrax in a Murine Model of Infection
- TGF-b2 Induction Regulates Invasiveness of -Transformed Leukocytes and Disease Susceptibility
- The Origin of Intraspecific Variation of Virulence in an Eukaryotic Immune Suppressive Parasite
- CO Acts as a Signalling Molecule in Populations of the Fungal Pathogen
- SV2 Mediates Entry of Tetanus Neurotoxin into Central Neurons
- MAP Kinase Phosphatase-2 Plays a Critical Role in Response to Infection by
- Glycosylation Focuses Sequence Variation in the Influenza A Virus H1 Hemagglutinin Globular Domain
- Potentiation of Epithelial Innate Host Responses by Intercellular Communication
- Fcγ Receptor I Alpha Chain (CD64) Expression in Macrophages Is Critical for the Onset of Meningitis by K1
- ANK, a Host Cytoplasmic Receptor for the Cell-to-Cell Movement Protein, Facilitates Intercellular Transport through Plasmodesmata
- Analysis of the Initiating Events in HIV-1 Particle Assembly and Genome Packaging
- Evolution of Linked Avirulence Effectors in Is Affected by Genomic Environment and Exposure to Resistance Genes in Host Plants
- Structural Basis of HIV-1 Neutralization by Affinity Matured Fabs Directed against the Internal Trimeric Coiled-Coil of gp41
- Hepatitis C Virus (HCV) Evades NKG2D-Dependent NK Cell Responses through NS5A-Mediated Imbalance of Inflammatory Cytokines
- Host Cell Invasion and Virulence Mediated by Ssa1
- Global Gene Expression in Urine from Women with Urinary Tract Infection
- Should the Human Microbiome Be Considered When Developing Vaccines?
- HapX Positively and Negatively Regulates the Transcriptional Response to Iron Deprivation in
- Enhancing Oral Vaccine Potency by Targeting Intestinal M Cells
- Herpes Simplex Virus Reorganizes the Cellular DNA Repair and Protein Quality Control Machinery
- The Female Lower Genital Tract Is a Privileged Compartment with IL-10 Producing Dendritic Cells and Poor Th1 Immunity following Infection
- Zn Inhibits Coronavirus and Arterivirus RNA Polymerase Activity and Zinc Ionophores Block the Replication of These Viruses in Cell Culture
- Cryo Electron Tomography of Native HIV-1 Budding Sites
- Crystal Structure and Size-Dependent Neutralization Properties of HK20, a Human Monoclonal Antibody Binding to the Highly Conserved Heptad Repeat 1 of gp41
- Modelling the Evolution and Spread of HIV Immune Escape Mutants
- The Arabidopsis Resistance-Like Gene Is Activated by Mutations in and Contributes to Resistance to the Bacterial Effector AvrRps4
- Platelet-Activating Factor Receptor Plays a Role in Lung Injury and Death Caused by Influenza A in Mice
- Genetic and Structural Basis for Selection of a Ubiquitous T Cell Receptor Deployed in Epstein-Barr Virus Infection
- Ubiquitin-Regulated Nuclear-Cytoplasmic Trafficking of the Nipah Virus Matrix Protein Is Important for Viral Budding
- Pneumolysin Activates the NLRP3 Inflammasome and Promotes Proinflammatory Cytokines Independently of TLR4
- Immune Evasion by : Differential Targeting of Dendritic Cell Subpopulations
- Survival in Selective Sand Fly Vector Requires a Specific -Encoded Lipophosphoglycan Galactosylation Pattern
- PLOS Pathogens
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Zn Inhibits Coronavirus and Arterivirus RNA Polymerase Activity and Zinc Ionophores Block the Replication of These Viruses in Cell Culture
- The Female Lower Genital Tract Is a Privileged Compartment with IL-10 Producing Dendritic Cells and Poor Th1 Immunity following Infection
- Crystal Structure and Size-Dependent Neutralization Properties of HK20, a Human Monoclonal Antibody Binding to the Highly Conserved Heptad Repeat 1 of gp41
- The Arabidopsis Resistance-Like Gene Is Activated by Mutations in and Contributes to Resistance to the Bacterial Effector AvrRps4
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



