-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
Species Complex: Ecology, Phylogeny, Sexual Reproduction, and Virulence
article has not abstract
Published in the journal: Species Complex: Ecology, Phylogeny, Sexual Reproduction, and Virulence. PLoS Pathog 10(10): e32767. doi:10.1371/journal.ppat.1004397
Category: Pearls
doi: https://doi.org/10.1371/journal.ppat.1004397Summary
article has not abstract
The Paracoccidioides Genus and Paracoccidioidomycosis
Paracoccidioidomycosis (PCM) is a deep systemic mycosis caused by human fungal pathogens of the Paracoccidioides genus. The disease is geographically restricted to subtropical areas of Latin America (from south of Mexico to north of Argentina) with a high prevalence in Brazil, Colombia, Venezuela, and Argentina [1]. The annual incidence rate in Brazil is 10–30 infections per million inhabitants, and the mean mortality rate is 1.4 per million inhabitants per year, making this disease the highest cause of mortality among systemic mycoses [2]. PCM is endemic in rural populations and mainly affects individuals engaged in agricultural activities, who inhale aerosols containing fungal material during manipulation of the soil.
Molecular evolutionary studies place the genus Paracoccidioides in the thermodimorphic fungal pathogen clade related to the family Ajellomycetaceae (Ascomycetes), which includes the Blastomyces, Histoplasma, and Emmonsia genera, and with which it shares a common ancestor, Lacazia loboi. PCM can be caused by two species Paracoccidioides brasiliensis and P. lutzii [3]. P. brasiliensis has been considered a single species since its discovery, although several studies including molecular and morphological data support the split of P. brasiliensis into two species [3], [4]. P. lutzii is composed of a single monophyletic and recombining population so far found in central, southwest, and north Brazil and Ecuador [3]–[5]. On the other hand, P. brasiliensis contains a complex of at least four different cryptic species (S1, PS2, PS3 and PS4; Figure 1A [6]). P. brasiliensis S1 represents a monophyletic and recombining population widely distributed in South America and has been associated with the majority of cases of PCM detected up until the present time. Strains belonging to P. brasiliensis S1 have previously been recovered from armadillos, soil, and penguin feces [6]. P. brasiliensis PS2 is a paraphyletic and recombining population identified so far only in Brazil and Venezuela [6]. P. brasiliensis PS3 is comprised of a monophyletic and clonal population that has been recovered in humans and armadillos in endemic regions of Colombia [6]. P. brasiliensis PS4 was recently identified and is composed of a monophyletic population of clinical isolates from Venezuela [5], [7]. Besides the typical bicorn cocked hat – and barrel-shaped conidia produced by both species, P. lutzii frequently produces elongated rod-shaped conidia, a characteristic feature that may be used for species identification [3]. Because of the difficulties of conidia production in the laboratory and slight morphological differences among species, molecular identification of Paracoccidioides species has become the most common tool of choice. Several molecular markers have already been applied in population studies of the Pararacoccidioides genus, and for multilocus sequencing typing, gp43, arf, β-tub, and hsp70 loci are the best choices for species delineation [4], [6].
Fig. 1. 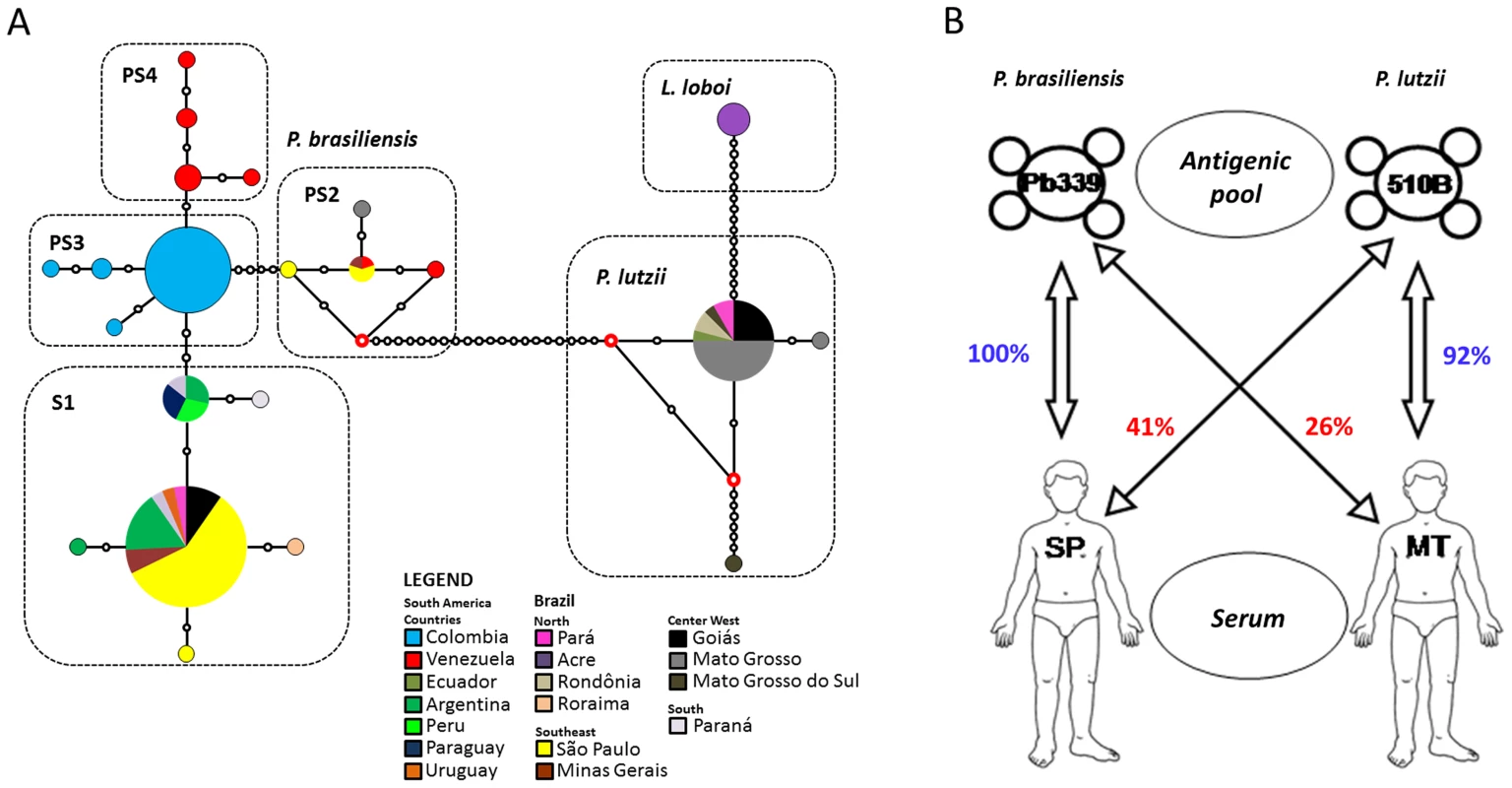
(A) Median-joining haplotypic network distribution of the <i>Paracoccidioides</i> genus and <i>L. loboi</i> based on the <i>gp43</i> marker. The size of the circumference is proportional to the haplotype frequency, and colors vary according to the sampling location of each haplotype. Red dots (median vectors) are hypothetical missing intermediates, and black dots represent each mutation site. (B) Schematic representation of serum/antigen compatibility among patients and isolates from Mato Grosso and São Paulo used in serological tests. The illustration shows the low immunogenic specificity when serum and antigenic pool from different states are reacted <em class="ref">[41]</em>. Phylogeography of the Paracoccidioides Genus
The finding of the cryptic species in the genus Paracoccidioides has led us to explore the evolutionary mechanisms that were responsible for the current geographic distribution of its five phylogenetic species (S1, PS2, PS3, PS4, and P. lutzii). Phylogeographic inferences from three different loci revealed simultaneous geographic expansions of S1 isolates. This represents a dispersal by distance, leading to a nonsexual population (PS3) that does not produce any gene flow between other species, and a long-distance colonization or a fragmentation resulting in the separation of Venezuelan PS4 species [3]–[5], [7]. The dispersal event that resulted in PS3 has been confirmed, with the remaining divergence processes due to vicariance events. Despite the great stability of the Guiana and Brazilian shields, the uplift of the Andes and episodic marine inclusions 61 million years ago, 20 million years ago, and 11.8–7.0 million years ago may have favored vicariance between Paracoccidioides and Lacazia. This may have occurred by creating new and empty ecological niches represented by wetlands and/or totally submerged areas, as well as by the simultaneous emergence of riverine Cetacean mammals. The recent dispersal of PS3 to Colombia may also be explained by the complete submersion of Colombian territory by the Pebas/Solimões lake, derived from marine incursions in the late Miocene era [8] and indicating a recent occupation. As there are no clear geographic barriers, the most challenging task is to explain the speciation processes that have given rise to S1, PS2, and P. lutzii in the very stable Brazilian shield. Indeed, the prevalence of P. lutzii in central-western Brazil and its relatively close proximity to the S1 and PS2 occurrence areas suggests a parapatric speciation. With regards to the divergence between S1 and PS2 and the current sympatry observed between them, probable differences in preferences for substrates and resources in their saprobe lifestyle that might have triggered a disruptive selection should be considered [5]. However, more studies are required to complete the biogeographical puzzle of the Paracoccidioides genus. These include mapping of the cryptic species in environmental, rather than clinical, samples, as well as searching for saprobe differences between sympatric species.
Ecology of P. brasiliensis and P. lutzii
Onygenalean (Ascomycota) organisms have typically evolved by adapting to two distinct ecological niches, the first represented by saprobic conditions in soil and the second by the live tissues of animal hosts. Genomic adaptations such as loss of carbohydrate-degrading enzymes, gain of proteases, and the ability to produce infective conidia allowing long association with the mammalian host are more adapted to a biotrophic lifestyle [9], [10]. Epidemiological evidence indicates that the saprobic forms of P. brasiliensis and P. lutzii may occur in some restricted and/or protected soil conditions, in places containing natural and anthropic disturbed vegetation near water sources [11]. Isolation of P. brasiliensis directly from its saprobic form has proved to be difficult. However, the fungus has been repeatedly cultured from the armadillo species Dasypus novemcinctus and Cabassous centralis in endemic PCM areas [12], [13] and, in unique cases, from dogs and two-toed sloths [14], [15]. Additional evidence of the infection of several wild and domestic animals has also been provided by intradermal, serological, histopathological, and molecular tests, revealing a broad distribution and adaptation to mammalian hosts [16].
Although outbreaks of PCM have not been documented, the geographical distribution of the disease is heterogeneous and is associated with moderate-to-high precipitation rates, mild temperatures, and fertile soils. The disease occurs in areas such as the central-western and northern regions of Brazil where agricultural activities are more commonly employed. Climatic anomalies, such as those triggered by the 1982–1983 El Niño event, have been associated with an excess of acute PCM cases when compared with the number of expected cases for the same period. This indicates the presence of a temporal cluster of the disease in the state of São Paulo, Brazil, occurring in the year 1985 [17]. Climatic conditions resulting in an atypical increase in soil water storage in 1982–1983 and in an increase in the absolute air humidity in 1984 may have contributed firstly to fungus growth and then to conidial dispersal. This evidently follows the “grow and blow” model already proposed for coccidioidomycosis outbreaks [18]. Experimental studies have indicated that the several genetic groups or cryptic species of P. brasiliensis have different abilities in producing the infective conidia, and this may in turn produce differential rates of infection. For example, the S1 and PS2 sympatric cryptic species of P. brasiliensis occur at a disproportional rate of approximately 9∶1 in both patient and armadillo isolates. At the same time, isolates of the S1 genotype produce many more conidia than PS2 isolates [5].
Reproductive Modes in the Paracoccidioides Species Complex
Paracoccidioides was considered an asexual and clonal microorganism for many years [1]. The anamorph of Paracoccidioides is characterized by multiple budding yeast cells that grow at 37°C in mammalian tissues or by mycelia that produce chlamydospores or conidia at 25°C in the environment. Recently, population genetics and comparative genomic studies have provided evidence for different breeding strategies in the Paracoccidioides genus. Recombination events were detected in both P. lutzii and P. brasiliensis (S1 and PS2), and the P. brasiliensis PS3 population was considered clonal [4], [6]. The mating type locus was identified in the three sequenced genomes, and a single copy of MAT1-1 or MAT1-2 was found, thus suggesting a bipolar mating system [9]. According to Torres et al. [19], MAT gene distribution was evaluated with regards to the country of origin and phylogenetic species, revealing a 1∶1 ratio of MAT1-1 and MAT1-2. Additionally, Teixeira et al. [20] tested the MAT gene distribution in 98 Paracoccidioides isolates and revealed a slight (2-fold) prevalence of the MAT1-2 idiomorph. Unexpectedly, both the MAT1-1 and MAT1-2 genes were identified in 13 of the clinical isolates, suggesting that homothallism may exist in the Paracoccidioides genus.
Orthologs of mating and meiotic regulators that have been well characterized in a wide range of fungi were found by comparative genomics to be highly conserved in Paracoccidioides and other Ajellomycetacean sexual fungi [9], [20]. The biological functionality of α-pheromone and its receptor was elucidated using heterologous expression of these Paracoccidioides genes in the corresponding Saccharomyces cerevisiae null mutants [21]. Features related to sexual reproduction, such as coiled constricted hyphae and knob-like structures, were observed in Paracoccidioides species, indicating the formation of young ascocarps. In addition, multiple nuclei were found in coiled constricted hyphae, possibly as a consequence of nuclear migration during mating. Unfortunately, no cleistothecium or ascus production has so far been detected. The presence and expression of sexual machinery as well the ability to produce sex-related structures indicates that mating may occur in the Paracoccidioides life cycle.
Virulence Factors Associated with Dimorphism and Host Adaptation
The most probable environmental habitat of mycelial-phase Paracoccidioides is the soil. Once conidia or mycelia fragments are inhaled into the lung alveoli, the fungus shifts its morphology to a yeast phase because of temperature, hormones, and immune response. This step is crucial for the survival and maintenance of Paracoccidioides and other dimorphic fungi in hosts. The dimorphic transition promotes changes in the cell wall composition and carbohydrate polymer structure. Additionally, the presence of an outermost layer of α-1,3-glucan in the P. brasiliensis yeast cell wall has been proposed as a protective shield against host defense [22], [23].
The yeast transcriptional profile indicating the diversion of pyruvate from the glycolytic pathway into the glyoxylate cycle is consistent with a lower oxygen level in infected tissues [24]. Stress adaptations that induce genes encoding molecular chaperones, such as heat shock proteins (HSPs), are a common trait of Paracoccidioides during dimorphism and exposition to different host niches. These adaptations may be indispensable for fungal virulence upon infection [25]. In contrast, some HSP genes are down-regulated when mycelia cells are incubated in vitro with estradiol or human female serum, in some part explaining the predominance of the disease in adult males [26]. The ability of fungal cells to adhere to host cells is also vital for the initial steps of the infection process. Phospholipase B, involved in the fungus-macrophage interaction, and PbHad32p, a hydrolase involved in adherence to host cells, have both been proposed to be important for the initial steps of the virulence process [27], [28]. The genes encoding glyceraldehyde-3-phosphate-dehydrogenase (GAPDH), identified as a cell wall–associated host-adhesion molecule in Paracoccidioides, and enolase, which binds to fibronectin, have also been linked to the initial adhesion of the fungus to lung epithelial cells and alveolar macrophages upon infection [29]. New strategies for gene function studies using antisense RNA technology have recently emerged, overcoming problems with gene knockout due to multinucleated cells and unstable transformants [30]. The depletion of P. brasiliensis–encoding genes Cdc42 [31], AOX [32], Gp43 [33], SCONC [34], HSP90 [35], P27 [36], and Rbt5 [37] contributed significantly to the elucidation of host-pathogen interaction, pathogen resistance, and virulence in those species.
In years to come, more information on virulence processes will emerge from the accumulated data from transcriptome and complete genome releases at our disposal. This will pave the way for the identification of possible mechanisms to control the initial steps of infection which, when available to clinicians, will benefit patients.
Paracoccidioides Species Complex and Its Impact on Clinical and Serological Aspects of PCM
Since the discovery of the cryptic speciation in the genus Paracoccidioides, some important regional features of the disease were discussed regarding its impact on the current statement of PCM. The acute or subacute ( juvenile PCM) and chronic (adult PCM) are the two main forms of the disease; however, the presentation and course of the disease may vary from case to case [16]. The first discrimination between the PCM pathology related to geographical origin was observed by Barbosa et al. [38], in which patients from the central region of Brazil had predominantly lymphoabdominal forms not shared by patients from the south and southeast, a finding later confirmed by Andrade [39]. Is there a possibility that lymphoabdominal forms are associated with the pathology caused by P. lutzii and not by P. brasiliensis? Are there different pathologies caused by different species of Paracoccidioides? This possibility should not be ruled out and must be fully investigated in order to verify possible associations of cryptic species and different clinical manifestations of PCM. In addition to clinical manifestation, issues addressed to treatment have been raised by Hahn et al. [40] who found that patients infected with P. lutzii had good responses to trimethoprim-sulfamethoxazole while those infected with P. brasiliensis relapsed with the same drug administration. Moreover, there are known to be a high number of patients coming from the north-central region of Brazil with PCM with low or no immunoreactivity [41], [42]. Immunodiffusion tests with antigens produced by isolates from São Paulo (P. brasiliensis strain 339) crossed with sera from patients of Mato Grosso have low positivity (Figure 1B). Recently, serological tests confirm that sera from patients with PCM due to P. lutzii are able to recognize cell-free antigens from P. lutzii; however, sera from patients with PCM due to P. brasiliensis could not recognize any P. lutzii antigens [43]. Undoubtedly, these issues are critical and need an urgent mobilization to improve the methods of diagnosis and therapy that can specifically detect and effectively combat the Paracoccidioides species of a given patient with PCM.
Zdroje
1. San-BlasG, Nino-VegaG, IturriagaT (2002) Paracoccidioides brasiliensis and paracoccidioidomycosis: molecular approaches to morphogenesis, diagnosis, epidemiology, taxonomy and genetics. Med Mycol 40 : 225–242.
2. CoutinhoZF, SilvaD, LazeraM, PetriV, OliveiraRM, et al. (2002) Paracoccidioidomycosis mortality in Brazil (1980–1995). Cad Saude Publica 18 : 1441–1454.
3. TeixeiraMD, TheodoroRC, OliveiraFF, MachadoGC, HahnRC, et al. (2013) Paracoccidioides lutzii sp. nov.: biological and clinical implications. Med Mycol 52 : 19–28.
4. TeixeiraMM, TheodoroRC, de CarvalhoMJ, FernandesL, PaesHC, et al. (2009) Phylogenetic analysis reveals a high level of speciation in the Paracoccidioides genus. Mol Phylogenet Evol 52 : 273–283.
5. TheodoroRC, Teixeira MdeM, FelipeMS, Paduan KdosS, RibollaPM, et al. (2012) Genus paracoccidioides: Species recognition and biogeographic aspects. PLoS ONE 7: e37694.
6. MatuteDR, McEwenJG, PucciaR, MontesBA, San-BlasG, et al. (2006) Cryptic speciation and recombination in the fungus Paracoccidioides brasiliensis as revealed by gene genealogies. Mol Biol Evol 23 : 65–73.
7. Salgado-SalazarC, JonesLR, RestrepoA, McEwenJG (2010) The human fungal pathogen Paracoccidioides brasiliensis (Onygenales: Ajellomycetaceae) is a complex of two species: phylogenetic evidence from five mitochondrial markers. Cladistics 26 : 12.
8. WesselinghFP, SaloJA (2006) A Miocene perspective on the evolution of the Amazonian biota. Scripta Geol 133 : 19.
9. DesjardinsCA, ChampionMD, HolderJW, MuszewskaA, GoldbergJ, et al. (2011) Comparative genomic analysis of human fungal pathogens causing paracoccidioidomycosis. PLoS Genet 7: e1002345.
10. SharptonTJ, StajichJE, RounsleySD, GardnerMJ, WortmanJR, et al. (2009) Comparative genomic analyses of the human fungal pathogens Coccidioides and their relatives. Genome Res 19 : 1722–1731.
11. RestrepoA, McEwenJG, CastanedaE (2001) The habitat of Paracoccidioides brasiliensis: how far from solving the riddle? Med Mycol 39 : 233–241.
12. BagagliE, SanoA, CoelhoKI, AlquatiS, MiyajiM, et al. (1998) Isolation of Paracoccidioides brasiliensis from armadillos (Dasypus noveminctus) captured in an endemic area of paracoccidioidomycosis. Am J Trop Med Hyg 58 : 505–512.
13. CorredorGG, CastanoJH, PeraltaLA, DiezS, ArangoM, et al. (1999) Isolation of Paracoccidioides brasiliensis from the nine-banded armadillo Dasypus novemcinctus, in an endemic area for paracoccidioidomycosis in Colombia. Rev Iberoam Micol 16 : 216–220.
14. de FariasMR, CondasLA, RibeiroMG, Bosco SdeM, MuroMD, et al. (2011) Paracoccidioidomycosis in a dog: case report of generalized lymphadenomegaly. Mycopathologia 172 : 147–152.
15. Trejo-ChavezA, Ramirez-RomeroR, Ancer-RodriguezJ, Nevarez-GarzaAM, Rodriguez-TovarLE (2011) Disseminated paracoccidioidomycosis in a Southern two-toed sloth (Choloepus didactylus). J Comp Pathol 144 : 231–234.
16. BoccaAL, AmaralAC, TeixeiraMM, SatoPK, Shikanai-YasudaMA, et al. (2013) Paracoccidioidomycosis: eco-epidemiology, taxonomy and clinical and therapeutic issues. Future Microbiol 8 : 1177–1191.
17. BarrozoLV, BenardG, SilvaME, BagagliE, MarquesSA, et al. (2010) First description of a cluster of acute/subacute paracoccidioidomycosis cases and its association with a climatic anomaly. PLoS Negl Trop Dis 4: e643.
18. TameriusJD, ComrieAC (2011) Coccidioidomycosis incidence in Arizona predicted by seasonal precipitation. PLoS ONE 6: e21009.
19. TorresI, GarciaAM, HernandezO, GonzalezA, McEwenJG, et al. (2010) Presence and expression of the mating type locus in Paracoccidioides brasiliensis isolates. Fungal Genet Biol 47 : 373–380.
20. Teixeira MdeM, TheodoroRC, Derengowski LdaS, NicolaAM, BagagliE, et al. (2013) Molecular and morphological data support the existence of a sexual cycle in species of the genus Paracoccidioides. Eukaryot Cell 12 : 380–389.
21. Gomes-RezendeJA, Gomes-AlvesAG, MeninoJF, CoelhoMA, LudovicoP, et al. (2012) Functionality of the Paracoccidioides mating alpha-pheromone-receptor system. PLoS ONE 7: e47033.
22. RappleyeCA, GoldmanWE (2006) Defining virulence genes in the dimorphic fungi. Annu Rev Microbiol 60 : 281–303.
23. San-BlasG, San-BlasF, SerranoLE (1977) Host-parasite relationships in the yeastlike form of Paracoccidioides brasiliensis strain IVIC Pb9. Infect Immun 15 : 343–346.
24. DerengowskiLS, TavaresAH, SilvaS, ProcopioLS, FelipeMS, et al. (2008) Upregulation of glyoxylate cycle genes upon Paracoccidioides brasiliensis internalization by murine macrophages and in vitro nutritional stress condition. Med Mycol 46 : 125–134.
25. FelipeMS, TorresFA, MaranhaoAQ, Silva-PereiraI, Pocas-FonsecaMJ, et al. (2005) Functional genome of the human pathogenic fungus Paracoccidioides brasiliensis. FEMS Immunol Med Microbiol 45 : 369–381.
26. ShankarJ, WuTD, ClemonsKV, MonteiroJP, MirelsLF, et al. (2011) Influence of 17beta-estradiol on gene expression of Paracoccidioides during mycelia-to-yeast transition. PLoS ONE 6: e28402.
27. SoaresDA, de AndradeRV, SilvaSS, BoccaAL, Soares FelipeSM, et al. (2010) Extracellular Paracoccidioides brasiliensis phospholipase B involvement in alveolar macrophage interaction. BMC Microbiol 10 : 241.
28. HernandezO, AlmeidaAJ, TamayoD, TorresI, GarciaAM, et al. (2012) The hydrolase PbHAD32 participates in the adherence of Paracoccidioides brasiliensis conidia to epithelial lung cells. Med Mycol 50 : 533–537.
29. BarbosaMS, BaoSN, AndreottiPF, de FariaFP, FelipeMS, et al. (2006) Glyceraldehyde-3-phosphate dehydrogenase of Paracoccidioides brasiliensis is a cell surface protein involved in fungal adhesion to extracellular matrix proteins and interaction with cells. Infect Immun 74 : 382–389.
30. MeninoJF, AlmeidaAJ, RodriguesF (2012) Gene knockdown in Paracoccidioides brasiliensis using antisense RNA. Methods Mol Biol 845 : 187–198.
31. AlmeidaAJ, CunhaC, CarmonaJA, Sampaio-MarquesB, CarvalhoA, et al. (2009) Cdc42p controls yeast-cell shape and virulence of Paracoccidioides brasiliensis. Fungal Genet Biol 46 : 919–926.
32. RuizOH, GonzalezA, AlmeidaAJ, TamayoD, GarciaAM, et al. (2011) Alternative oxidase mediates pathogen resistance in Paracoccidioides brasiliensis infection. PLoS Negl Trop Dis 5: e1353.
33. TorresI, HernandezO, TamayoD, MunozJF, LeitaoNPJr, et al. (2013) Inhibition of PbGP43 expression may suggest that gp43 is a virulence factor in Paracoccidioides brasiliensis. PLoS ONE 8: e68434.
34. MeninoJF, SaraivaM, Gomes-RezendeJ, SturmeM, PedrosaJ, et al. (2013) P. brasiliensis virulence is affected by SconC, the negative regulator of inorganic sulfur assimilation. PLoS ONE 8: e74725.
35. TamayoD, MunozJF, TorresI, AlmeidaAJ, RestrepoA, et al. (2013) Involvement of the 90 kDa heat shock protein during adaptation of Paracoccidioides brasiliensis to different environmental conditions. Fungal Genet Biol 51 : 34–41.
36. TorresI, HernandezO, TamayoD, MunozJF, GarciaAM, et al. (2014) Paracoccidioides brasiliensis PbP27 gene: knockdown procedures and functional characterization. FEMS Yeast Res 14 : 270–280.
37. BailaoEF, ParenteJA, PigossoLL, de CastroKP, FonsecaFL, et al. (2014) Hemoglobin uptake by Paracoccidioides spp. is receptor-mediated. PLoS Negl Trop Dis 8: e2856.
38. BarbosaW, DaherR, OliveiraAR (1968) Lymphatic abdominal forms of South American blastomycosis. Rev InstMed Trop São Paulo 10 : 12.
39. AndradeALSS (1983) Paracoccidoidomycosis. The contribution to the study of lymphatic-abdominal form. Rev Patol Trop 12 : 91.
40. HahnRC, MacedoAM, FontesCJ, BatistaRD, SantosNL, et al. (2003) Randomly amplified polymorphic DNA as a valuable tool for epidemiological studies of Paracoccidioides brasiliensis. J Clin Microbiol 41 : 2849–2854.
41. BatistaJJr, de CamargoZP, FernandesGF, VicentiniAP, FontesCJ, et al. (2010) Is the geographical origin of a Paracoccidioides brasiliensis isolate important for antigen production for regional diagnosis of paracoccidioidomycosis? Mycoses 53 : 176–180.
42. Queiroz JuniorLD, de CamargoZP, TadanoT, RodriguesAM, TakararaDT, et al. (2014) Serological and antigenic profiles of clinical isolates of Paracoccidioides spp. from Central Western Brazil. Mycoses 57 : 466–72.
43. GegembauerG, AraujoLM, PereiraEF, RodriguesAM, PaniagoAM, et al. (2014) Serology of Paracoccidioidomycosis Due to Paracoccidioides lutzii. PLoS Negl Trop Dis 8: e2986.
Štítky
Hygiena a epidemiológia Infekčné lekárstvo Laboratórium
Článek Identification of the Microsporidian as a New Target of the IFNγ-Inducible IRG Resistance SystemČlánek Human Cytomegalovirus Drives Epigenetic Imprinting of the Locus in NKG2C Natural Killer CellsČlánek APOBEC3D and APOBEC3F Potently Promote HIV-1 Diversification and Evolution in Humanized Mouse ModelČlánek Role of Non-conventional T Lymphocytes in Respiratory Infections: The Case of the Pneumococcus
Článok vyšiel v časopisePLOS Pathogens
Najčítanejšie tento týždeň
2014 Číslo 10- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Očkování proti virové hemoragické horečce Ebola experimentální vakcínou rVSVDG-ZEBOV-GP
- Koronavirus hýbe světem: Víte jak se chránit a jak postupovat v případě podezření?
-
Všetky články tohto čísla
- Theory and Empiricism in Virulence Evolution
- -Related Fungi and Reptiles: A Fatal Attraction
- Adaptive Prediction As a Strategy in Microbial Infections
- Antimicrobials, Stress and Mutagenesis
- A Novel Function of Human Pumilio Proteins in Cytoplasmic Sensing of Viral Infection
- Social Motility of African Trypanosomes Is a Property of a Distinct Life-Cycle Stage That Occurs Early in Tsetse Fly Transmission
- Autophagy Controls BCG-Induced Trained Immunity and the Response to Intravesical BCG Therapy for Bladder Cancer
- Identification of the Microsporidian as a New Target of the IFNγ-Inducible IRG Resistance System
- mRNA Structural Constraints on EBNA1 Synthesis Impact on Antigen Presentation and Early Priming of CD8 T Cells
- Infection Causes Distinct Epigenetic DNA Methylation Changes in Host Macrophages
- Neutrophil Crawling in Capillaries; A Novel Immune Response to
- Live Attenuated Vaccine Protects against Pulmonary Challenge in Rats and Non-human Primates
- The ESAT-6 Protein of Interacts with Beta-2-Microglobulin (β2M) Affecting Antigen Presentation Function of Macrophage
- Characterization of Uncultivable Bat Influenza Virus Using a Replicative Synthetic Virus
- HIV Acquisition Is Associated with Increased Antimicrobial Peptides and Reduced HIV Neutralizing IgA in the Foreskin Prepuce of Uncircumcised Men
- Uses a Unique Ligand-Binding Mode for Trapping Opines and Acquiring A Competitive Advantage in the Niche Construction on Plant Host
- Involvement of a 1-Cys Peroxiredoxin in Bacterial Virulence
- Ethanol Stimulates WspR-Controlled Biofilm Formation as Part of a Cyclic Relationship Involving Phenazines
- Densovirus Is a Mutualistic Symbiont of a Global Crop Pest () and Protects against a Baculovirus and Bt Biopesticide
- Insights into Intestinal Colonization from Monitoring Fluorescently Labeled Bacteria
- Mycobacterial Antigen Driven Activation of CD14CD16 Monocytes Is a Predictor of Tuberculosis-Associated Immune Reconstitution Inflammatory Syndrome
- Lipoprotein LprG Binds Lipoarabinomannan and Determines Its Cell Envelope Localization to Control Phagolysosomal Fusion
- Dampens the DNA Damage Response
- MicroRNAs Suppress NB Domain Genes in Tomato That Confer Resistance to
- Novel Cyclic di-GMP Effectors of the YajQ Protein Family Control Bacterial Virulence
- Vaginal Challenge with an SIV-Based Dual Reporter System Reveals That Infection Can Occur throughout the Upper and Lower Female Reproductive Tract
- Detecting Differential Transmissibilities That Affect the Size of Self-Limited Outbreaks
- One Small Step for a Yeast - Microevolution within Macrophages Renders Hypervirulent Due to a Single Point Mutation
- Expression Profiling during Arabidopsis/Downy Mildew Interaction Reveals a Highly-Expressed Effector That Attenuates Responses to Salicylic Acid
- Human Cytomegalovirus Drives Epigenetic Imprinting of the Locus in NKG2C Natural Killer Cells
- Interaction with Tsg101 Is Necessary for the Efficient Transport and Release of Nucleocapsids in Marburg Virus-Infected Cells
- The N-Terminus of Murine Leukaemia Virus p12 Protein Is Required for Mature Core Stability
- Sterol Biosynthesis Is Required for Heat Resistance but Not Extracellular Survival in
- Allele-Specific Induction of IL-1β Expression by C/EBPβ and PU.1 Contributes to Increased Tuberculosis Susceptibility
- Host Cofactors and Pharmacologic Ligands Share an Essential Interface in HIV-1 Capsid That Is Lost upon Disassembly
- APOBEC3D and APOBEC3F Potently Promote HIV-1 Diversification and Evolution in Humanized Mouse Model
- Structural Basis for the Recognition of Human Cytomegalovirus Glycoprotein B by a Neutralizing Human Antibody
- Systematic Analysis of ZnCys Transcription Factors Required for Development and Pathogenicity by High-Throughput Gene Knockout in the Rice Blast Fungus
- Epstein-Barr Virus Nuclear Antigen 3A Promotes Cellular Proliferation by Repression of the Cyclin-Dependent Kinase Inhibitor p21WAF1/CIP1
- The Host Protein Calprotectin Modulates the Type IV Secretion System via Zinc Sequestration
- Cyclophilin A Associates with Enterovirus-71 Virus Capsid and Plays an Essential Role in Viral Infection as an Uncoating Regulator
- A Novel Alpha Kinase EhAK1 Phosphorylates Actin and Regulates Phagocytosis in
- The pH-Responsive PacC Transcription Factor of Governs Epithelial Entry and Tissue Invasion during Pulmonary Aspergillosis
- Sensing of Immature Particles Produced by Dengue Virus Infected Cells Induces an Antiviral Response by Plasmacytoid Dendritic Cells
- Co-opted Oxysterol-Binding ORP and VAP Proteins Channel Sterols to RNA Virus Replication Sites via Membrane Contact Sites
- Characteristics of Memory B Cells Elicited by a Highly Efficacious HPV Vaccine in Subjects with No Pre-existing Immunity
- HPV16-E7 Expression in Squamous Epithelium Creates a Local Immune Suppressive Environment via CCL2- and CCL5- Mediated Recruitment of Mast Cells
- Dengue Viruses Are Enhanced by Distinct Populations of Serotype Cross-Reactive Antibodies in Human Immune Sera
- CD4 Depletion in SIV-Infected Macaques Results in Macrophage and Microglia Infection with Rapid Turnover of Infected Cells
- A Sialic Acid Binding Site in a Human Picornavirus
- Contact Heterogeneity, Rather Than Transmission Efficiency, Limits the Emergence and Spread of Canine Influenza Virus
- Myosins VIII and XI Play Distinct Roles in Reproduction and Transport of
- HTLV-1 Tax Stabilizes MCL-1 via TRAF6-Dependent K63-Linked Polyubiquitination to Promote Cell Survival and Transformation
- Species Complex: Ecology, Phylogeny, Sexual Reproduction, and Virulence
- A Critical Role for IL-17RB Signaling in HTLV-1 Tax-Induced NF-κB Activation and T-Cell Transformation
- Exosomes from Hepatitis C Infected Patients Transmit HCV Infection and Contain Replication Competent Viral RNA in Complex with Ago2-miR122-HSP90
- Role of Non-conventional T Lymphocytes in Respiratory Infections: The Case of the Pneumococcus
- Kaposi's Sarcoma-Associated Herpesvirus Induces Nrf2 during Infection of Endothelial Cells to Create a Microenvironment Conducive to Infection
- A Relay Network of Extracellular Heme-Binding Proteins Drives Iron Acquisition from Hemoglobin
- Glutamate Secretion and Metabotropic Glutamate Receptor 1 Expression during Kaposi's Sarcoma-Associated Herpesvirus Infection Promotes Cell Proliferation
- Reduces Malaria and Dengue Infection in Vector Mosquitoes and Has Entomopathogenic and Anti-pathogen Activities
- PLOS Pathogens
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Novel Cyclic di-GMP Effectors of the YajQ Protein Family Control Bacterial Virulence
- MicroRNAs Suppress NB Domain Genes in Tomato That Confer Resistance to
- The ESAT-6 Protein of Interacts with Beta-2-Microglobulin (β2M) Affecting Antigen Presentation Function of Macrophage
- Characterization of Uncultivable Bat Influenza Virus Using a Replicative Synthetic Virus
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



