-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
HPV16 Down-Regulates the Insulin-Like Growth Factor Binding Protein 2 to Promote Epithelial Invasion in Organotypic Cultures
The human papillomaviruses (HPV) are the etiological agents of cervical cancer and the disease progresses through the pre-malignant phases of cervical intraepithelial neoplasia I, II and III (CINI-III), before becoming an invasive carcinoma. Therefore identifying factors, which regulate the transition through the premalignant phases and onto invasive cancer would be of importance clinically, to identify patients at risk of progressing from CIN I to CIN III. We show that expression of E6 and E7 proteins from the high risk HPV16, causes reduced expression of the IGF binding protein 2 (IGFBP2) and this correlates with progression from CIN I to CIN III. By modulating IGFBP2 levels in epithelial cells, we have demonstrated that reduction of IGFBP2 levels is a driving event in epithelial invasion. We have gone on to show that de-regulation of expression of IGFBP2 is due to histone deacetylation of the promoter, which can be reversed by histone deacetylase inhibitors.
Published in the journal: HPV16 Down-Regulates the Insulin-Like Growth Factor Binding Protein 2 to Promote Epithelial Invasion in Organotypic Cultures. PLoS Pathog 11(6): e32767. doi:10.1371/journal.ppat.1004988
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1004988Summary
The human papillomaviruses (HPV) are the etiological agents of cervical cancer and the disease progresses through the pre-malignant phases of cervical intraepithelial neoplasia I, II and III (CINI-III), before becoming an invasive carcinoma. Therefore identifying factors, which regulate the transition through the premalignant phases and onto invasive cancer would be of importance clinically, to identify patients at risk of progressing from CIN I to CIN III. We show that expression of E6 and E7 proteins from the high risk HPV16, causes reduced expression of the IGF binding protein 2 (IGFBP2) and this correlates with progression from CIN I to CIN III. By modulating IGFBP2 levels in epithelial cells, we have demonstrated that reduction of IGFBP2 levels is a driving event in epithelial invasion. We have gone on to show that de-regulation of expression of IGFBP2 is due to histone deacetylation of the promoter, which can be reversed by histone deacetylase inhibitors.
Introduction
Metastasis involves multiple steps, so defining the processes which regulate cancer cell invasion are crucial for understanding the initiation of the metastatic process. In particular, it will be important to monitor the molecular events that occur in the transition from a hyper-proliferative epithelium to an invasive epithelium and determine their functions. High-risk human papillomavirus (HPV) types are responsible for the transformation of the cervical epithelium and subsequent cervical cancer. Expression of the ‘early’ HPV genes E6 and E7 has been identified to be sufficient to immortalise primary human keratinocytes [1,2] and are required for continued proliferation of infected cells however whether this is sufficient to transform cells into a malignant form is still disputed [2–4]. E6 and E7 proteins immortalize epithelial cells through their ability to inactivate the cell cycle checkpoints regulated by the retinoblastoma protein (pRb) and p53 resulting in enhanced proliferation and loss of differentiation [5–7]. If not cleared, the HPV infection can persist resulting in progression to invasive disease [8]. However, not all HPV infections of the cervix lead to progressive disease and so knowledge of the alterations during transition from low grade, CIN 1, to high grade disease, CIN 3 and eventual invasive disease may yield novel molecular biomarkers that distinguish lesions with a propensity to progress to invasive disease from lesions that will remain premalignant [9].
During the development of cervical cancer, numerous molecular events have been described, including: altered viral gene expression [10,11], regulation of immune-response [12], activation of proliferative signalling pathways [13–15], modification of chromatin [16–19], and regulation of pro-invasive genes, such as matrix metalloproteases (MMPs) [11,20]. In the present study, we have investigated the factors and mechanisms which influence the invasive behaviour of the epithelium. We have examined the ability of the high-risk HPV16 E6 and E7 genes to transform primary human foreskin keratinocytes (HFKs) into an invasive epithelium and have identified a crucial role for the IGF (Insulin-like growth factor) signalling pathway in the progression to invasive growth. The invasive potential of E6/7 expressing keratinocytes is enhanced following dramatic down-regulation of insulin-like growth factor binding protein 2 (IGFBP2), resulting from enhanced histone deacetylase 3 activity at the IGFBP2 promoter. IGFBP2 has been shown to have both pro-tumourgenic properties and tumour suppressive functions, although the former tend to be independent of IGF/IGF receptor signalling [21]. In this study, we have found that IGFBP2 acts to suppress IGFI/II stimulation of the IGF receptor 1 (IGF1R) but in its absence, IGFI/II signalling, in conjunction with the stromal derived growth factor, keratinocyte growth factor (KGF), stimulates the AKT pathway leading to invasion. Significantly, we have observed that IGFBP2 expression is inhibited in high-grade pre-malignant cervical lesions infected with HPV16 and propose that this down-regulation is a required step in the initiation of the invasion process.
Results
The high-risk HPVs are a causal factor for cervical and infection is observed in a proportion of head and neck cancers. Whilst E6 and E7 expressing keratinocytes are immortalized, we have observed in three-dimensional organotypic cultures that they do not possess the ability to invade into the stroma [1,2]. The stromal compartment also regulates the invasive behaviour of the epithelium [22–24], and we have recently demonstrated that pRb-depleted human foreskin fibroblasts (HFFs) promote epithelial invasion, i.e. breakdown of the basement membrane and growth into the underlying collagen layer (S1 Fig). This invasion is driven through altered secretion of the keratinocyte growth factor [22]. Organotypic cultures generated using early-passage (passage 3–10) HPV16 E6/7 expressing HFKs are refractory to the pro-invasive signals from pRb-depleted HFFs. However, with continued passage (late passage) (i.e. >passage 14), these cells acquire the ability to invade (Fig 1A). We hypothesized that following extended passage. E6/7-HFKs may secrete growth factors or cytokines that could alter invasive behaviour of the epithelium.
Fig. 1. Prolonged passage of HPV16 E6/7 immortalised keratinocytes generates an invasive epithelium. 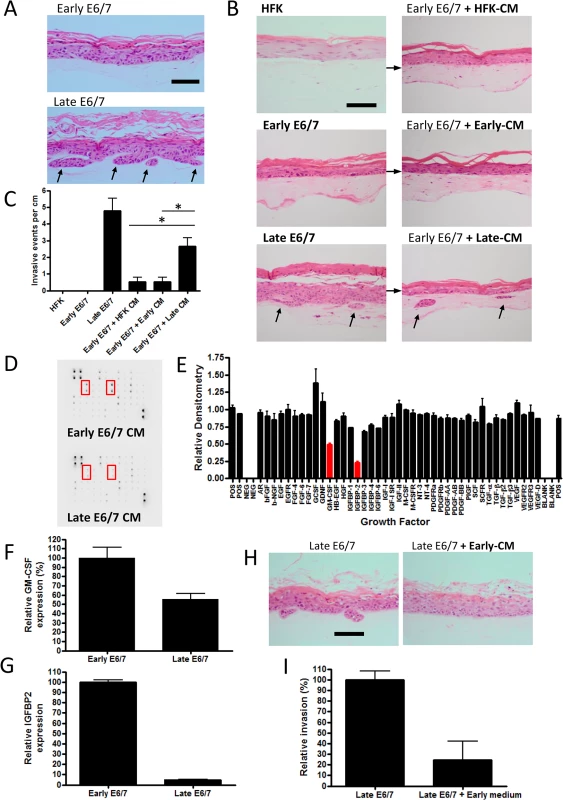
A) H+E stained sections of organotypic cultures containing early and late passage E6/7-HFKs cultured with pro-invasive pRb-depleted fibroblasts. Late-passage but not early passage E6/7-HFKs invade into the collagen matrix. B) Medium transfer from late-passage E6/7-HFKs (Late-CM) can promote invasion in early pass E6/7-HFKs unlike medium from normal HFKs (HFK-CM) and early-passage E6/7-HFKs (Early-CM). The frequency of invasion is shown in C), data represents the average of three independent experiments, error bars represent standard error of the means (SEM). CM = conditioned medium. D) Growth factor array analysis of conditioned medium, which is quantified in E) demonstrates late-passage E6/7-HFKs secrete less IGFBP2 and GM-CSF. F and G) GM-CSF and IGFBP2 mRNA transcript levels are reduced in late-passage E6/7-HFKs compared to non-invasive early pass E6/7-HFKs. n = 3, error bars represent SEM. H) Medium transfer from non-invasive early E6/7-HFKs blocks invasion of late-passage E6/7-HFK cultures as quantified in I). n = 3, error bars represent SEM. Scale bars represent 100 μM. To test this hypothesis, conditioned medium (CM) from monolayer cultures of invasive cells, normal HFKs and early-passage E6/7-HFKs was transferred daily to organotypic cultures containing non-invasive early E6/7-HFKs. Medium taken from late-passage E6/7-HFKs was sufficient to induce invasion of the previously non-invasive cells, although not to the same level as late passage E6/7-HFKs (Fig 1B and 1C), suggesting invasive late-passage E6/7-HFKs were generating a pro-invasive environment. Subsequently, conditioned medium from normalised numbers of early and late-passage E6/7-HFKs were subjected to growth factor array analysis, which measured the levels of 41 growth factors. Surprisingly, the pro-invasive late-passage E6/7-HFKs did not secrete additional growth factors/cytokines but produced significantly lower levels of the insulin-like growth factor binding protein, IGFBP2, and the granulocyte-macrophage colony-stimulating factor (GM-CSF) at the protein (Fig 1D and 1E) and mRNA level (Fig 1F and 1G) in cell extracts. This result suggested that an inhibitor of invasion was lost as cells acquired invasive behaviour, indeed, when media from non-invasive, early pass E6/7-HFKs was transferred to invasive late-passage E6/7-HFKs, we observed a complete inhibition of invasion (Fig 1H and 1I).
Since IGFBP2 expression was the most dramatically altered factor (>90% loss, p<0.001), we wanted to investigate its role in the invasive phenotype of late passage E6/7-HFKs. Western blot and real-time analysis from three independently generated E6/7-HFK lines confirmed that late-passage E6/7-HFKs produced very low levels of IGFBP2 in comparison to early-passage E6/7-HFKs as well as primary HFKs (Fig 2A and 2B, and S2A Fig). Down-regulation of IGFBP2 also occurred with continued passage of E6/7-HFKs maintained in co-culture with J2-3T3 fibroblasts in E-medium (S2B Fig). Further examination of the IGFBP family identified that there was a modest increase in IGFBP3 but only IGFBP2 was significantly regulated following continued passage of E6/7-HFKs (Fig 2C). These results implied that IGFBP2 may be down-regulated either as a consequence of long term culture or prolonged HPV16 E6/7 expression. The expression of IGFBP2 was therefore monitored in primary human foreskin keratinocytes and immortalised keratinocytes (immortalised either by hTERT or through co-culture with J2-3T3 mouse fibroblasts and the Rock inhibitor Y27632 [23]). IGFBP2 was expressed at high levels in immortalized cells relative to late passage E6/7 keratinocytes (S2C and S2D Fig). Furthermore, we established that HPV16 E6/7 were able to mediate down-regulation of IGFBP2 when introduced into these immortalised cells whereas control cells transfected with vector only did not (S2E and S2F Fig). This implied that the down-regulation of IGFBP2 was a result of prolonged HPV16 E6/7 expression. To test this we targeted E6 and E7 in late passage E6/7-HFKs with siRNA, which resulted in re-expression of p53, pRb, and IGFBP2 and concomitantly inhibited invasion in organotypic cultures (S3A–S3D Fig). There is also a correlation between IGFBP2 levels and HPV in publicly available microarray datasets from various cervical cancer cell lines [24] where HPV positive cervical cancer cell lines were shown to have substantially reduced IGFBP2 expression compared to HPV negative cervical cell lines (Fig 2D). We independently confirmed this at protein and RNA levels in C33a, Caski and Hela cell lines (Fig 2E and 2F).
Fig. 2. IGFBP2 is transcriptionally repressed in late-passage E6/7-HFKs and HPV positive cervical cancer cell lines. 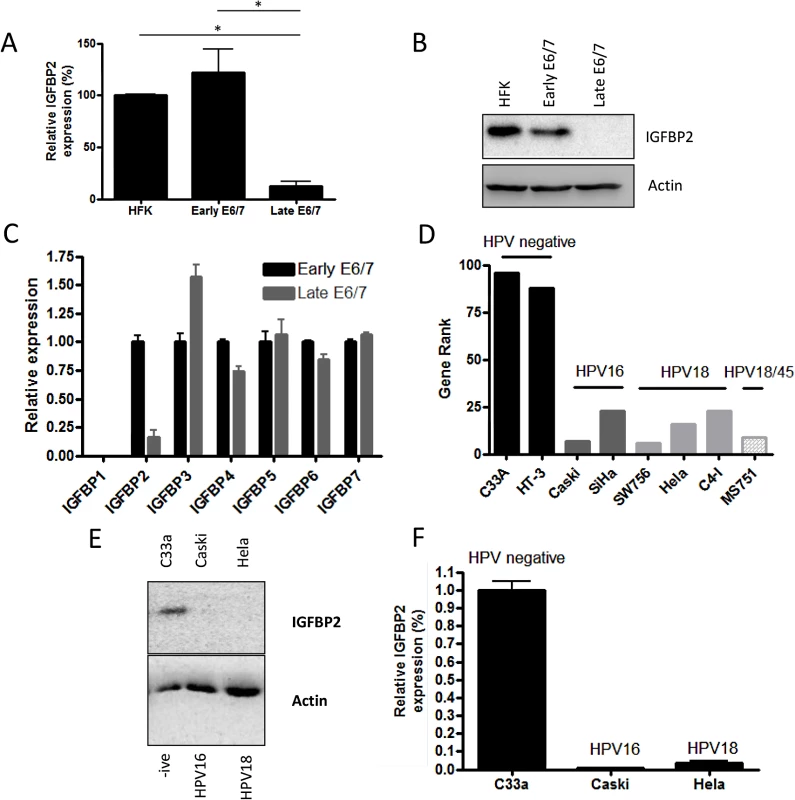
A) Continued expression of E6/7 in HFKs leads to dramatic down-regulation of IGFBP2 mRNA levels, n = 4, error bars represent SEM, this is also observed at a protein level (B). C) Real-time PCR analysis of the IGFBP family of proteins demonstrated a specific down regulation of IGFBP2 expression. n = 3, error bars represent SEM. Note IGFBP1 is not expressed by HFKs but was readily detected in human fibroblasts (S4A Fig). D) Gene rank analysis of IGFBP2 expression in cervical cells lines identified that IGFBP2 expression is low in HPV positive cell lines in comparison to cervical HPV negative cell lines (C33A and HT-3). E) The results in D were confirmed by western blot (E) and real-time PCR analysis (F) for the C33a, Caski and Hela cervical cell lines. n = 3, error bars represent SEM. To establish a role for IGFBP2 in the invasion process, recombinant IGFBP2 was added to organotypic cultures containing invasive late-passage E6/7-HFKs. Addition of physiological quantities of IGFBP2 to the cultures resulted in inhibition of epithelial invasion in a dose dependent manner (Fig 3A and 3B, and S4B and S4C Fig). As IGFBP3 was found to be elevated in late passage cultures and is thought to promote invasion, we assessed whether IGFBP3 addition further induced epithelial invasion in late passage E6/7-HFKs (S4D and S4E Fig), but found no further effect of IGFBP3 addition. This was also established in a modified organotypic raft culture containing the HPV16 positive cervical cancer cell line, Caski (S4F and S4G Fig), where IGFBP2, but not IGFBP3, significantly inhibited invasion of the epithelial cells. To further assess the effects of IGFBP2 loss on the invasive potential of the epithelium, IGFBP2 levels were stably depleted in early-passage non-invasive E6/7-HFKs using two different shRNA molecules. IGFBP2 knockdown was confirmed by Western blot and real-time PCR analysis (Fig 3C and 3D), and resulted in enhanced invasion (Fig 3E and 3F). IGFBP2 was also depleted in primary HFKs (Fig 3G), however, this did not result in the generation of an invasive epithelium (Fig 3E and 3F) suggesting that IGFBP2 acts as a brake to pro-invasive signalling mediated by E6 and E7 and following continued cell expansion, this brake is lost.
Fig. 3. IGFBP2 is a regulator of the invasion process. 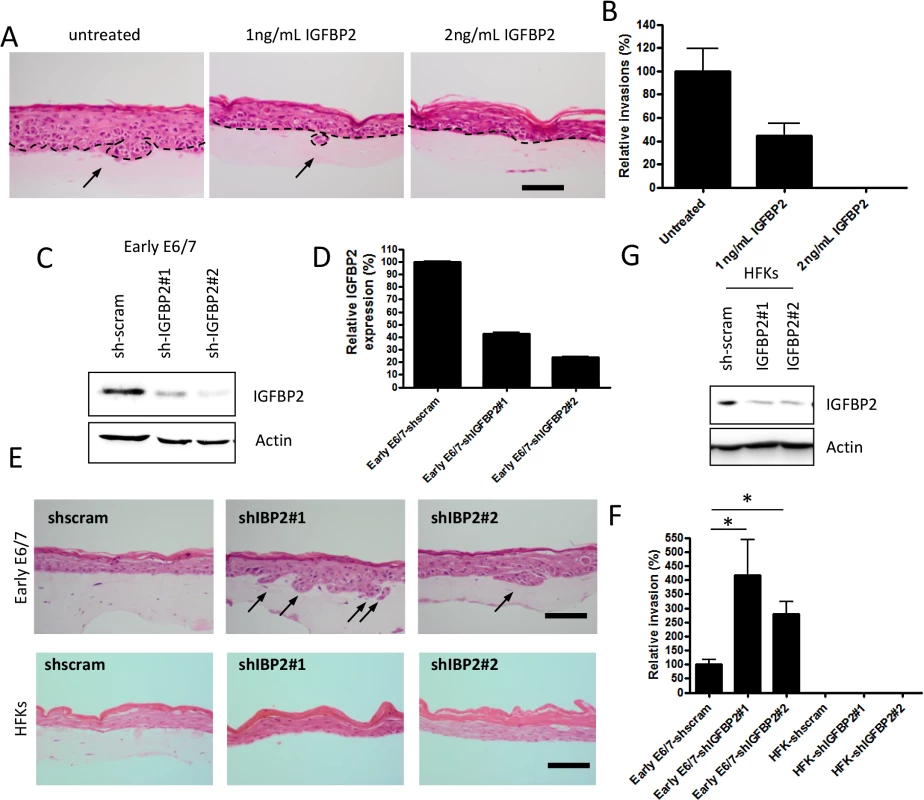
A) Recombinant IGFBP2 was added to organotypic cultures at the indicated concentrations over a 14 day period. Addition of IGFBP2 inhibited the invasive behaviour of late-passage E6/7-HFKs and the invasive frequencies are quantified in B). n = 3, error bars represent SEM. C) IGFBP2 levels were depleted from early-passage E6/7-HFKs as demonstrated by western blot (C) and real-time analysis (D). n = 3, error bars represent SEM. E) Depletion of IGFBP2 from early-passage E6/7-HFKs resulted in enhanced invasive potential as quantified in F). However this was not observed when IGFBP2 was depleted in primary HFKs (G, also E and F). n = 3, error bars represent SEM. Scale bars represent 100 μM. IGFBP2 expression is regulated by a variety of factors, including the IGF system itself [25] and insulin [26], however in E6/7 expressing keratinocytes, we found this was not the case (S4H Fig). Epigenetic mechanisms have been associated with regulation IGFBP2 expression [27–29], so next we wanted to determine if one or more of these mechanisms played a role in IGFBP2 regulation and if manipulation of the epigenetic factors would restore the IGFBP2 levels in late pass E6/7-HFKs. We have identified that as E6/7 immortalised keratinocytes are passaged, there is acquisition of methylation marks at CpG islands close to the transcriptional start of IGFBP2 in late passage cells only (S5A and S5B Fig). However addition of 5-aza-C was unable to restore expression of IGFBP2 in the invasive cells (S5C and S5D Fig) suggesting DNA methylation may not be the critical regulator of IGFBP2. HDAC inhibitors have been shown to elevate expression of IGFBP2 in cells which readily express the protein [28,29], so to determine if this had occurred in late passage cells, which exhibit low levels of IGFBP2, we added the pan-HDAC inhibitors sodium butyrate (SB) or trichostatin A (TSA) to invasive E6/7-expressing keratinocytes and Hela cells. The inhibitors restored both IGFBP2 mRNA and protein (Fig 4A and 4B) in late E6/E7 keratinocytes, as well as in Hela cells (S6 Fig). Since these results suggest that there is increased histone deacetylation in the invasive keratinocytes resulting in reduced expression of IGFBP2, we determined the expression of HDACs during the transition to an invasive epithelium. HDAC 1, 2, 3, 5 and 6 were elevated in the invasive epithelial cells (Fig 4C), and cell survival assays suggested that these invasive cells are sensitive to HDAC inhibition (S7 Fig). Interestingly, addition of low doses (IC25) of the HDAC inhibitors TSA, SAHA (both pan inhibitors) and romidepsin (Romi, a class I inhibitor, which inhibits HDACs 1, 2, 3 but also HDAC4 and 6), which do not inhibit proliferation, was sufficient to reduce invasive frequency of the late passage E6/7-HFKs (Fig 4D and 4E).
Fig. 4. IGFBP2 expression is repressed through enhanced HDAC function. 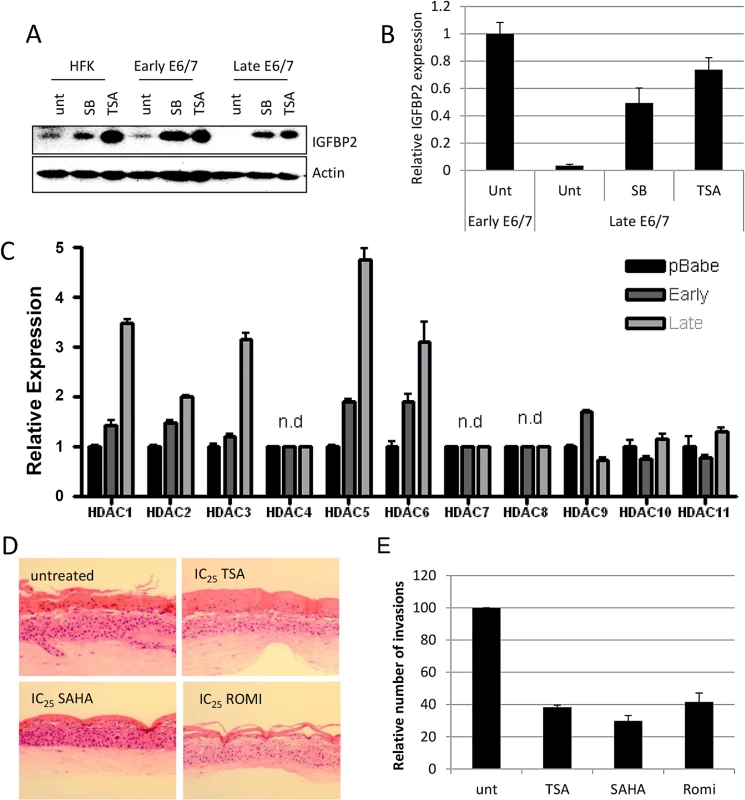
A) Addition of 1 μM Trichostatin A (TSA) or 5 mM sodium butyrate (SB) restores expression of IGFBP2 in late passage E6/7-HFKs to levels similar to primary keratinocytes, as demonstrated by Western blot and real-time PCR (B). n = 3, error bars represent SEM. C) The expression levels of histone deacetylases were assessed by real-time PCR in control primary keratinocytes (pBabe) early and late passage E6/7-HFKs. n = 3, representative experiment shown, error bars represent standard deviation (SD). D) Addition of HDAC inhibitors to organotypic cultures significantly reduced the invasive frequency of the epithelium, as quantified in (E). n = 3, error bars represent SEM. To further evaluate the mechanism through which IGFBP2 expression is regulated by histone modifications, selective HDAC inhibitors (proprietary inhibitors of HDAC6 (HDAC6i), HDAC1 and 2 (HDAC1/2i) and HDAC3 (HDAC3i)) and entinostat, which is another class I inhibitor, were employed. Inhibitors of HDAC3, the class I inhibitor, entinostat and to a lesser extent the HDAC1/2 inhibitor were sufficient to restore IGFBP2 RNA and protein expression (Fig 5A and 5B). Using a commercially available HDAC3 selective inhibitor, RGFP966, IGFBP2 expression was also restored (S8 Fig). To further confirm a specific role for HDAC3 in the regulation of IGFBP2 expression, HDAC1-3 were individually depleted using siRNA and results showed that depletion of HDAC3 was sufficient to restore both mRNA and protein expression of IGFBP2 (Fig 5C and 5D).
Fig. 5. HDAC3 is a critical regulator of IGFBP2 expression. 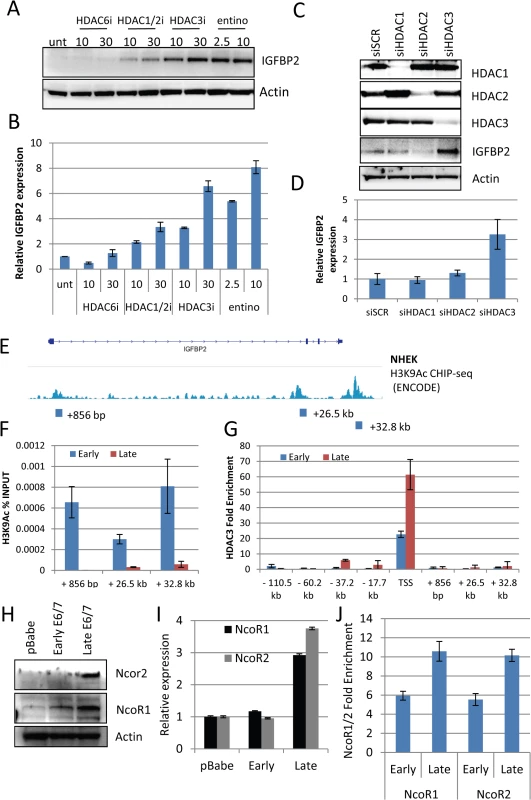
A) Using selective (HDAC6i, HDAC1/2i and HDAC3i) HDAC inhibitors and a class I inhibitor (entinostat – entino), we were able to restore IGFBP2 expression in late passage E6/7-HFKs both at the protein and transcriptional level (B). n = 3, representative experiment shown, error bars represent SD. C) Following 48 hours treatment with siRNA targeting HDAC1, 2 or 3, western blot analysis confirmed the specific depletion of individual HDACs siRNA and restoration of IGFBP2 following HDAC3 depletion. D) This was also confirmed at the transcriptional level n = 3, error bars represent SEM. E) Encode histone 3 lysine 9 acetylation (H3K9Ac) CHIP-seq from normal human epithelial keratinocytes (NHEK) at the IGFBP2 locus. Blocks represent primer locations to assess the levels of H3K9Ac binding at these sites. F) H3K9Ac CHIP-qPCR at the three sites identified in E, demonstrates the loss of the modification in late passage E6/7-HFKs which lack IGFBP2 expression. n = 3, error bars represent SEM G) HDAC3 CHIP-qPCR was conducted at locations throughout the IGFBP2 locus, predicted from previous HDAC3 CHIP-seq experiments in human and mouse derived cells. HDAC3 binding was enriched at the transcriptional start site (TSS) and was enhanced in late passage E6/7-HFKs. n = 3, error bars represent SEM. H) Western blot of NcoR1 and NcoR2 in HFKs, early and late passage E6/7-HFKs. I) The co-repressors NcoR1 and NcoR2 are elevated in late passage E6/7 cells at the protein and transcriptional level (I). n = 3, error bars represent SEM J) CHIP-qPCR analysis also demonstrated enhanced binding of both NcoR1 and NcoR2 at the TSS. Average of two experiments shown, error bars represent SD. To assess the function of HDAC3 in regulating IGFBP2 expression we utilised publicly available data, which examined histone modifications around the IGFBP2 locus in primary keratinocytes [30] (Fig 5E). Three histone 3 lysine 9 (H3K9) acetylation sites, which can be altered by HDAC3 [31], were identified. Using CHIP-qPCR with anti-H3K9Ac CHIP-tested antibodies [30], we observed that the acetylation at these three sites is lost in invasive cells, which do not express IGFBP2 (Fig 5F), suggesting a mechanism for the loss of expression of IGFBP2 in late passage cells. In addition, the IGFBP2 promoter is bivalent, containing both active and repressive histone modifications within the promoter region, with the activating modifications proposed to predominate over the repressive elements [32]. Our results suggest that the loss of the activating marks allows the repressive elements to predominate leading to repression of the gene expression as previously suggested [33]. To support this suggestion, we showed, using ChIP-qPCR, that there is an increase in the presence of HDAC3 at the transcriptional start site, but no significant enrichment of HDAC3 at other sites within the IGFBP2 locus (Fig 5G). Furthermore, HDAC3 resides in a repressive complex with NcoR1 (NcoR1) and NcoR2 (SMRT) [34] resulting in an active repressive complex [35]. Analysis of NcoR1/2 at the protein and transcriptional levels showed marked elevation in invasive cells (Fig 5H and 5I) and also an enrichment at the Transcriptional Start Site (TSS) of IGFBP2 (Fig 5J).
Having established that IGFBP2 expression is lost in late passage E6/7-HFKs we next wanted to identify the significance of this loss and how it affected downstream signalling in the invasive cells. Since IGFBP2 has been shown to function through preventing IGF1 and IGF2 from binding to the IGF1-receptor [36–38], we treated E6/7-HFKs with IGFBP2 prior to IGF1/2 treatment and established that IGFBP2 is acting to block IGF1/2 induced AKT and ERK activation (Fig 6A). However, primary human foreskin keratinocytes (HFK) and early-passage E6/7-HFKs were unresponsive to IGF1 and IGF2 treatment (Fig 6B). These results implied that IGF-signalling is enhanced in the invasive cells, as a result of the loss of IGFBP2. The expression of the IGF-receptors 1 and 2 (IGF1R and IGF2R) in invasive versus non-invasive E6/7-HFKs was assessed by real-time and Western blot analysis and shown to be elevated in invasive cells whereas the related insulin receptor was unaltered (Fig 6C and 6D). This also mirrors observations that IGF1R expression is elevated in CIN3 cervical lesions [13,39].
Fig. 6. IGFBP2 blocks pro-invasive IGF1R signalling. 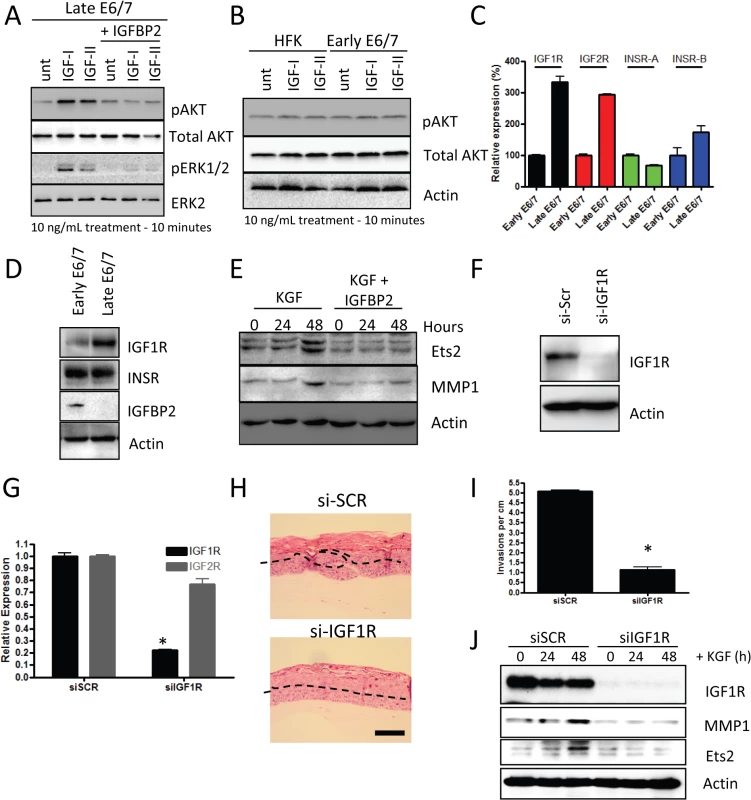
A) Late-passage E6/7-HFKs were pre-treated with 10ng/mL IGFBP2 for 1 hour prior to 10 minutes of 10ng/ml of IGF1/2 treatment, as described in the Methods section. IGF1/2 induced activation of AKT and ERK pathways, which was inhibited by IGFBP2. B) Similar treatment of primary HFKs and early-passage E6/7-HFKs showed that these cells do not activate AKT in response to IGF1/2 treatment. C) The expression of the IGF and insulin receptors (IGF1R/2R and INSR-A/B, respectively) were assessed by real-time PCR and demonstrated significant increases in IGF1R and IGF2R mRNA. n = 3, error bars represent SEM. This was also detected at a protein level for IGF1R in cycling cultures of late passage E6/7-HFKs D). E) IGFBP2 blocks signalling pathways mediated by KGF, including those that result in activation of Ets1 and MMP1. F) The role of IGF1R signalling in regulating invasion was assessed by depletion of IGF1R using siRNA (siIGF1R) and knockdown confirmed by western blotting (F) and real-time PCR (G). n = 3, error bars represent SEM. H) siIGF1R treatment inhibited the invasion process, as quantified in I). Average of three experiments, error bars represent SEM. J) IGF1R knockdown inhibited the KGF-dependent activation of Ets2 and MMP1. Scale bars represent 100 μM. Previous work has demonstrated that invasion of the E6/7-HFKs relies on the secretion of keratinocyte growth factor (KGF or FGF7) from stromal fibroblasts acting on the Fibroblast growth factor receptor 2b (KGFR/FGFR2b) on epithelial cells [22]. Therefore, we next investigated whether IGFBP2 could alter these effects. We treated late-passage E6/7-HFKs with KGF in the presence or absence of IGFBP2 and showed that IGFBP2 was sufficient to inhibit KGF induction of ETS2 and MMP1, known modulators of the invasive process (Fig 6E) [40,41]. This implied a crucial role for the IGF pathway in the regulation of invasion. To test this hypothesis, IGF1R levels were depleted by siRNA in invasive late-passage E6/7-HFKs and depletion confirmed at the protein and mRNA levels (Fig 6F and 6G). Depletion of IGF1R in the epithelium significantly reduced the frequency of invasions in organotypic rafts, suggesting that the IGF signalling pathway is pro-invasive (Fig 6H and 6I). We also tested whether IGF1R-depleted cells respond to the KGF pro-invasive stimulus, and similar to IGFBP2 treatment of these cells, KGF was unable to activate ETS2 and MMP1 in the absence of IGFR1 (Fig 6J).
The ability of IGF signalling to alter the pro-invasive signalling of KGF implied that the two pathways were connected and it has recently been reported that KGF functions in a protease dependent manner, specifically activating A Disintegrin And Metalloprotease 17 (ADAM17) [42]. We also confirmed in our cells that KGF activates downstream signalling events in a protease dependent manner, using the protease inhibitor GM6001 (Fig 7A). Furthermore, we found that by depleting IGF1R in the late-passage E6/7-HFKs the activation of AKT was inhibited following KGF treatment, suggesting that KGF-induced activation of AKT is both a protease-dependent and IGF1R-dependent process (Fig 7B). Late-passage E6/7-HFKs were compared to early-passage cells in terms of their expression of the ADAM family of proteins and ADAM17 expression was found to be elevated in these cells (Fig 7C). We then tested whether KGF induced activation of AKT is ADAM17 dependent using siRNA. ADAM17 was efficiently depleted (Fig 7D and 7E) and this prevented activation of the AKT pathway (Fig 7F and 7G). ADAM17 has been shown to shed various growth factors from cells, including IGF [43] so we determined if IGF is secreted from E6/7-HFKs following KGF treatment. IGF was found to be secreted from invasive E6/7-HFKs on KGF treatment, however, following knockdown of ADAM17 this secretion was inhibited (Fig 7H), implying that KGF induced ADAM17 activation leads to enhanced shedding of IGF from invasive cells and drives activation of the AKT pathway through activation of IGF1R.
Fig. 7. Signalling mediated by KGF requires IGF1R. 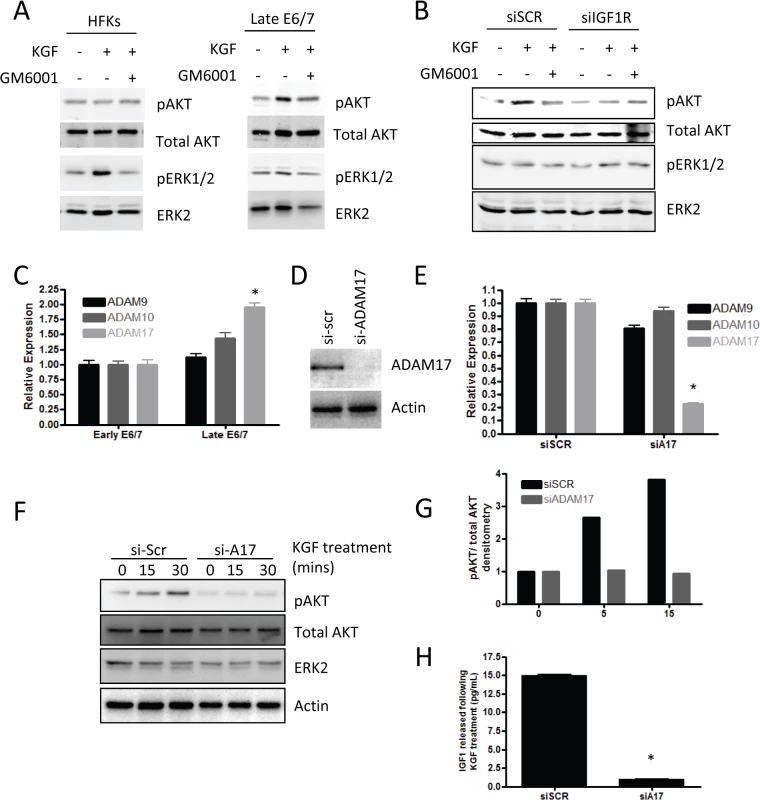
A) KGF treatment of primary HFKs leads to activation of the AKT pathway in late-passage E6/7-HFKs only. B) siRNA depletion of IGF1R (siIGF1R) prevents the activation of AKT by KGF suggesting that IGF1R forms part of the signalling pathway. C) ADAM17 expression is elevated in late-passage, invasive, E6/7-HFKs, compare to early-passage E6/7-HFKs. n = 3, error bars represent SEM. D) Knockdown of ADAM17. n = 4, representative experiment shown, error bars represent SD, confirmed by western blot and real-time PCR (E) resulted in inhibition of AKT activation by KGF (F), as quantified in (G). H) Following 30 mins treatment of KGF, IGF1 was shed from late-passage E6/7-HFKs in an ADAM17 dependent manner. n = 4, error bars represent SEM. In order to establish the clinical relevance of our findings, expression of IGFBP2 was assessed in pre-malignant cervical intraepithelial neoplasia’s (CIN) by immunohistochemistry and dual immunofluorescence, utilising p16INK4A (p16) staining to distinguish premalignant cells from normal cells. IGFBP2 was readily detected in uninfected normal cervical epithelium (Fig 8A, p16 negative region, white arrow), however in regions where HPV16 had infected the epithelium, IGFBP2 was dramatically reduced in CIN3 lesions (Fig 8A, p16 positive regions, red arrow). Following our initial observations we evaluated IGFBP2 expression in a blinded manner in 40 CIN1 lesions and 39 CIN3 lesions, all of which had previously been identified as HPV16 positive [44]. IGFBP2 was found to be down-regulated in 43% of CIN1 lesions whilst in CIN3 lesions, 85% of the samples showed down-regulation (Fig 8B and 8C). There was a significant difference in the expected ratio of samples with IGFBP2 loss when comparing CIN1 and CIN3 lesions using the Fischer’s exact test, Chi-square test and Z-test (p<0.001). IGFBP2 was also found to be down-regulated in invasive disease (Fig 8B and 8C), although there was no difference in the proportions of tumours with reduced IGFBP2 at the various stages examined (Fig 8D). We had follow-up data for 13 patients with CIN1 where IGFBP2 was reduced. From this group, 10 patients progressed to CIN3, the other 3 patients either regressed (1 case) or remained CIN1 (2 cases) (Fig 8E). The results imply that IGFBP2 is commonly down-regulated at advanced stages of infection, correlating with the effects of prolonged HPV16 E6 and E7 expression and reduced levels of IGFBP2 in CIN1 disease may indicate a propensity to progress to a high grade. We have also investigated whether IGFBP2 expression is regulated in other cancers associated with HPV infection. HPV infection has been observed in head and neck cancers, where it is associated with between 30–60% of oro-pharyngeal cancers [45]. We have utilised publically available gene array datasets to assess the expression of IGFBP2 in oro-pharyngeal cancers [46]. In these studies HPV infection was detected by immunohistochemistry of the surrogate marker p16 and the samples were sub-divided into p16 positive and p16 negative groups and microarray datasets were analysed for IGFBP2 and p16 expression. In p16 positive cancers, IGFBP2 expression was significantly reduced (Fig 8F), suggesting that down-regulation of IGFBP2 expression is likely associated with HPV infection in oro-pharyngeal cancers.
Fig. 8. IGFBP2 is frequently down regulated in HPV16 infected CIN3 lesions. 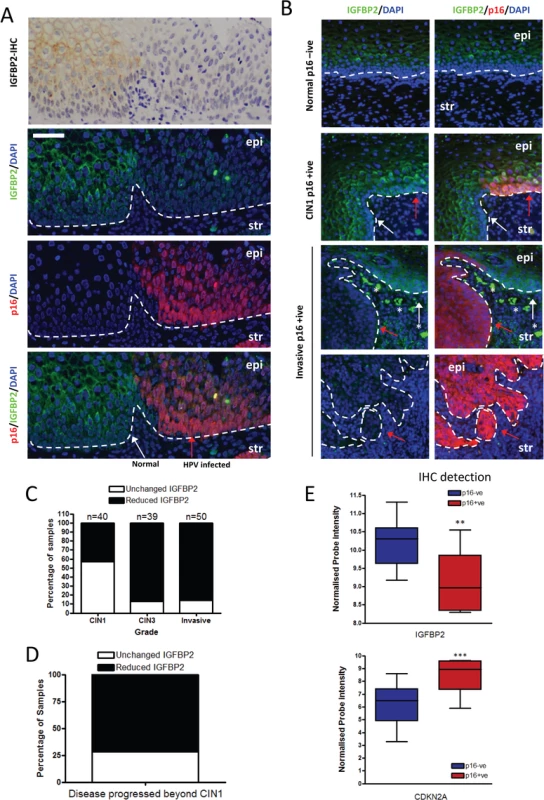
A) Immunohistochemical (IHC) and immunofluorescence staining of IGFBP2 localises the protein in the cytoplasm of epithelial cells of the cervix with regions of low IGFBP2 observed. In order to assess whether these were in HPV infected regions the same sample was co-stained with IGFBP2 and p16, the latter a surrogate marker of HPV infection. IGFBP2 was found to be commonly down-regulated in HPV infected regions (CIN3). HPV infected epithelium is indicated with a red arrow, and neighbouring normal tissue with a white arrow. Epi = epithelium; str = stroma. B) To further address the role of IGFBP2 in the progression of cervical cancer, 40 CIN1, 39 CIN3 lesions and 50 invasive carcinomas were assessed for IGFBP2 staining intensity and scored as-, +, ++, +++, for normal and p16 positive regions. If IGFBP2 scores decreased by a factor of 2 or more between p16 positive compared to normal regions then samples were identified as ‘reduced IGFBP2’. Shown are representative images of CIN1 where IGFBP2 expression was un-altered and invasive disease where IGFBP2 is reduced in p16 positive regions compared to neighbouring normal epithelium *indicates non-specific staining of red blood cells. Arrows are as stated above. C) Quantification across all samples. D) We had data for 13 patients who had low levels of IGFBP2 in the original CIN1 biopsy. 10 patients progressed, whilst 1 patient regressed and 2 had persistent CIN1 lesions. E) Analysis of publically available microarray datasets for IGFBP2 in oro-pharyngeal cancers demonstrated that in tumours where p16 is readily detected by IHC, IGFBP2 is reduced compared to tumours with no p16 staining. p16 expression in these dataset was also assessed by mRNA expression and was used as a surrogate of HPV infection. In summary, we have shown that IGFBP2 is reduced in an E6/7 dependent manner over the passage of human keratinocytes leading to invasion in our 3-dimensional model system. Our in vivo studies with cervical premalignancies show that IGFBP2 expression is reduced with severity of disease from CIN1 to CIN3. The reduced IGFBP2 expression leads to activation of the IGF/IGFR pathways, which through cross talk with the KGF/FGFR2b complex can drive invasion (Fig 9). The results suggest that the IGF/IGFR/IGFBP2 axis would make a logical target for further investigation for potential treatment of cervical cancers.
Fig. 9. Proposed mechanism of IGFBP2 function. 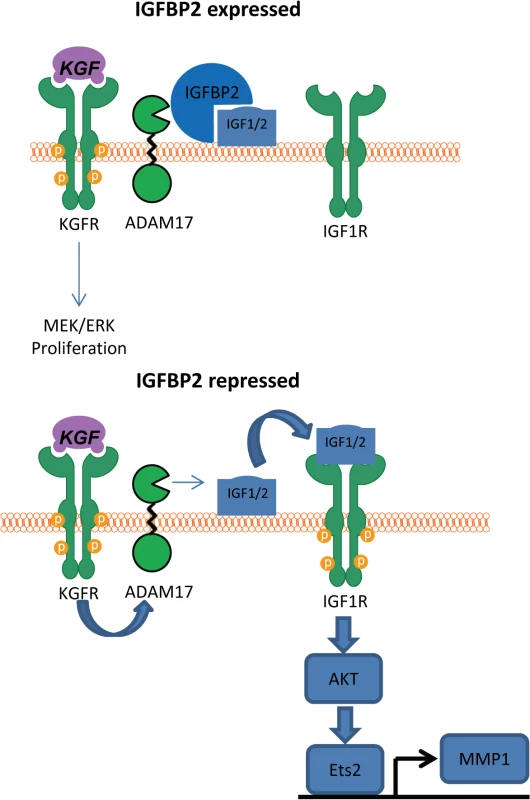
In the presence of IGFBP2, IGF1/2 cannot be released from the cell surface and therefore cannot activate the IGF1 receptor. However, when IGFBP2 is lost, KGF activation of ADAM17 leads to cleavage of the unprotected IGF1 leading to IGF1R activation and subsequent AKT activation, which we have previously demonstrated leads to expression of MMP1 [22]. Discussion
Invasion of the hyper-proliferative cervical epithelium into the surrounding stroma is an important event in progressive disease and here we have identified a crucial role for IGFBP2 in controlling this invasion process. Our results suggest that the prolonged expression of E6/7 proteins can generate an invasive epithelium through depletion of IGFBP2 expression, which in turn leads to signalling through the IGF1R in cross-talk with the FGFR2b. These results are also in keeping with previous results which demonstrated that HPV16 E7 can transform fibroblasts, in an IGF1R dependent manner [47]. We propose that IGFBP2 functions as a brake preventing the HPV-infected epithelium from invading into the underlying stroma.
Our in vitro findings are mirrored in cervical cancer specimens where IGFBP2 expression is commonly lost in 85% of CIN3 lesions, which progress to invasive disease with high incidence, if left untreated [48], while CIN1 lesions do not. We did however observed that 43% of CIN1 lesions have reduced IGFBP2, and our preliminary data indicates that a significant proportion of patients with CIN1 disease who later progressed to a higher grade lesion, had reduced levels of IGFBP2 in the HPV infected epithelium of the original CIN1 biopsy. As this has only been examined in a limited number of cases, future studies are required to confirm that IGFBP2 levels may indicate patients at risk of progression. If the reduction of IGFBP2 levels identifies a sub-group of CIN1 lesions that have the propensity to progress, this could be useful clinically, as these patients could be monitored more closely to detect disease progression. There is a possibility that the CIN1 lesions where IGFBP2 were down regulated were mis-classified, however, grading of lesions was conducted by two pathologists with overall agreement in each case.
Mechanistically our results demonstrate a critical role for IGF-signalling in driving the invasive process. The IGF pathway is well known to be modulated in cancer and is known to promote neoplastic growth [13,39]. The IGF receptors are expressed in a variety of cancers, and in vivo studies have demonstrated that cancer cells have a dependency for IGF1. Following prolonged expression of HPV16 E6 and E7 the IGF pathway becomes activated i) through loss of IGFBP2 and ii) through enhanced expression of the IGF-receptors. Here we demonstrate that re-addition or re-expression of IGFBP2 through HDAC inhibitor treatment, blocks IGF-signalling and is sufficient to inhibit epithelial invasion, while reciprocal knockdown of IGFBP2 in non-invasive E6/7-HFKs resulted in enhanced invasion, demonstrating a critical role for the pathway in the invasion process. We have further demonstrated that the loss of IGFBP2 allows pro-invasive signals derived from the stroma to enhance epithelial invasion, and this is conducted via IGF1R. It has previously been demonstrated that the keratinocyte growth factor functions via activating the metalloprotease ADAM17 [42] and here we show this is also the case, and there is preferential activation of the AKT pathway. We have previously demonstrated that the AKT pathway is activated in cervical cancer specimens [6] and have demonstrated it as a key component of epithelial invasion [22]. Here we show that the activation of AKT by KGF was dependent on IGF1R (Fig 9), and this can be modulated by IGFBP2 and ADAM17. Signalling via the IGF1R pathway has been proposed to be an important determinant of cervical cancer progression, since elevated expression of IGF1R has been observed in cervical specimens which positively correlated with stage of the CIN lesions [13,39] and further highlights the importance of the IGF-axis in HPV infection and incidence of CIN lesions [49]. This, together with our data, suggests that an activated IGF1R pathway promotes a pro-invasive phenotype in the cervical epithelium. An important caveat is that our in vitro model utilises fibroblasts which promote epithelium invasion [22], and reduction of IGFBP2 in the epithelium alone, may not be sufficient to drive invasion in situ. As CIN lesions take a number of years to progress to invasive disease during this time the stroma may be ‘activated’ which in combination with loss of IGFBP2 can drive epithelium invasion. In line with this hypothesis detection of cancer associated myofibroblasts has been observed in the stroma of cervical cancers and is correlated with poor prognosis [50].
IGFBP2 has been demonstrated to have both tumour suppressive and oncogenic properties in different cancer types. Our data show IGFBP2 as an inhibitor of the invasion process in our 3-D model of cervical pre-cancer and in the main, IGFBP2 tumour suppressive functions are those which antagonise IGF signalling [38,51,52], although IGF-independent pro-apoptotic functions of IGFBP2 have also been described [53]. In examples where IGFBP2 functions in an oncogenic manner, these functions appear to be independent of IGF and are mediated via integrin alpha 5 [54,55] which leads to inhibition of PTEN and ultimately activates the AKT pathway [56]. These oncogenic functions of IGFBP2 were not observed in our studies which may be due to functions of the HPV E6 and E7, which have been shown to down-regulate various integrins, including alpha 5 [57]. Whilst the HPV vaccine will ultimately reduce the incidence of cervical cancer if administered universally, there still remains a generation of women who will require intervention. Since IGFBP2 is itself a potential target for therapeutic intervention [58], and has been shown to inhibit the growth of breast cancer cells in vivo [59], we propose that since HDAC3 is involved in reducing expression of IGFBP2, HDAC inhibitors maybe a useful tool to treat patients with progressive disease.
Methods
Ethics statement
Ethical approval for human materials used in this study was from the Northern Ireland Tumour Bank (NIB11-0001).
Cell culture
Primary human foreskin keratinocytes, derived from neonatal foreskins were maintained in Epilife containing growth supplement (Life-Technologies). Primary human foreskin fibroblasts (Cascade Biologics), the cervical cancer cell lines: C33a and Caski (from existing laboratory stocks, frozen at low passage), and Hela’s (American Type Culture Collection) were maintained in DMEM with 10% FBS. hTERT immortalised HFKs were obtained from the Rheinwald lab and maintained in E-medium with mitomycin C arrested J2-3T3 feeders. Immortalisation of HFKs using J2-3T3 and Rock inhibitor, Y27632, was conducted as previously described [23] (n = 2), to compare IGFBP2 expression levels all cells were grown in E-medium with J2-3T3s.
Reagents
Recombinant IGFBP2, IGFBP3, IGF-I, IGF-II, KGF (Peprotech) and protease inhibitor, GM6001 (Millipore) were prepared according to manufacturer’s protocols. Short term treatments were conducted in confluent monolayers generated following 2 days growth of 200,000 HFKs in 6 well plates and medium was replaced with Epilife without growth supplement for 24 hours before treatment with individual growth factors. To assess the effects of IGFBP2 on IGF1/2 signalling, HFKs were grown as above then switched to low glucose, serum free DMEM for 24 hours and samples pre-treated for 1 hour with IGFBP2 prior to IGF1/2 treatment. Samples were lysed in RIPA buffer for 20 minutes on ice. To assess the impact of KGF on ETS2 and MMP1 induction confluent monolayers grown on 100 μg/mL collagen I before commencing KGF treatment in fresh Epilife with growth supplement. Proteins were harvested using urea buffer. Organotypic cultures were grown in the presence of pRb-depleted fibroblasts in E-medium as previously described [5], stained with haemotoxylin and eosin and invasions per cm of raft were counted. In Fig 1B and 1H media was changed each day. A modified organotypic system was used for the Caski cell line as described previously [60] with the exception that matrigel was replaced with an equivalent volume of egg-white, as previously described [61]. Real-time PCR was conducted using Roche Lightcycler480, primers are described in S1 Table and gene expression data was normalised to RPLPO.
Generation of primary cell lines and siRNA
E6/7-HFKs and pRb-depleted HFFs were generated as previously described [6], [22,62]. shIGFBP2#1 and #2 were from the pRSC retrovirus shRNA library (UCL) with the following target sequences, GTGGAGAACCACGTGGACA and CGGAGCAGGTTGCAGACAA. siRNA sequences used in this study were; siE6/7: GCACACACGUAGACAUUCGdTdT as previously described [63]. siHDAC1 and 2 were from Qiagen. siHDAC3: GAUGCUGAACCAUGCACCUTT as previously described [64]. siIGF1R: CAAUGAGUACAACUACCGCdTdT as previously described [65], siRNA targeting ADAM17 was purchased from Dharmacon siADAM17 smartpool. siRNA transfections were conducted using RNAiMAX (Life-Technologies).
Antibodies, arrays, ELISA and CHIPs
Immunofluorescence (IF) and immunohistochemistry staining procedures were previously described [22]. Cervical intraepithelial neoplasia samples (CIN1 and CIN3) were from New Mexico and invasive cervical cancers were from Belfast Health and Social trust and a TMA containing tumour and matched normal tissue (Abcam, Ab178142). IGFBP2 staining intensity and scored as-, +, ++, +++, for normal and p16 positive regions. If IGFBP2 scores decreased by a factor of 2 or more between p16 positive compared to normal regions then samples were identified as reduced IGFBP2. Antibodies used for IF were as follows: IGFBP2 (Cell-Signaling Technologies #3922, 1 : 200), p16 (BD-Biosciences, 554079, 1 : 200). Western blot analysis utilised the following antibodies: Cell-Signaling Technologies: IGFBP2 (#3922, 1 : 1000), phospho-AKTser473 (#4060, 1 : 2000), total AKT (#2920, 1 : 1000) HDAC1 (#5356, 1 : 1000) and HDAC2 (#2545, 1 : 1000), Santa-Cruz: p53 (sc-126, 1 : 1000), pERK1/2 (sc-7383, 1 : 1000), ERK2 (sc-154-G, 1 : 1000), IGF1R (sc-713, 1 : 1000), INS-R (sc711, 1 : 1000), Ets2 (sc-351, 1 : 2000), MMP1 (sc-58377, 1 : 1000) and HDAC3 (sc-11417, 1 : 1000), Sigma-aldrich: beta-actin (A2228, 1 : 10,000) and ADAM17 (SAB3500367, 1 : 1000) NcoR1 (Bethyl laboratories A301-145A, 1 : 1000) and NcoR2 (Abcam Ab24551, 1 : 1000). Human Growth factor array from RayBiotech was conducted according to manufacturer’s protocols and arrays were analysed using densitometry software, Fluorchem SP. Human IGF-1 ELISA was from RayBiotech. CHIP-qPCR experiments were conducted as previously described [66] using 3 x106 cells per immunoprecipitation. Chip antibodies used in this study as stated above, HDAC3 (2μg per CHIP), NcoR1 (2μg per CHIP), NcoR2 (10μL per CHIP) and H3K9Ac (2.5μg per CHIP). Real-time PCR for CHIP-qPCR was conducted using Roche Lightcycler480, primers are described in S2 Table.
Supporting Information
Zdroje
1. Hudson JB, Bedell MA, McCance DJ, Laiminis LA. Immortalization and altered differentiation of human keratinocytes in vitro by the E6 and E7 open reading frames of human papillomavirus type 18. J Virol. 1990;64(2):519–26. 2153221; PubMed Central PMCID: PMCPMC249139.
2. Münger K, Phelps WC, Bubb V, Howley PM, Schlegel R. The E6 and E7 genes of the human papillomavirus type 16 together are necessary and sufficient for transformation of primary human keratinocytes. J Virol. 1989;63(10):4417–21. 2476573; PubMed Central PMCID: PMCPMC251060.
3. Rudlowski C, Becker AJ, Schroder W, Rath W, Büttner R, Moser M. GLUT1 messenger RNA and protein induction relates to the malignant transformation of cervical cancer. Am J Clin Pathol. 2003;120(5):691–8. doi: 10.1309/4KYN-QM58-62JW-2GD7 14608894.
4. Dürst M, Dzarlieva-Petrusevska RT, Boukamp P, Fusenig NE, Gissmann L. Molecular and cytogenetic analysis of immortalized human primary keratinocytes obtained after transfection with human papillomavirus type 16 DNA. Oncogene. 1987;1(3):251–6. 2838778.
5. McCance DJ, Kopan R, Fuchs E, Laimins LA. Human papillomavirus type 16 alters human epithelial cell differentiation in vitro. Proc Natl Acad Sci U S A. 1988;85(19):7169–73. 2459699; PubMed Central PMCID: PMCPMC282145.
6. Menges CW, Baglia LA, Lapoint R, McCance DJ. Human papillomavirus type 16 E7 up-regulates AKT activity through the retinoblastoma protein. Cancer Res. 2006;66(11):5555–9. doi: 10.1158/0008-5472.CAN-06-0499 16740689.
7. Wong PP, Pickard A, McCance DJ. p300 alters keratinocyte cell growth and differentiation through regulation of p21(Waf1/CIP1). PLoS One. 2010;5(1):e8369. doi: 10.1371/journal.pone.0008369 20084294; PubMed Central PMCID: PMCPMC2805707.
8. Woodman CB, Collins SI, Young LS. The natural history of cervical HPV infection: unresolved issues. Nat Rev Cancer. 2007;7(1):11–22. doi: 10.1038/nrc2050 17186016.
9. Wentzensen N. Triage of HPV-positive women in cervical cancer screening. Lancet Oncol. 2013;14(2):107–9. doi: 10.1016/S1470-2045(12)70568-5 23261357.
10. Pastuszak-Lewandoska D, Bartosińska-Dyc A, Migdalska-Sęk M, Czarnecka KH, Nawrot E, Domańska D, et al. HPV16 E6*II gene expression in intraepithelial cervical lesions as an indicator of neoplastic grade: a pilot study. Med Oncol. 2014;31(3):842. doi: 10.1007/s12032-014-0842-6 24436016.
11. Shulzhenko N, Lyng H, Sanson GF, Morgun A. Ménage à trois: an evolutionary interplay between human papillomavirus, a tumor, and a woman. Trends Microbiol. 2014. doi: 10.1016/j.tim.2014.02.009 24674660.
12. Overmeer RM, Henken FE, Bierkens M, Wilting SM, Timmerman I, Meijer CJ, et al. Repression of MAL tumour suppressor activity by promoter methylation during cervical carcinogenesis. J Pathol. 2009;219(3):327–36. doi: 10.1002/path.2598 19662663.
13. Kuramoto H, Hongo A, Liu YX, Ojima Y, Nakamura K, Seki N, et al. Immunohistochemical evaluation of insulin-like growth factor I receptor status in cervical cancer specimens. Acta Med Okayama. 2008;62(4):251–9. 18766208.
14. Schrevel M, Gorter A, Kolkman-Uljee SM, Trimbos JB, Fleuren GJ, Jordanova ES. Molecular mechanisms of epidermal growth factor receptor overexpression in patients with cervical cancer. Mod Pathol. 2011;24(5):720–8. doi: 10.1038/modpathol.2010.239 21252859.
15. Su PH, Lin YW, Huang RL, Liao YP, Lee HY, Wang HC, et al. Epigenetic silencing of PTPRR activates MAPK signaling, promotes metastasis and serves as a biomarker of invasive cervical cancer. Oncogene. 2013;32(1):15–26. doi: 10.1038/onc.2012.29 22330137.
16. Lin Z, Bazzaro M, Wang MC, Chan KC, Peng S, Roden RB. Combination of proteasome and HDAC inhibitors for uterine cervical cancer treatment. Clin Cancer Res. 2009;15(2):570–7. doi: 10.1158/1078-0432.CCR-08-1813 19147762; PubMed Central PMCID: PMCPMC2714480.
17. Hyland PL, McDade SS, McCloskey R, Dickson GJ, Arthur K, McCance DJ, et al. Evidence for alteration of EZH2, BMI1, and KDM6A and epigenetic reprogramming in human papillomavirus type 16 E6/E7-expressing keratinocytes. J Virol. 2011;85(21):10999–1006. doi: 10.1128/JVI.00160-11 21865393; PubMed Central PMCID: PMCPMC3194988.
18. McLaughlin-Drubin ME, Crum CP, Münger K. Human papillomavirus E7 oncoprotein induces KDM6A and KDM6B histone demethylase expression and causes epigenetic reprogramming. Proc Natl Acad Sci U S A. 2011;108(5):2130–5. doi: 10.1073/pnas.1009933108 21245294; PubMed Central PMCID: PMCPMC3033314.
19. Steenbergen RD, Snijders PJ, Heideman DA, Meijer CJ. Clinical implications of (epi)genetic changes in HPV-induced cervical precancerous lesions. Nat Rev Cancer. 2014;14(6):395–405. doi: 10.1038/nrc3728 24854082.
20. Gius D, Funk MC, Chuang EY, Feng S, Huettner PC, Nguyen L, et al. Profiling microdissected epithelium and stroma to model genomic signatures for cervical carcinogenesis accommodating for covariates. Cancer Res. 2007;67(15):7113–23. doi: 10.1158/0008-5472.CAN-07-0260 17671178.
21. Hoeflich A, Reisinger R, Lahm H, Kiess W, Blum WF, Kolb HJ, et al. Insulin-like growth factor-binding protein 2 in tumorigenesis: protector or promoter? Cancer Res. 2001;61(24):8601–10. 11751371.
22. Pickard A, Cichon AC, Barry A, Kieran D, Patel D, Hamilton P, et al. Inactivation of Rb in stromal fibroblasts promotes epithelial cell invasion. EMBO J. 2012;31(14):3092–103. doi: 10.1038/emboj.2012.153 22643222; PubMed Central PMCID: PMCPMC3400012.
23. Chapman S, Liu X, Meyers C, Schlegel R, McBride AA. Human keratinocytes are efficiently immortalized by a Rho kinase inhibitor. J Clin Invest. 2010;120(7):2619–26. doi: 10.1172/JCI42297 20516646; PubMed Central PMCID: PMCPMC2898606.
24. Scotto L, Narayan G, Nandula SV, Arias-Pulido H, Subramaniyam S, Schneider A, et al. Identification of copy number gain and overexpressed genes on chromosome arm 20q by an integrative genomic approach in cervical cancer: potential role in progression. Genes Chromosomes Cancer. 2008;47(9):755–65. doi: 10.1002/gcc.20577 18506748.
25. Ernst CW, White ME. Hormonal regulation of IGF-binding protein-2 expression in proliferating C2C12 myoblasts. J Endocrinol. 1996;149(3):417–29. 8691100.
26. Schmid C, Schläpfer I, Waldvogel M, Meier PJ, Schwander J, Böni-Schnetzler M, et al. Differential regulation of insulin-like growth factor binding protein (IGFBP)-2 mRNA in liver and bone cells by insulin and retinoic acid in vitro. FEBS Lett. 1992;303(2–3):205–9. 1376696.
27. Chiba T, Yokosuka O, Fukai K, Kojima H, Tada M, Arai M, et al. Cell growth inhibition and gene expression induced by the histone deacetylase inhibitor, trichostatin A, on human hepatoma cells. Oncology. 2004;66(6):481–91. doi: 10.1159/000079503 15452378.
28. Yang YJ, Song TY, Park J, Lee J, Lim J, Jang H, et al. Menin mediates epigenetic regulation via histone H3 lysine 9 methylation. Cell Death Dis. 2013;4:e583. doi: 10.1038/cddis.2013.98 23579270; PubMed Central PMCID: PMCPMC3668625.
29. Biernacka KM, Uzoh CC, Zeng L, Persad RA, Bahl A, Gillatt D, et al. Hyperglycaemia-induced chemoresistance of prostate cancer cells due to IGFBP2. Endocr Relat Cancer. 2013;20(5):741–51. doi: 10.1530/ERC-13-0077 23959956.
30. Bernstein BE, Birney E, Dunham I, Green ED, Gunter C, Snyder M, et al. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012;489(7414):57–74. doi: 10.1038/nature11247 22955616; PubMed Central PMCID: PMCPMC3439153.
31. Bhaskara S, Knutson SK, Jiang G, Chandrasekharan MB, Wilson AJ, Zheng S, et al. Hdac3 is essential for the maintenance of chromatin structure and genome stability. Cancer Cell. 2010;18(5):436–47. doi: 10.1016/j.ccr.2010.10.022 21075309; PubMed Central PMCID: PMCPMC3004468.
32. Barski A, Cuddapah S, Cui K, Roh TY, Schones DE, Wang Z, et al. High-resolution profiling of histone methylations in the human genome. Cell. 2007;129(4):823–37. doi: 10.1016/j.cell.2007.05.009 17512414.
33. Bernstein BE, Kamal M, Lindblad-Toh K, Bekiranov S, Bailey DK, Huebert DJ, et al. Genomic maps and comparative analysis of histone modifications in human and mouse. Cell. 2005;120(2):169–81. doi: 10.1016/j.cell.2005.01.001 15680324.
34. Karagianni P, Wong J. HDAC3: taking the SMRT-N-CoRrect road to repression. Oncogene. 2007;26(37):5439–49. doi: 10.1038/sj.onc.1210612 17694085.
35. Guenther MG, Barak O, Lazar MA. The SMRT and N-CoR corepressors are activating cofactors for histone deacetylase 3. Mol Cell Biol. 2001;21(18):6091–101. 11509652; PubMed Central PMCID: PMCPMC87326.
36. Kiepe D, Ulinski T, Powell DR, Durham SK, Mehls O, Tönshoff B. Differential effects of insulin-like growth factor binding proteins-1, -2, -3, and -6 on cultured growth plate chondrocytes. Kidney Int. 2002;62(5):1591–600. doi: 10.1046/j.1523-1755.2002.00603.x 12371959.
37. Firth SM, Baxter RC. Cellular actions of the insulin-like growth factor binding proteins. Endocr Rev. 2002;23(6):824–54. 12466191.
38. Gockerman A, Prevette T, Jones JI, Clemmons DR. Insulin-like growth factor (IGF)-binding proteins inhibit the smooth muscle cell migration responses to IGF-I and IGF-II. Endocrinology. 1995;136(10):4168–73. 7545099.
39. Moreno-Acosta P, Gamboa O, Sanchez de Gomez M, Cendales R, Diaz GD, Romero A, et al. IGF1R gene expression as a predictive marker of response to ionizing radiation for patients with locally advanced HPV16-positive cervical cancer. Anticancer Res. 2012;32(10):4319–25. 23060553.
40. Butticè G, Duterque-Coquillaud M, Basuyaux JP, Carrère S, Kurkinen M, Stéhelin D. Erg, an Ets-family member, differentially regulates human collagenase1 (MMP1) and stromelysin1 (MMP3) gene expression by physically interacting with the Fos/Jun complex. Oncogene. 1996;13(11):2297–306. 8957070.
41. Ropiquet F, Huguenin S, Villette JM, Ronflé V, Le Brun G, Maitland NJ, et al. FGF7/KGF triggers cell transformation and invasion on immortalised human prostatic epithelial PNT1A cells. Int J Cancer. 1999;82(2):237–43. 10389758.
42. Maretzky T, Evers A, Zhou W, Swendeman SL, Wong PM, Rafii S, et al. Migration of growth factor-stimulated epithelial and endothelial cells depends on EGFR transactivation by ADAM17. Nat Commun. 2011;2 : 229. doi: 10.1038/ncomms1232 21407195; PubMed Central PMCID: PMCPMC3074487.
43. Kyula JN, Van Schaeybroeck S, Doherty J, Fenning CS, Longley DB, Johnston PG. Chemotherapy-induced activation of ADAM-17: a novel mechanism of drug resistance in colorectal cancer. Clin Cancer Res. 2010;16(13):3378–89. doi: 10.1158/1078-0432.CCR-10-0014 20570921; PubMed Central PMCID: PMCPMC2896550.
44. Wheeler CM, Hunt WC, Cuzick J, Langsfeld E, Pearse A, Montoya GD, et al. A population-based study of human papillomavirus genotype prevalence in the United States: baseline measures prior to mass human papillomavirus vaccination. Int J Cancer. 2013;132(1):198–207. doi: 10.1002/ijc.27608 22532127.
45. Marur S, D'Souza G, Westra WH, Forastiere AA. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 2010;11(8):781–9. doi: 10.1016/S1470-2045(10)70017-6 20451455.
46. Thurlow JK, Peña Murillo CL, Hunter KD, Buffa FM, Patiar S, Betts G, et al. Spectral clustering of microarray data elucidates the roles of microenvironment remodeling and immune responses in survival of head and neck squamous cell carcinoma. J Clin Oncol. 2010;28(17):2881–8. doi: 10.1200/JCO.2009.24.8724 20458058.
47. Steller MA, Zou Z, Schiller JT, Baserga R. Transformation by human papillomavirus 16 E6 and E7: role of the insulin-like growth factor 1 receptor. Cancer Res. 1996;56(21):5087–91. 8895768.
48. McCredie MR, Sharples KJ, Paul C, Baranyai J, Medley G, Jones RW, et al. Natural history of cervical neoplasia and risk of invasive cancer in women with cervical intraepithelial neoplasia 3: a retrospective cohort study. Lancet Oncol. 2008;9(5):425–34. doi: 10.1016/S1470-2045(08)70103-7 18407790.
49. Harris TG, Burk RD, Yu H, Minkoff H, Massad LS, Watts DH, et al. Insulin-like growth factor axis and oncogenic human papillomavirus natural history. Cancer Epidemiol Biomarkers Prev. 2008;17(1):245–8. doi: 10.1158/1055-9965.EPI-07-0686 18199731.
50. Horn LC, Schreiter C, Canzler A, Leonhardt K, Einenkel J, Hentschel B. CD34(low) and SMA(high) represent stromal signature in uterine cervical cancer and are markers for peritumoral stromal remodeling. Ann Diagn Pathol. 2013;17(6):531–5. doi: 10.1016/j.anndiagpath.2013.05.009 24183311.
51. Pereira JJ, Meyer T, Docherty SE, Reid HH, Marshall J, Thompson EW, et al. Bimolecular interaction of insulin-like growth factor (IGF) binding protein-2 with alphavbeta3 negatively modulates IGF-I-mediated migration and tumor growth. Cancer Res. 2004;64(3):977–84. 14871828.
52. Diehl D, Hessel E, Oesterle D, Renner-Müller I, Elmlinger M, Langhammer M, et al. IGFBP-2 overexpression reduces the appearance of dysplastic aberrant crypt foci and inhibits growth of adenomas in chemically induced colorectal carcinogenesis. Int J Cancer. 2009;124(9):2220–5. doi: 10.1002/ijc.24193 19142966.
53. Frommer KW, Reichenmiller K, Schutt BS, Hoeflich A, Ranke MB, Dodt G, et al. IGF-independent effects of IGFBP-2 on the human breast cancer cell line Hs578T. J Mol Endocrinol. 2006;37(1):13–23. doi: 10.1677/jme.1.01955 16901920.
54. Holmes KM, Annala M, Chua CY, Dunlap SM, Liu Y, Hugen N, et al. Insulin-like growth factor-binding protein 2-driven glioma progression is prevented by blocking a clinically significant integrin, integrin-linked kinase, and NF-κB network. Proc Natl Acad Sci U S A. 2012;109(9):3475–80. doi: 10.1073/pnas.1120375109 22345562; PubMed Central PMCID: PMCPMC3295320.
55. Schütt BS, Langkamp M, Rauschnabel U, Ranke MB, Elmlinger MW. Integrin-mediated action of insulin-like growth factor binding protein-2 in tumor cells. J Mol Endocrinol. 2004;32(3):859–68. 15171717.
56. Perks CM, Vernon EG, Rosendahl AH, Tonge D, Holly JM. IGF-II and IGFBP-2 differentially regulate PTEN in human breast cancer cells. Oncogene. 2007;26(40):5966–72. doi: 10.1038/sj.onc.1210397 17369847.
57. Hodivala KJ, Pei XF, Liu QY, Jones PH, Rytina ER, Gilbert C, et al. Integrin expression and function in HPV 16-immortalised human keratinocytes in the presence or absence of v-Ha-ras. Comparison with cervical intraepithelial neoplasia. Oncogene. 1994;9(3):943–8. 8108139.
58. Galea CA, Mobli M, McNeil KA, Mulhern TD, Wallace JC, King GF, et al. Insulin-like growth factor binding protein-2: NMR analysis and structural characterization of the N-terminal domain. Biochimie. 2012;94(3):608–16. doi: 10.1016/j.biochi.2011.09.012 21951978.
59. Soh CL, McNeil K, Owczarek CM, Hardy MP, Fabri LJ, Pearse M, et al. Exogenous administration of protease-resistant, non-matrix-binding IGFBP-2 inhibits tumour growth in a murine model of breast cancer. Br J Cancer. 2014;110(12):2855–64. doi: 10.1038/bjc.2014.232 24853186; PubMed Central PMCID: PMCPMC4056053.
60. Chioni AM, Grose R. FGFR1 cleavage and nuclear translocation regulates breast cancer cell behavior. J Cell Biol. 2012;197(6):801–17. doi: 10.1083/jcb.201108077 22665522; PubMed Central PMCID: PMCPMC3373409.
61. Kaipparettu BA, Kuiatse I, Tak-Yee Chan B, Benny Kaipparettu M, Lee AV, Oesterreich S. Novel egg white-based 3-D cell culture system. Biotechniques. 2008;45(2):165–8, 70–1. doi: 10.2144/000112883 18687065.
62. Pickard A, Cichon AC, Menges C, Patel D, McCance DJ. Regulation of epithelial differentiation and proliferation by the stroma: a role for the retinoblastoma protein. J Invest Dermatol. 2012;132(12):2691–9. doi: 10.1038/jid.2012.201 22696061; PubMed Central PMCID: PMCPMC3443514.
63. Tang S, Tao M, McCoy JP, Zheng ZM. The E7 oncoprotein is translated from spliced E6*I transcripts in high-risk human papillomavirus type 16 - or type 18-positive cervical cancer cell lines via translation reinitiation. J Virol. 2006;80(9):4249–63. doi: 10.1128/JVI.80.9.4249–4263.2006 16611884; PubMed Central PMCID: PMCPMC1472016.
64. Senese S, Zaragoza K, Minardi S, Muradore I, Ronzoni S, Passafaro A, et al. Role for histone deacetylase 1 in human tumor cell proliferation. Mol Cell Biol. 2007;27(13):4784–95. doi: 10.1128/MCB.00494-07 17470557; PubMed Central PMCID: PMCPMC1951481.
65. Rochester MA, Riedemann J, Hellawell GO, Brewster SF, Macaulay VM. Silencing of the IGF1R gene enhances sensitivity to DNA-damaging agents in both PTEN wild-type and mutant human prostate cancer. Cancer Gene Ther. 2005;12(1):90–100. doi: 10.1038/sj.cgt.7700775 15499378.
66. McDade SS, Patel D, Moran M, Campbell J, Fenwick K, Kozarewa I, et al. Genome-wide characterization reveals complex interplay between TP53 and TP63 in response to genotoxic stress. Nucleic Acids Res. 2014. doi: 10.1093/nar/gku299 24823795.
Štítky
Hygiena a epidemiológia Infekčné lekárstvo Laboratórium
Článek Clearance of Pneumococcal Colonization in Infants Is Delayed through Altered Macrophage TraffickingČlánek An Model of Latency and Reactivation of Varicella Zoster Virus in Human Stem Cell-Derived NeuronsČlánek Protective mAbs and Cross-Reactive mAbs Raised by Immunization with Engineered Marburg Virus GPsČlánek Specific Cell Targeting Therapy Bypasses Drug Resistance Mechanisms in African TrypanosomiasisČlánek Peptidoglycan Branched Stem Peptides Contribute to Virulence by Inhibiting Pneumolysin ReleaseČlánek HIV Latency Is Established Directly and Early in Both Resting and Activated Primary CD4 T CellsČlánek Sequence-Specific Fidelity Alterations Associated with West Nile Virus Attenuation in Mosquitoes
Článok vyšiel v časopisePLOS Pathogens
Najčítanejšie tento týždeň
2015 Číslo 6- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Očkování proti virové hemoragické horečce Ebola experimentální vakcínou rVSVDG-ZEBOV-GP
- Koronavirus hýbe světem: Víte jak se chránit a jak postupovat v případě podezření?
-
Všetky články tohto čísla
- Introducing “Research Matters”
- Exploring Host–Pathogen Interactions through Biological Control
- Analysis of Bottlenecks in Experimental Models of Infection
- Expected and Unexpected Features of the Newly Discovered Bat Influenza A-like Viruses
- Clearance of Pneumococcal Colonization in Infants Is Delayed through Altered Macrophage Trafficking
- Recombinant Murine Gamma Herpesvirus 68 Carrying KSHV G Protein-Coupled Receptor Induces Angiogenic Lesions in Mice
- TRIM30α Is a Negative-Feedback Regulator of the Intracellular DNA and DNA Virus-Triggered Response by Targeting STING
- Targeting Human Transmission Biology for Malaria Elimination
- Two Cdc2 Kinase Genes with Distinct Functions in Vegetative and Infectious Hyphae in
- An Model of Latency and Reactivation of Varicella Zoster Virus in Human Stem Cell-Derived Neurons
- Protective mAbs and Cross-Reactive mAbs Raised by Immunization with Engineered Marburg Virus GPs
- Virulence Factors of Induce Both the Unfolded Protein and Integrated Stress Responses in Airway Epithelial Cells
- Peptide-MHC-I from Endogenous Antigen Outnumber Those from Exogenous Antigen, Irrespective of APC Phenotype or Activation
- Specific Cell Targeting Therapy Bypasses Drug Resistance Mechanisms in African Trypanosomiasis
- An Ultrasensitive Mechanism Regulates Influenza Virus-Induced Inflammation
- The Role of Human Transportation Networks in Mediating the Genetic Structure of Seasonal Influenza in the United States
- Host Delivery of Favorite Meals for Intracellular Pathogens
- Complement-Opsonized HIV-1 Overcomes Restriction in Dendritic Cells
- Inter-Seasonal Influenza is Characterized by Extended Virus Transmission and Persistence
- A Critical Role for CLSP2 in the Modulation of Antifungal Immune Response in Mosquitoes
- Twilight, a Novel Circadian-Regulated Gene, Integrates Phototropism with Nutrient and Redox Homeostasis during Fungal Development
- Surface-Associated Lipoproteins Link Virulence to Colitogenic Activity in IL-10-Deficient Mice Independent of Their Expression Levels
- Latent Membrane Protein LMP2A Impairs Recognition of EBV-Infected Cells by CD8+ T Cells
- Bank Vole Prion Protein As an Apparently Universal Substrate for RT-QuIC-Based Detection and Discrimination of Prion Strains
- Neuronal Subtype and Satellite Cell Tropism Are Determinants of Varicella-Zoster Virus Virulence in Human Dorsal Root Ganglia Xenografts
- Molecular Basis for the Selective Inhibition of Respiratory Syncytial Virus RNA Polymerase by 2'-Fluoro-4'-Chloromethyl-Cytidine Triphosphate
- Structure of the Virulence Factor, SidC Reveals a Unique PI(4)P-Specific Binding Domain Essential for Its Targeting to the Bacterial Phagosome
- Activated Brain Endothelial Cells Cross-Present Malaria Antigen
- Fungal Morphology, Iron Homeostasis, and Lipid Metabolism Regulated by a GATA Transcription Factor in
- Peptidoglycan Branched Stem Peptides Contribute to Virulence by Inhibiting Pneumolysin Release
- A Macrophage Subversion Factor Is Shared by Intracellular and Extracellular Pathogens
- A Novel AT-Rich DNA Recognition Mechanism for Bacterial Xenogeneic Silencer MvaT
- Reovirus FAST Proteins Drive Pore Formation and Syncytiogenesis Using a Novel Helix-Loop-Helix Fusion-Inducing Lipid Packing Sensor
- The Role of ExoS in Dissemination of during Pneumonia
- IRF-5-Mediated Inflammation Limits CD8 T Cell Expansion by Inducing HIF-1α and Impairing Dendritic Cell Functions during Infection
- Discordant Impact of HLA on Viral Replicative Capacity and Disease Progression in Pediatric and Adult HIV Infection
- Crystal Structure of USP7 Ubiquitin-like Domains with an ICP0 Peptide Reveals a Novel Mechanism Used by Viral and Cellular Proteins to Target USP7
- HIV Latency Is Established Directly and Early in Both Resting and Activated Primary CD4 T Cells
- HPV16 Down-Regulates the Insulin-Like Growth Factor Binding Protein 2 to Promote Epithelial Invasion in Organotypic Cultures
- The νSaα Specific Lipoprotein Like Cluster () of . USA300 Contributes to Immune Stimulation and Invasion in Human Cells
- RSV-Induced H3K4 Demethylase KDM5B Leads to Regulation of Dendritic Cell-Derived Innate Cytokines and Exacerbates Pathogenesis
- Leukocidin A/B (LukAB) Kills Human Monocytes via Host NLRP3 and ASC when Extracellular, but Not Intracellular
- Border Patrol Gone Awry: Lung NKT Cell Activation by Exacerbates Tularemia-Like Disease
- The Curious Road from Basic Pathogen Research to Clinical Translation
- From Cell and Organismal Biology to Drugs
- Adenovirus Tales: From the Cell Surface to the Nuclear Pore Complex
- A 21st Century Perspective of Poliovirus Replication
- Is Development of a Vaccine against Feasible?
- Waterborne Viruses: A Barrier to Safe Drinking Water
- Battling Phages: How Bacteria Defend against Viral Attack
- Archaea in and on the Human Body: Health Implications and Future Directions
- Degradation of Human PDZ-Proteins by Human Alphapapillomaviruses Represents an Evolutionary Adaptation to a Novel Cellular Niche
- Natural Variants of the KPC-2 Carbapenemase have Evolved Increased Catalytic Efficiency for Ceftazidime Hydrolysis at the Cost of Enzyme Stability
- Potent Cell-Intrinsic Immune Responses in Dendritic Cells Facilitate HIV-1-Specific T Cell Immunity in HIV-1 Elite Controllers
- The Mammalian Cell Cycle Regulates Parvovirus Nuclear Capsid Assembly
- Host Reticulocytes Provide Metabolic Reservoirs That Can Be Exploited by Malaria Parasites
- The Proteome of the Isolated Containing Vacuole Reveals a Complex Trafficking Platform Enriched for Retromer Components
- NK-, NKT- and CD8-Derived IFNγ Drives Myeloid Cell Activation and Erythrophagocytosis, Resulting in Trypanosomosis-Associated Acute Anemia
- Successes and Challenges on the Road to Cure Hepatitis C
- BRCA1 Regulates IFI16 Mediated Nuclear Innate Sensing of Herpes Viral DNA and Subsequent Induction of the Innate Inflammasome and Interferon-β Responses
- A Structural and Functional Comparison Between Infectious and Non-Infectious Autocatalytic Recombinant PrP Conformers
- Phosphorylation of the Peptidoglycan Synthase PonA1 Governs the Rate of Polar Elongation in Mycobacteria
- Human Immunodeficiency Virus Type 1 Nef Inhibits Autophagy through Transcription Factor EB Sequestration
- Sequence-Specific Fidelity Alterations Associated with West Nile Virus Attenuation in Mosquitoes
- EBV BART MicroRNAs Target Multiple Pro-apoptotic Cellular Genes to Promote Epithelial Cell Survival
- Single-Cell and Single-Cycle Analysis of HIV-1 Replication
- TRIM32 Senses and Restricts Influenza A Virus by Ubiquitination of PB1 Polymerase
- The Herpes Simplex Virus Protein pUL31 Escorts Nucleocapsids to Sites of Nuclear Egress, a Process Coordinated by Its N-Terminal Domain
- Host Transcriptional Response to Influenza and Other Acute Respiratory Viral Infections – A Prospective Cohort Study
- PLOS Pathogens
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- HIV Latency Is Established Directly and Early in Both Resting and Activated Primary CD4 T Cells
- Battling Phages: How Bacteria Defend against Viral Attack
- A 21st Century Perspective of Poliovirus Replication
- Adenovirus Tales: From the Cell Surface to the Nuclear Pore Complex
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



