-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
The miR9863 Family Regulates Distinct Alleles in Barley to Attenuate NLR Receptor-Triggered Disease Resistance and Cell-Death Signaling
Plants rely on cell-surface and intracellular immune receptors to sense pathogen invasion and to mediate defense responses. However, uncontrolled activation of immune responses is harmful to plant growth and development. Small RNAs have recently been shown to fine-tune the expression of intracellular immune receptors and contribute to the regulation of defense signaling in dicot plants, while similar processes have not been well documented in monocot grain crops, such as barley and wheat. Here, we show that, in barley, some members of the miR9863 family target a subset of Mla alleles that confer race-specific disease resistance to the powdery mildew fungus. These miRNAs act on Mla transcripts by cleavage and translational repression. Production of a type of trans-acting small RNAs, designated as phasiRNAs, enhances the effects of miRNA regulation on Mla targets. We propose that Mla-mediated immune signaling is fine-tuned by the miRNAs at later stage of MLA activation to avoid overloading of immune responses in barley cells.
Published in the journal: The miR9863 Family Regulates Distinct Alleles in Barley to Attenuate NLR Receptor-Triggered Disease Resistance and Cell-Death Signaling. PLoS Genet 10(12): e32767. doi:10.1371/journal.pgen.1004755
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1004755Summary
Plants rely on cell-surface and intracellular immune receptors to sense pathogen invasion and to mediate defense responses. However, uncontrolled activation of immune responses is harmful to plant growth and development. Small RNAs have recently been shown to fine-tune the expression of intracellular immune receptors and contribute to the regulation of defense signaling in dicot plants, while similar processes have not been well documented in monocot grain crops, such as barley and wheat. Here, we show that, in barley, some members of the miR9863 family target a subset of Mla alleles that confer race-specific disease resistance to the powdery mildew fungus. These miRNAs act on Mla transcripts by cleavage and translational repression. Production of a type of trans-acting small RNAs, designated as phasiRNAs, enhances the effects of miRNA regulation on Mla targets. We propose that Mla-mediated immune signaling is fine-tuned by the miRNAs at later stage of MLA activation to avoid overloading of immune responses in barley cells.
Introduction
Plants have evolved two major classes of immune receptors for pathogen recognition and defense activation [1]. Pattern recognition receptors (PRRs) detect conserved pathogen associated molecular patterns (PAMP) and trigger PAMP-triggered immunity (PTI) [2], whilst intracellular NB-LRR (NLR) receptors recognize isolate-specific pathogen effectors and trigger effector-triggered immunity (ETI), often accompanied by a hypersensitive reaction (HR) [3], [4]. PTI and ETI are believed to share highly overlapping signaling networks for effective immune responses [5]. A plant genome may contain hundreds of genes encoding NLR receptors [6]. Improper activation of some NLR receptors is accompanied by autoimmunity that is harmful to plant growth [7], [8], thus, expression of NLR genes is likely under tight control at different levels.
Plant small RNAs are noncoding RNAs that largely fall into two groups, microRNAs (miRNAs) and small interfering RNAs (siRNAs). Plant small RNAs have important roles in plant growth and development, abiotic stress responses and antiviral defense responses [9]–[11]. Increasing evidence indicates that plant small RNAs also participate in PTI and ETI responses against bacterial and fungal pathogens [12], [13]. For example, the miR393 and miR393* pair are involved in Arabidopsis PTI responses against Pseudomonas syringae pv. tomato (Pst) [14]–[16] and hvu-miR398 is regulated by the barley Mla NLR immune receptor, influencing ETI responses to the powdery mildew fungus [17]. Further, the natural antisense (NAT) RNA, nat-siRNAATGB2, and a long siRNA, lsiRNA-1, contribute to Arabidopsis RPS2-mediated ETI responses to Pst (avrRpt2) [18], [19]. Interestingly, recent examples indicate that plant small RNA pathways are required for defense responses against Verticllium dahliae [20] and are themselves targets of effectors secreted by oomycetes and fungi [21], [22].
MicroRNAs are 21–24 nucleotides (nt) endogenous small RNAs that repress gene expression in plants and animals [23]. In plants, the majority of miRNAs originating from MIR genes are processed by the DICER-LIKE1 (DCL1) enzyme from long primary miRNA transcripts that form imperfect stem-loop structures [9], [10], [24]. The mature miRNAs are loaded into an ARGONAUTE (AGO) protein to form functional RNA-induced silencing complex (RISC) for target gene silencing [25]. Plant miRNAs mediate target silencing through at least two modes of action, i.e., target mRNA cleavage and translational repression [24], [26]. Unlike animal miRNAs, plant miRNAs are highly complementary to their targets and earlier studies have shown that plant miRNAs guide target mRNA cleavage via AGO slicers [27]–[29], thus guiding RNA cleavage was believed to be the major activity of plant miRNAs [9]. Increasing evidence has indicated that plant miRNAs also inhibit target mRNA translation [30]–[36], and in some cases, plant miRNAs may regulate targets through both mRNA cleavage and translation inhibition [30], [32], [35]. Recent work has demonstrated that the ALTERED MERISTEM PROGRAM1 (AMP1)-dependent activity of miR398 in repressing translation of the CSD2 gene on the ER [37], further clarifying the role of plant miRNAs in inhibition of target gene translation.
The role of plant miRNAs in modulating immunity has recently been shown in diverse plant species, particularly their involvement in post-transcriptional control of NLR receptors [38]–[41]. In Medicago truncatula, highly abundant miRNA families, e.g., miR2109, miR2118 and miR1507, act as master regulators and target sites encoding highly conserved domains of a large group of NLR receptors [38], and this regulatory circuit can be extended into non-legume plant species, e.g., potato [38]. In N. benthamiana, two miRNAs, miR6019 and miR6020, were shown to regulate the TIR-NB-LRR (TNL)-type N receptor gene and to attenuate N-mediated resistance against Tobacco mosaic virus [39], while in other Solanaceae species the miR482/2118 superfamily and additional miRNA families target both the TNL - and CC-NB-LRR (CNL)-type receptor genes [39], [40]. Moreover, ath-miR472, a miR482-related Arabidopsis miRNA, was recently reported to act together with RDR6 to target CNL genes, including RPS5 and SUMM2, and to modulate both PTI and ETI against bacterial pathogens through post-transcriptional regulation of a subset of CNL genes [41].
Plant NLR-targeting miRNAs are in general of 22-nt in length, and they are able to trigger the production of secondary siRNAs that form a feed-forward regulation loop to amplify the suppression effect on NLR genes [38]–[42]. These trans - or cis-acting small interfering RNAs are usually in phase with the 5′-end of the cleavage site of the target transcripts, thus called phased secondary siRNAs (phasiRNAs) [43]. While miRNAs of 22-nt rather than 21-nt are believed to be the preferential triggers of phasiRNAs [44]–[46], the asymmetrical miRNA duplex structure is also important in triggering secondary siRNAs [47]. Despite the involvement of miRNAs and phasiRNAs in modulating plant immunity against pathogens in diverse plant species, the functional relevance of NLR-targeting miRNAs and the secondary siRNAs has not been well addressed in plant-fungal interactions, particularly for Poaceae species.
In barley, Mildew resistance locus a (Mla) contains a large number of alleles encoding NLR immune receptors that have verified or anticipated race-specific disease resistance activity against the powdery mildew fungus, Blumeria graminis f. sp. hordei (Bgh) [48]. Recent findings indicate that upon pathogen recognition MLAs dynamically integrate PTI and ETI immune signaling pathways in both nuclear and cytoplasmic compartments to mediate defense responses [49], [50]. Upon activation MLA associates in the nucleus with transcription factors (TFs), i.e., WRKY and MYB TFs, to initiate extensive transcriptional reprogramming that is critical for effective immune responses [51]–[55], whereas MLA triggers cell-death signaling in the cytosol through as yet unknown components that are probably conserved in monocots and dicots [56]–[58]. Recent data show that Mla mediates control of barley miR398 that represses chloroplast HvSOD1 accumulation, thus de-repressing HR cell-death signaling in response to Bgh challenge [17]. Previous studies have also shown that the activity of some MLAs is tightly regulated at post-translational level by the co-chaperone Required for Mla12 resistance 1 (RAR1) [59]–[61]. Nevertheless, whether and how Mla alleles are regulated at post-transcriptional level in barley remains unknown.
Here, we show that members of the Triticeae-specific miR9863 family differentially regulate a subset of barley Mla alleles at the post-transcriptional level. Unique single-nucleotide-polymorphism (SNP) sites in mature miR9863s and the miR9863-binding site of Mla alleles determine regulation specificity. Furthermore, we demonstrate in barley and N. benthamiana that miR9863s trigger the biogenesis of phasiRNAs and together these small RNAs form a regulation network for controlling Mla expression. Overexpression of miR9863 members specifically attenuates MLA1, but not MLA10-mediated disease resistance against Bgh in barley or induction of cell-death signaling in N. benthamiana, signifying the miR9863 family in fine-tuning immunity triggered by MLA receptors.
Results
Identification of the miR9863 family in barley and wheat
To investigate the role of miRNAs in barley (Hordeum vulgare L.) resistance to the powdery mildew pathogen, Bgh, we constructed small RNA (sRNA) libraries using samples derived from healthy and Bgh-infected leaves of the near-isogenic line P01 that harbors the Mla1 resistance allele [62]. Deep sequencing and data analyses of the sRNA libraries identified miRNAs that were either up - or down-regulated after Bgh infection. We searched in our sequence data for miRNAs potentially targeting Mla alleles and found that members of one miRNA family are complementary to the coding sequence for the region adjacent and with two-amino acids overlapping with the RNBS-D motif in Mla1 (Fig. 1A and 1D). Amino acid sequence alignment indicated that this potential miRNA target site is highly conserved among Mla alleles, as well as some R genes from wheat and related species (S1 Figure). The same miRNA family was also ascertained by an independent small RNA sequencing project [63] and described in previous small RNA deep sequencing studies in barley and bread wheat (Triticum aestivum L.). This family was originally designated in wheat as miR2009 with four members, i.e., miR2009a, miR2009b, miR2009c and miR2009d (S1 Table) [64]–[66]. However, the plant miR2009 was not formally included in miRBase20.0, while a sea urchin miRNA was already registered as miR-2009 [67]. Thus, the name of the wheat miR2009 was most likely assigned arbitrarily and thus bears the same name as that of the sea urchin miRNA. Therefore, we have renamed the plant miRNA family with its own unique designator in miRBase20.0, miR9863. We set out to search for barley and wheat EST sequences containing miR9863 members, and to BLAST wheat and barley genome databases. We found that miR9863a could be aligned to the wheat EST, CK193889, while miR9863c aligned to another wheat EST, DR736484 (S2A Figure). Interestingly, miR9863b and miR9863d overlap by 17 nucleotides, and cluster with miR9863a on the same wheat EST (i.e., CK193889) with 244 nt in-between (S2A Figure; S1 Table). In barley, miR9863b and miR9863d also align to one barley cDNA sequence, AK364228, overlapping by 17 nt (S2A Figure; S1 Table). Since miR9863b and miR9863d are two miRNAs that likely resulted from overlapping processing of the same locus, we designated these two miRNAs as miR9863b.1 and miR9863b.2 (Fig. 1A; S1 Table) [68]. The miRNA and miRNA* sequence and the flanking EST sequences together can form a typical hairpin structures, with the miRNA and miRNA* on the stem region (Fig. 1B; S2A Figure). Thus, these analyses indicate that the miR9863 family is present in the Triticeae, and members of this family could be potential regulators of Mla alleles.
Fig. 1. miR9863 family members target Mla1 transcripts in barley. 
(A) Alignment of Mla1 transcript with miR9863 family members from barley and wheat. Shown above the alignment are the MLA1 domain structure (CC, NB, ARC and LRR), the amino acid sequence of the miRNA target region and the neighboring RNBS-D motif (in red). hvu, Hordeum vulgare; tae, Triticum aestivum. (B) Predicted secondary structures of hvu-MIR9863a, hvu -MIR9863b and tae-MIR9863c precursors. Since hvu-MIR9863c could not be amplified from barley cultivars (i.e. Pallas and Golden promise), we used tae-MIR9863c for generating miR9863c in this study. The hvu-MIR9863b precursor generates both miR9863b.1 and miR9863b.2 that partially overlap with each other. (C) Ethidium bromide stained agarose gel of 5′ RACE products obtained from the barley line P01 (a Pallas near-isogenic line containing the Mla1 allele). 5′ RACE products were generated by using total RNAs isolated from P01, and ‘a’, ‘b’, and ‘c’ indicate three different products derived from different cleavage sites (see below). (D) Cleavage sites in the Mla1 transcript confirmed by the sequencing of 5′ RACE products. Arrows indicate the cleavage sites, ‘a’, ‘b’ and ‘c’, corresponding to three products shown in (C); numbers below the arrows show frequency of clones with matching 5′-RACE product from this site out of total clones confirmed by sequencing. Next we attempted to isolate the precursors of the miR9863 family from both barley and wheat. While we could readily amplify all three progenitors from wheat, i.e., MIR9863a, MIR9863c and MIR9863b, we could only amplify two progenitors from barley, MIR9863a and MIR9863b, but not MIR9863c (Fig. 1B). MIR9863c also was not present in an independent deep sequencing data set of barley cv. Morex, both from non-inoculated and Bgh inoculated leaves [63]. It is possible that miR9863c may be genotype specific in barley, or an intron-derived microRNA.
Sequence analysis revealed that the MIR9863a progenitor from barley and wheat are highly conserved, and the same is true for MIR9863b (S2A Figure). Since MIR9863a and MIR9863b are aligned in tandem on the same wheat EST (CK193889), this suggests the existence of a wheat polycistron of MIR9863a-MIR9863b, which may not be present in barley (S2A Figure). To test whether these potential miRNA progenitors can generate miRNAs, we transiently expressed them in N. benthamiana by Agrobacterium-mediated infiltration and found the accumulation of 22-nt mature miRNAs in all cases (S2B Figure). Further, we examined the expression of the miR9863 family in different plant species (S3 Figure). Interestingly, we found that they are expressed in all tested barley and bread wheat cultivars, but do not accumulate in other tested monocots, e.g., Brachypodium distachyon, rice (Oryza sativa) and maize (Zea mays), nor in the dicots N. benthamiana and A. thaliana (S3 Figure). Although searching of the monocot small RNA library using miR9863 sequences found some sorghum small RNAs matching miR9863a [63], however, we could not further retrieve any flanking sequences from sorghum EST or genome database that can form a miRNA precursor structure [69]. Furthermore, searching of several sRNA libraries from Brachypodium distachyon [66], [70], [71], which has been served as a model for the Triticeae tribe, did not find any matching sequences to the miR9863 sequences. We also screened NCBI genome and EST databases of rice and Arabidopsis using the mature miR9863 sequences (allowing no more than 2 mismatches), but failed to identify a single match. Together, these suggest that the MIR9863 loci may be Triticeae-specific innovation.
The miR9863 family targets barley Mla1 for cleavage
To examine the function of the miR9863 members in directing Mla1 transcript cleavage in barley, we conducted 5′-RACE using Bgh infected P01 (harboring the Mla1 allele) leaf samples. We could detect three major cleavage products from Mla1 transcripts after gel-electrophoresis (Fig. 1C, marked by ‘a’, ‘b’, and ‘c’). Sequencing of clones derived from band ‘a’ revealed two cleavage positions within the miR9863-binding site in Mla1 transcripts (Fig. 1C and 1D). To test the specificity of miR9863a binding, we conducted site-directed mutagenesis by replacing the ‘T’ with an ‘A’ at the position next to the 1st cleavage site, and ‘CT’ with ‘GA’ across the 2nd cleavage site, resulting in Mla1-T1266A and Mla1-CT1270GA. This should result in a loop at the respective cleavage site in the miRNA/target duplex (S4A Figure). We next determined the efficiency of miR9863a on wild-type (WT) Mla1 and the two Mla1 variants by transient expression in N. benthamiana. Upon co-expression of MIR9863a with WT Mla1, MLA1 protein was decreased to a level that was barely detectable by Western blot analysis (S4B Figure, lane 2). By contrast, MLA1-CT1270GA or MLA1-T1266A protein abundance was not affected or only slightly reduced (S4B Figure, lanes 3 and 4, 5 and 6, respectively), compared to the empty vector (EV) controls (S4B Figure, lane 1). These results suggest the strict pairing at the 10th to 11th nt and the 15th nt positions in the miR9863a-binding site of Mla1 is necessary for proper regulation of miR9863a. Sequencing of clones derived from band ‘b’ and ‘c’ revealed two additional cleavage sites downstream of the miR9863-binding position (Fig. 1C and 1D). We speculate that these two cleavage sites were derived from in-phase degradation triggered by secondary siRNAs (see below). In summary, the potentially Triticeae-specific miR9863 family has four members, and some members of this miRNA family direct the cleavage of Mla1 transcripts at the miRNA-binding site.
miR9863 family members differentially regulate Mla1
Functional differentiation is usually observed among different miRNA members that belong to the same family [31], [72], [73]. To determine the specificity of individual miR9863 members for Mla1, we co-expressed C-terminal HA-tagged MLA1 with progenitor MIR9863a, MIR9863b or MIR9863c, respectively, in N. benthamiana through Agrobacterium-mediated infiltration (Fig. 1B; Fig. 2). MLA1 accumulation was fully or significantly blocked by the expression of MIR9863a or MIR9863b but not by MIR9863c, compared to EV control (Fig. 2A, 2B and 2C). Since MIR9863b should simultaneously generate miR9863b.1 and miR9863b.2 (Fig. 1B), we could not differentiate the effect of these two miRNAs (Fig. 2B). To address this, we utilized the Arabidopsis precursor Ath-MIR173 as a backbone and constructed artificial miRNA expression vectors, aMIR9863a, aMIR9863b.1 and aMIR9863b.2 (S5 Figure) [17], [44], [74]. In N. benthamiana these aMIRNA precursors could respectively generate miR9863a, miR9863b.1, or miR9863b.2 to a comparable level (Fig. 2D). Further co-expression of aMIR9863a with Mla1 fully repressed MLA1 accumulation in N. benthamiana (Fig. 2E, lanes 1 and 2). Importantly, aMIR9863b.1 expression could largely block MLA1 accumulation whilst aMIR9863b.2 expression has only a subtle effect, resulting in accumulation of MLA1 at ∼15%, or ∼60% of that in EV control, respectively (Fig. 2E, lanes 3 and 4, lanes 5 and 6). These results demonstrate that Mla1 is differentially regulated by miR9863 members, with the highest efficiency specified by miR9863a and miR9863b.1, less so by miR9863b.2, and none by miR9863c.
Fig. 2. miR9863a, miR9863b.1 and miR9863b.2, but not miR9863c, regulate Mla1. 
(A) to (C) miR9863 family members differentially regulate Mla1 gene shown by transient gene expression assay in N. benthamiana. Agrobacteria strain expressing Mla1-3HA were co-infiltrated into N. benthamiana leaves with strains containing the respective miRNA precursors, hvu-MIR9863a (A), hvu-MIR9863b (B) or tae-MIR9863c (C). Western blotting was used to detect MLA1-3HA or actin levels at 36 hours post Agro-infiltration (hpai); and the number below the blots indicates relative protein levels as calculated by Image J. Empty vector (EV) was used as a negative control and rubisco served as a loading control. The asterisks indicate non-specific signals. Experiments were performed at least two times with similar results. (D) Expression of mature miR9863a, miR9863b.1 and miR9863b.2 using artificial MIRNA backbone. The mature miR9863a, miR9863b.1 or miR9863b.2 sequences were engineered into the Arabidopsis MIR173 precursor (see S5 Figure) and then artificial MIRNA precusors (aMIRNA) were expressed in N. benthamiana by Agro-infiltration. The levels of mature miR9863s were detected by RNA gel blot analysis at 36 hpai. miR156 and 5S rRNA were employed as loading controls. (E) Determination of the regulation efficiency of miR9863a, miR9863b.1 or miR9863b.2 on Mla1. The aMIR9863a, aMIR9863b.1 or aMIR9863b.2 was respectively co-expressed with Mla1 in N. benthamiana, and MLA1 protein levels were determined at 36 hpai by immunoblotting. It is interesting to note that miR9863a and miR9863b.2 differ markedly in their regulation of Mla1, but have only a single nucleotide difference (Fig. 1A; S6A Figure). To further characterize this observation, we introduced a ‘T’ to ‘C’ point mutation in the MIR9863a progenitor, resulting in MIR9863a-T9C that should produce a miR9863a variant sequentially same as miR9863b.2 (S6B Figure). Indeed, unlike MIR9863a that fully blocked MLA1 accumulation, MIR9863a-T9C co-expression with Mla1 led to high MLA1 level to ∼80% of that in EV control (S6C Figure, panel a); RNA gel-blotting detected equal expression level of the respective miRNAs (S6C Figure, panel b). These data indicate that a single nucleotide difference between miR9863a and miR9863b.2 can influence the regulation of Mla1.
miR9863 family members regulate only a subset of Mla genes
Cloned Mla alleles encode highly sequence related CNL-subtype NLR receptors [48], [59], [75]–[78]. Sequence alignment using 29 Mla short sequences covering only the miR9863 target site shows that these Mla sequences are almost identical except for 1 or 2 adjacent nucleotides (Fig. 3A). These two adjacent nucleotides were predicted to complement with the 2nd–3rd nt of miR9863a/b.2 or the 7th–8th nt of miR9863b.1, respectively. Based on these SNP haplotypes, 29 Mla alleles were classified into three groups, group I, II and III (Fig. 3A). To determine whether there are differences in miR9863 regulation on different Mla groups, we randomly selected from each group two Mla genes, i.e., Mla28 and Mla32 from group I, Mla2 and Mla6 from group II, and Mla10 and Mla12 from group III, and each of them was co-expressed with precursor MIR9863a or MIR9863b in N. benthamiana, respectively (Fig. 1B, Fig. 3B). Western analysis revealed that the accumulation of MLA28 and MLA32 (group I) was fully blocked by expression of MIR9863a and partially blocked by expression of MIR9863b (Fig. 3B, left panels). By contrast, the accumulation of MLA2 and MLA6 (group II, Fig. 3B middle panel), or MLA10 and MLA12 (group III, Fig. 3B right panel) were not affected by expression of MIR9863a or MIR9863b, compared to the EV control. Together, these data strongly suggest that only group I Mla members are regulated by miR9863a and miR9863b.1/b.2 in planta.
Fig. 3. miR9863a and miR9863b.1/b.2 specifically regulate group I Mla alleles. 
(A) Mla alleles are classified into three groups according to the SNP haplotype in the miR9863 binding site. The two SNPs differ among Mla groups are highlighted. (B) Mla alleles of group I, but not group II and III, are regulated by miR9863a and miR9863b.1/b.2. Mla genes of group I (Mla28, Mla32), group II (Mla2, Mla6) and group III (Mla10, Mla12) were respectively co-expressed with either MIR9863a (upper panels) or MIR9863b (lower panels) in N. benthamiana as described in Fig. 2. Protein levels of MLA or actin were determined by immunoblotting with an anti-HA or anti-actin antibody; Rubisco was included as a loading control. The asterisks indicate non-specific signals. To further verify the above finding that 1–2 SNPs in the Mla allele miR9863-binding site dictate miRNA specificity, we conducted targeted mutagenesis at these nt positions for each individual Mla from groups I, II, or III (Fig. 4A). First, we replaced the ‘TC’ (nt at 1278 and 1279) in Mla1 (from group I) with ‘GC’ or ‘GA’ to mimic the SNP haplotype of group II or III Mla, and reciprocally we replaced ‘GC’ in Mla2 and Mla6, as well as ‘GA’ in Mla10 and Mla12 to ‘TC’ to mimic the SNP haplotype of group I Mla (Fig. 4A). Upon co-expression of MIR9863a with WT Mla1 or two Mla1 variants (Mla1-TC1278GC, Mla1-TC1278GA), we could not detect by Western analysis any WT MLA1 accumulation, but the levels of two MLA1 variants are comparable to that in the EV control (Fig. 4B), suggesting loss of miR9863a effects on mutated Mla1 genes. In contrast, when MIR9863a was co-expressed with WT Mla2 and Mla6, or the respective Mla variants (Fig. 4C), as well as with WT Mla10 and Mla12, or their variants (Fig. 4D), we detected the expression of the WT MLAs similar to EV control, but no accumulation was detected for all mutant variants, compared to the EV control (Fig. 4C and 4D). This indicates a gain of function for miR9863a for Mla variant genes that mimic the group I Mla SNP haplotype. We obtained similar gain of function results for MIR9863b (S7 Figure). These results suggest that miR9863 targeting specificity on Mla alleles is largely determined by the two SNP compositions in the miR9863-binding site of each Mla.
Fig. 4. SNP variations in Mla alleles dictate miR9863 specificity. 
(A) Alignment of miR9863 target site of WT and mutated Mla alleles. SNPs in bold font and color-coded represent Mla type of group I (black), II (magenta) or III (red). Nucleotide substitutions TC1278GC or TC1278GA were introduced into Mla1; GC1278TC into Mla2 and Mla6, and GA1278TC into Mla10 and Mla12. (B) to (D) Determination of miR9863a regulation specificity on WT or mutated Mla alleles from different groups. Using transient gene expression assays in N. benthamiana, Hvu-MIR9863a was respectively co-expressed with group I Mla1 alleles (B), or group II Mla2 and Mla6 alleles (C), or group III Mla10 and Mla12 alleles (D). The protein accumulation levels of indicated Mla isoforms and actin were determined by immunoblotting. The asterisks indicate non-specific signals. To further confirm miRNA-Mla specificity in barley, we conducted virus-induced gene silencing (VIGS) to knock-down expression of the miR9863 family in barley (Fig. 5). We first optimized a VIGS system by coupling a modified Barley stripe mosaic virus (BSMV)-induced gene silencing system with the expression of a short tandem target mimic (STTM), i.e., STTM-miR9863, designed to bind all barley miR9863 members (Fig. 5A) [79], [80] (see Materials and Methods). BSMV viral particles harboring the STTM-miR9863 structure were obtained from N. benthamiana and used to infect the near-isogenic barley line P01 (Mla1) or P03 (Mla6) (Fig. 5B, left and right half). As expected, we observed significantly reduced miR9863 accumulation in both P01 and P03 by BSMV: STTM-miR9863, compared to the BSMV-STTM-EV control by RNA blot analysis, while the accumulation of the non-related miR156 was not affected in both lines (Fig. 5B, panel a). Further qRT-PCR analyses validated the reduction of miR9863a/b.2 or miR9863c/b.1 in both lines infected with BSMV: STTM-miR9863, compared to BSMV-STTM-EV (Fig. 5B, panel b). Importantly, the reduced level of these miRNAs was inversely correlated with the increased Mla1 transcript level in P01, in contrast to the unchanged Mla6 transcript level in P03 (Fig. 5B, panel c). These data further verified that miR9863 family members specifically regulate group I Mla alleles. In summary, our results demonstrate that only a subset of Mla alleles are regulated by miR9863a and miR9863b.1/b.2, and two SNPs in the miR9863-binding site of Mla dictate miR9863 specificity.
Fig. 5. BSMV-VIGS silencing of miR9863 leads to increased transcript level of Mla1 but not Mla6 in barley. 
(A) Schematic representation of STTM-EV and STTM-miR9863 fragments used for the construction of pCaBS-γSTTM-EV and pCaBS-γSTTM-miR9863 for BSMV-mediated silencing (see Materials and Methods). (B) Comparison of expression levels of miR9863 members and Mla in BSMV infected barley plants. Pallas near-isogenic lines P01 (Mla1) and P03 (Mla6) were infected by BSMV harboring STTM-EV or STTM-miR9863 constructs, and systemic leaves with typical BSMV symptoms were collected for analysis. The level of miR9863 members was analyzed by RNA gel blot using miR156 as a loading control (panel a), or by qRT-PCR normalized against U6 (panel b). Mla1 and Mla6 expression levels were quantified by qRT-PCR normalized against actin (panel c). miRNA-directed phasiRNAs are critical for miR9863 regulation of Mla
Apart from many Bgh-responsive miRNA families in the sRNA libraries derived from infected barley leaf tissue (above, and see Materials and Methods), we identified additional sRNAs that perfectly match the Mla1 sense and antisense strand and are aligned downstream of the miR9863 target site (Fig. 6A, upper panel). These sRNAs are predominantly 21-nt in length (Fig. 6B) and biased for adenosine (A, ∼35%) and uridine (U, ∼20%) at the 5′-end position (Fig. 6C). Of the two abundant sRNAs at the 5′-proximal, one starts at 126 bp downstream of the miR9863a cleavage site (i.e., 1397 bp in Mla1), which corresponds to the 7th 21-nt register from the cleavage site; and the other starts at 504 bp downstream of the miR9863a cleavage site and corresponds to the 25th 21-nt register from the cleavage site (Fig. 6A, lower panel). These sRNAs are likely phasiRNAs [43] derived from the Mla1 transcripts, thus, for convenience of further analysis these two 21-nt register siRNAs are designated as phasiRNAI and phasiRNAII, respectively. To understand whether the biogenesis of these phasiRNAs are directly linked to miRNA action on its target, we co-expressed MIR9863b or MIR9863a with Mla1 in the heterologous N. benthamiana and then quantified the expression level of phasiRNAI and phasiRNAII (Fig. 6D, 6F and 6G). We found that phasiRNAI was significantly increased to ∼8 or 45 fold (Fig. 6F, bars 3 and 5), and phasiRNAII was increased to ∼2 to 2.5 fold (Fig. 6G, bars 3 and 5), compared to EV co-expressed with Mla1 (Fig. 6F and 6G, bar 2). These results indicate that miR9863b.1/b.2 and miR9863a trigger the biogenesis of phasiRNAs with Mla1 in N. benthamiana, and are consistent with our sRNA deep sequencing data obtained from barley.
Fig. 6. 22-nt miR9863s trigger the production of phasiRNAs with Mla1 transcripts. 
(A) Alignment of Mla1 sequence with 21-nt phased phasiRNAs. Raw reads of barley 21-nt phasiRNAs (y axis) obtained from deep sequencing were mapped to the sense strand (blue lines above the x axis) or antisense strand (red lines below the x axis) of the Mla1 sequence. Mla1 sequence adjacent to the miR9863 cleavage site was shown below the plot, and horizontal brackets below the sequence indicate 21-nt phasiRNAs, of which two most abundant ones (phasiRNAI and phasiRNAII) are indicated in the plot and in the sequence (in red). (B) Length of Mla1 phasiRNAs (x axis) was plotted to their raw reads value (y axis) obtained by sRNA deep sequencing in barley. (C) 5′-terminal nucleotide bias (x axis) of Mla1 phasiRNAs determined by raw reads (y axis) obtained by sRNA deep sequencing in barley. (D) Predicted secondary structure of precursors for expressing natural 22-nt miRNAs (MIR9863b, MIR9863a) and mutated 21-nt miRNAs (MIR9863b21 and MIR9863a21). (E) miR9863b.1/b.2 and miR9863a of 22-nt regulate Mla1 with higher efficiency than the mutated miRNAs of 21-nt. The mature miRNAs were detected by RNA gel blotting (panel a) with the mixture of [γ-32P]ATP labeled probes for miR9863a and miR9863b.1 (see supplemental Table 2 online), and 5S rRNA was employed as a loading control. MLA1 and actin protein levels were determined by immunoblotting (panel b), and rubisco is shown as loading control. (F) to (G) miR9863b.1/b.2 and miR9863a of 22-nt trigger the biogenesis of phasiRNAs with Mla1. Mla1 was co-expressed with EV or indicated miR9863 precursor in N. benthamiana, and the relative levels of phasiRNAI (F) and phasiRNAII (G) were measured by stem-loop quantitative RT-PCR (qRT-PCR) and normalized to U6 level. Letters above the bars (a-e) represent groups with significant differences [p<0.05, Tukey's honest significant difference (HSD) test]. Previous studies have demonstrated that miRNAs of 22-nt, rather than 21-nt, trigger secondary siRNA [38], [39], [43]–[45]. To test whether this also applies to miR9863 for Mla-dependent secondary siRNA production, we engineered the precursor of MIR9863b or MIR9863a by adding a cytosine in the miR9863* sequence to remove the asymmetric structure, resulting in MIR9863b21 and MIR9863a21 (Fig. 6D). This construct is predicted to produce 21-nt miRNAs that retain target-binding and cleavage activity [39], [44], [45]. Indeed, upon co-expression of each of these precursors with Mla1 in N. benthamiana, we detected by RNA gel blotting that MIR9863b21 and MIR9863a21 predominantly generated 21-nt miRNAs while the natural MIRNA precursors produced 22-nt miRNAs (Fig. 6E, panel a). Significantly, compared to the natural MIRNA precursors, MIR9863b21 and MIR9863a21 less effectively repressed MLA1 accumulation (Fig. 6E, panel b). Interestingly, we also observed marked reduction of secondary siRNAs, representing by phasiRNAI and phasiRNAII, in samples expressing mutated precursors (Fig. 6F and 6G, bar 4 and 6), compared to the WT precursors (Fig. 6F and 6G, bar 3 and 5). Together, these data suggest that the ineffective Mla1 regulation by the 21-nt-long miRNAs is partially due to the reduced level of secondary siRNAs. This was further supported by co-expressing MIR9863a or MIR9863a21 with a short Mla1 fragment encoding only the ARC domain fused with the mYFP-3HA tandem tag (S8 Figure). In samples expressing miR9863a21 of 21-nt, we detected almost no phasiRNAs by RNA gel blotting, compared to the substantial phasiRNA accumulation in samples expressing 22-nt natural miR9863a (S8B Figure, panel a). Similarly, the level of phasiRNAs was inversely correlated with the abundance of MLA1_ARC fusion protein (S8B Figure, panel b). In summary, these results demonstrate that 22-nt natural miR9863s are critical for the biogenesis of phasiRNAs of 21-nt in barley and N. benthamiana, and that these secondary phasiRNAs are important for effective miR9863 regulation of Mla target genes.
miR9863-mediated Mla regulation likely requires AGO1
The sorting of miRNAs to different AGO proteins is complex and influenced by several factors [24], [25], for example, the 5′ nucleotide identity and the length of miRNAs, and miRNA duplex structure. Arabidopsis AGO1 has been shown to associate with miRNAs with 5′ U [81] and to trigger the production of secondary siRNA when binding 22-nt miRNAs [25]. AGO genes have not yet been formerly annotated in barley genome although some sequences share high similarity with AtAGO1. To test whether AGO1 might be involved in miR9863-mediated regulation of Mla, we took advantage of the Tobacco rattle virus (TRV) mediated-VIGS to knock-down AGO1 in N. benthamiana using AGO4 as a control (S9 Figure). Arabidopsis AGO4 was shown to associate primarily with 24-nt siRNAs [81], [82]. As a technical control, the TRV-VIGS silencing of PHYTOENE DESATURASE (NbPDS) worked effectively, resulting in clear photobleaching phenotype on the upper systemic leaves (S9C Figure). Both NbAGO1 and NbAGO4 possess two alleles each, i.e., NbAGO1-1 and NbAGO1-2, NbAGO4-1 and NbAGO4-2, and these alleles share high sequence similarity [83]. Indeed, in our TRV-VIGS assays we observed co-suppression of both alleles of NbAGO1 or NbAGO4 when either one of the alleles was targeted for silencing (S9B Figure, top and middle panel). Interestingly, upon co-expression of Mla1 with MIR9863b or MIR9863a, MLA1 accumulation was similarly suppressed in TRV: 00 treated plants or NbAGO4-silenced plants (S9D Figure, 1st, 4th and 5th column). This is in contrast to NbAGO1-silenced plants, where MLA1 accumulation was unaffected, as compared to the TRV: 00 controls (S9D Figure, 2nd and 3rd column); the expression level of miR9863b or miR9863a was similar in all samples, however. Together, these data indicate that NbAGO1 rather than NbAGO4 is required for miR9863-mediated regulation of Mla1 in N. benthamiana.
Translational repression may contribute to miR9863-mediated regulation of Mla
Our data so far have showed that miR9863-directed Mla transcript cleavage and degradation play a role in the regulation of Mla. To examine whether translational inhibition directed by miR9863 may contribute to MLA suppression, again we co-expressed MIR9863a with Mla1 in N. benthamiana and then quantified Mla1 transcripts in parallel with MLA1 protein levels (Fig. 7). We monitored the level of three Mla1 amplicons (amplicon 1, Mla1407–783; amplicon 2, Mla11228–1582; amplicon 3 Mla12465–2621), positioned respectively upstream, over and downstream of the miR9863a cleavage site to reflect the levels of transcription, cleavage and cleavage-triggered decay of Mla1 transcripts (Fig. 7A) [84], at 16, 24, and 36 hours-post-Agro-infiltration (hpai). The corresponding MLA1 protein level was determined in samples collected 2 hrs later for each time point, allowing sufficient time for translation (Fig. 7B) [84]. At 16 hpai, we detected a basal level of the three Mla1 amplicons, but no accumulation of MLA1 protein (Fig. 7B, lanes 2 and 3). Later, at 24 hpai, we detected increased accumulation of Mla1 amplicons, as well as the presence of MLA1 protein in samples co-expressing the EV control (Fig. 7B, lane 4). However, the levels of the three Mla1 amplicons in samples co-expressing MIR9863a were 49–67% of that in EV control and MLA1 protein remained undetectable in the same samples (Fig. 7B, lane 5). At 36 hpai, Mla1 amplicons and MLA1 protein further increased in the EV control (Fig. 7B, lane 6), but the three Mla1 amplicons in MIR9863a-expressing samples were only 29–39% of the EV control (Fig. 7B, lane 7 vs. lane 6). It should be noted that these Mla1 transcript levels in the MIR9863a-expressing samples at 36 hpai are higher than those in the EV controls at 24 hpai (Fig. 7B, lane 7 vs. lane 4), yet MLA1 protein accumulation was not observed at 36 hpai, even though MLA1 protein was detected in the EV control at 24 hpai (Fig. 7B, lane 7 vs. lane 4). These findings suggest that in N. benthamiana Mla1 transcript levels have been uncoupled from accumulation of MLA1 protein in samples that Mla1 was co-expressed with miR9863, indicating apart from miR9863a-mediated transcript cleavage and decay, miR9863a-directed translational repression also plays a role in the regulation of Mla1 in this heterologous expression system.
Fig. 7. miR9863a regulates Mla1 mRNA and MLA1 protein in an uncoupled manner. 
(A) Schematic representation of Mla1-encoded domain structure and positions of amplicons for qRT-PCR quantification of Mla1 mRNA level. Amplicon 1, 2 or 3 positions at 5′, over or 3′ of the miR9863 cleavage site, respectively. (B) Quantification of Mla1 transcript and protein level at different time points post co-expression of Mla1-3HA with MIR9863a in N. benthamiana. The relative levels of three Mla1 amplicons were analyzed for samples of three time points, 16 and 24 and 36 hours-post-Agro-infiltration (hpai), by qRT-PCR quantification. For each amplicon, relative level was shown as fold-changes compared to the level at 16 hpai after first normalized to actin for each time point. The percentage above the bars indicates the remaining level of the amplicons relative to that of the EV control. Samples used for protein quantification were collected at 2 hrs after taking samples for mRNA quantification allowing sufficient time for protein translations. Western blots shown are exposed for more than 2 hrs. The asterisks indicate non-specific signals. All the experiments were performed at least twice with similar results. Overexpression of miR9863 members attenuates MLA-triggered disease resistance and cell-death signaling
To investigate the role of miR9863 members in MLA-mediated disease resistance to Bgh, we used a well-established single-cell transient assay [77], [85]. Plasmid constructs of miRNA precursor are co-delivered with a β-glucuronidase (GUS) reporter into barley epidermal cells by particle bombardment, and then the haustorial index (HI%) is scored to assess the frequency of fungal haustoria in transformed cells upon fungal inoculation (Fig. 8A and 8B). First, we respectively delivered plasmids of EV, MIR9863a, MIR9863c, or MIR9863b into near-isogenic barley line P01 (Mla1) and then inoculated conidiospores of Bgh K1 (AVRa1) to activate MLA1-mediated immune responses (Fig. 8A, left panel). We observed a low HI% of ∼3% in leaves receiving EV control, likely due to MLA1-triggered immunity against Bgh K1 (Fig. 8A, bar 1), whereas in leaves expressing MIR9863a or MIR9863b, we observed significantly increased HI% to ∼22% or 15% (Fig. 8A, bars 2 and 4), indicating MLA1-triggered immunity was compromised by the expression of miR9863a or miR9863b.1/b.2. The HI% was similar in P01 leaves expressing MIR9863c to that in the EV control (Fig. 8A, bar 3), consistent with that miR9863c does not interact with the Mla1 allele (Fig. 2C). Further, plasmids of MIR9863a, MIR9863c and MIR9863b were respectively delivered into leaves of near-isogenic barley line P09 (Mla10) and inoculated with Bgh isolate A6 (AVRa10) (Fig. 8A, right panel), and we observed comparable HI% of ∼6% to 7% in leaves expressing EV or respective miR9863 precursors (Fig. 8A, bars 5 to 8), suggesting that expression of any miR9863 members had no effect on Mla10-mediated disease resistance to Bgh, consistent with that Mla members of group III are not miR9863 targets (Fig. 3B). Taken together, overexpression of miR9863a and miR9863b.1/b.2 specifically attenuates MLA1 but not MLA10-mediated disease resistance against Bgh in barley.
Fig. 8. Overexpression of MIR9863a and MIR9863b attenuates Mla1-triggered disease resistance and cell-death signaling. 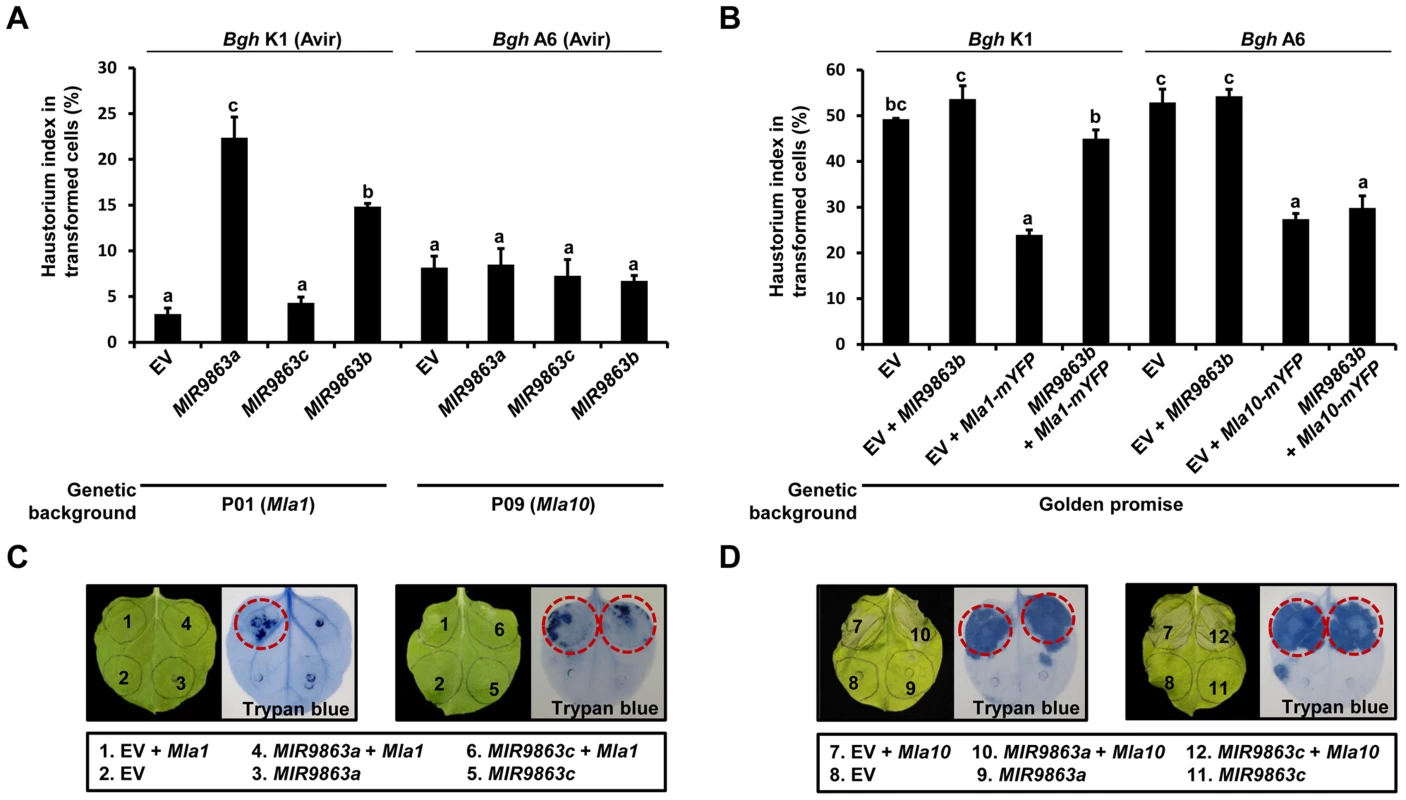
(A) Transient gene expression of MIR9863a, MIR9863c and MIR9863b in barley near-isogenic line containing Mla1 or Mla10. Empty vector or plasmids expressing indicated MIRNA precursors were delivered into leaf epidermal cells of indicated barley lines through particle bombardment, and spores of indicated avirulence Bgh isolates were inoculated 36 hrs post bombardment. Haustorial index shown are the percentage of cells that developed fungal haustoria in all transformed cells. (B) Transient gene expression assays in Golden promise (GP), a barley line susceptible to both Bgh K1 and Bgh A6. Indicated construct expressing MIR9863b was delivered alone or with Mla1-mYFP or Mla10-mYFP construct into GP leaf epidermal cells, and spores of Bgh K1 or Bgh A6 were inoculated at 4 hrs post bombardment. Error bars in (A) and (B) represent standard error (SE) from at least three replicates, and letters (a–c) above the bars represent groups with significant differences [p<0.05, Tukey's honest significant difference (HSD) test]. (C) to (D) Analysis of cell-death induction upon co-expressing Mla1 or Mla10 with MIR9863a and MIR9863c. The indicated constructs were expressed in N. benthamiana leaves by Agro-infiltration, and cell death was visualized by trypan blue staining at 48 hours-post-Agro-infiltration (hpai). Red circle indicates visible cell-death phenotype. To exclude non-specific effects on barley defense responses due to miRNA overexpression, for example basal immunity, we delivered EV alone or together with MIR9863b into leaves of Golden Promise (GP) that is susceptible to both Bgh isolates K1 and A6 (Fig. 8B). HI% scored on GP leaves expressing EV control, or co-expressing EV with MIR9863b, was ∼50% upon inoculation with Bgh K1 or A6, suggesting that expression of miR9863b.1/b.2 does not interfere with basal defense to Bgh (Fig. 8B, bars 1 and 2, bars 5 and 6). Furthermore, functional Mla1-mYFP or Mla10-mYFP fusion [52] was co-delivered into GP leaves with either EV or MIR9863b, respectively (Fig. 8B, bars 3 and 4, 7 and 8). Mla1-mYFP co-expression with EV or MIR9863b resulted in contrasting HI% for Bgh K1 (AVRa1), ∼24% vs. ∼45% (Fig. 8B, bars 3 and 4), indicating again that MLA1-mediated responses against Bgh K1 was compromised by miR9863b.1/b.2 expression; in contrast, Mla10-mYFP co-expression with EV or MIR9863b resulted in similar HI% of ∼25% to Bgh A6 (AVRa10), thus retained MLA10-triggered disease resistance to Bgh A6 (Fig. 8B, bars 7 and 8), consistent with the finding that Mla10 is not a miR9863 target (Fig. 3, Fig. 4).
MLA triggered cell-death is conserved in dicot plants [56], [57], thus, we reasoned that miR9863 targeting MLA may also affect this MLA activity in dicots. To test this possibility, we co-expressed MIR9863a or MIR9863c respectively with Mla1 (Fig. 8C), or with Mla10 (Fig. 8D), in N. benthamiana. Mla1 co-expression with EV effectively triggered cell death detected by trypan blue staining at 36∼48 hpai, whereas this cell-death was completely abolished in Mla1 co-expression with MIR9863a (Fig. 8C, left panel), but not with MIR9863c (Fig. 8C, right panel). Alternatively, Mla10 co-expression with EV triggered strong cell-death that was unaffected by expressing either MIR9863a or MIR9863c (Fig. 8D, left and right). These results indicate that miR9863a retains regulation specificity in suppressing MLA1, but not MLA10, to induce cell-death in dicot plants.
Expression of Mla1, miR9863 members and phasiRNAs during Bgh infection
To further understand the regulation of miR9863 on Mla target in the context of Bgh infections, we analyzed the expression of Mla1, miR9863 members and two phasiRNAs during incompatible barley-Bgh interactions (S10 Figure). A barley transgenic line expressing functional Mla1-HA fusion driven by Mla1 native promoter [61] was inoculated with Bgh K1 (AVRa1) and gene expression was monitored up to 72 hrs post inoculation (hpi). Mla1 transcripts were induced at 16 hpi by Bgh K1 and reached the highest level at 24 hpi (S10A Figure). Similar results were previously reported for the expression of Mla6 and Mla13 during incompatible interactions [76], [86]. During the same period of time from 16 to 24 hpi, we also detected by stem-loop qRT-PCR the increased expression of miR9863a/b.2 and miR9863c/b.1, as well as phasiRNAI and phasiRNAII (S10B and C Figure, Fig. 6A), noting that here we cannot distinguish miR9863a from miR9863b.2, or miR9863c from miR9863b.1, due to only one nt difference. Accumulation of Mla1 transcripts dropped from ∼4.0 fold at 24 hpi to ∼1.6 fold at 48 to 72 hpi (S10A Figure), and inversely, the expression level of miR9863c/b.1, and the two phasiRNAs was increased and reached to the highest level at 48 hpi and sustained at high level up to 72 hpi (S10 B and C Figure). Interestingly, miR9863a/b.2 expression was induced earlier (at 16 hpi) than miR9863c/b.1 (at 24 hpi), and the former maintained at similar level while the latter continued to increase up to 72 hpi (S10B Figure). These data confirm the induction of Mla gene expression in the early phase (16∼24 hpi) during incompatible interactions [76], [86] and suggest that, in the later phase of the interactions, the decreased Mla1 expression level is coupled to the induced or sustained expression of miR9863s and the phasiRNAs.
Discussion
We present here a regulatory network comprising miR9863 family members and the derived secondary phasiRNAs that target Mla alleles during barley-Bgh interactions. This post-transcriptional mechanism appears to utilize more than one mode of action in miRNA-mediated target regulation, i.e., Mla mRNA cleavage, shown in both barley and N. benthamiana (Fig. 1 and S4 Figure), and translational inhibition, shown in N. benthamiana (Fig. 7). Overexpression of miR9863 members attenuate group I MLA-mediated disease resistance to Bgh in barley and its heterologous cell-death response in N. benthamiana (Fig. 8). This dedicated miR9863 function is likely related to the specificity defined by the miR9863-Mla pair and dictated by SNP variations in mature miR9863s and the miRNA-targeting site in unique Mla alleles (Figs. 1, 3 and 4; S6 and S7 Figures). Furthermore, BSMV-mediated silencing of miR9863 in barley led to the up-regulation of Mla1, but not Mla6 transcripts (Fig. 5), and time-course expression analysis revealed that the down-regulation of Mla1 transcript accumulation is inversely correlated with the up-regulation of miR9863s and phasiRNAs in the late phase of barley-Bgh incompatible interactions (S10 Figure). Interestingly, for many Mla alleles, there also exists a correlation between their derepression of miR9863 regulation with their dependence on RAR1, a cochaperone required for accumulation of a subset NLR receptors (S11 Figure) (see below). Therefore, we propose that the miR9863 family plays a key role in targeting Mla alleles and in dampening the disease resistance and associated cell-death triggered by these NLR proteins.
miR9863 family target NLR genes in Triticeae species
Although miRNAs regulating NLR-encoding genes have recently been demonstrated in a wide variety of dicot species and perennial woody plants [38]–[41], [87], convincing evidence for similar regulatory mechanisms have been scarce in well characterized monocot species, for example, rice and maize. Based on earlier work, it was postulated that NLR genes in Poaceae species were not regulated by endogenous miRNA and siRNA regulatory networks [43]. However, regulation of barley Mla alleles by the miR9863 family demonstrates that these mechanisms are present in Poaceae species. Moreover, previous small RNA sequencing studies have identified various members of the miR9863 family [63]–[66], [88], suggesting functional conservation in barley and wheat. Through data mining and mRNA expression analyses, we show that the miR9863 family appears to be absent in dicot species N. benthamiana and A. thaliana, as well as monocots rice (O. sativa), maize (Z. mays) and B. distachyon, at least at the depth of coverage in current miRNA databases (S2 and S3 Figures). Although some putative sorghum small RNAs were found sequentially matching miR9863a, it appears that the miR9863 family is mainly expressed in barley and wheat, suggesting that miR9863 family might be Triticeae-specific. Since functional Mla homologs and orthologs exist in wheat, it will be interesting to know whether miR9863 members regulate these genes or other NLR genes as well (S1 Figure) [89], [90].
SNPs in miRNA and target miRNA-binding sites determine regulation specificity
Unlike animal miRNAs, plant miRNAs largely depend on their high sequence complementarity to target mRNAs for post-transcriptional gene regulation [9]. Indeed, Arabidopsis and rice genome-wide assessments of sequence variation have revealed lower levels of nucleotide variation and divergence in miRNAs and target binding sites than in their flanking sequences [73], [91], leading to hypothesis that strong purifying selection plays an important role in the interaction between plant miRNAs and their target binding sites. Somewhat surprisingly, similar findings were also reported in humans [92], [93]. Nevertheless, few SNP variations in miRNAs and their target binding sites have been identified in plants [73], [91], [94], [95], and some of them have been shown to contribute to phenotypic variations in Arabidopsis [95].
Our analyses of miR9863s and Mla target sequences have uncovered the role of miR9863 family SNPs in determining target specificity and potentially phenotypic variations (Figs. 1, 2 and 3; S6 Figure). The miR9863a and miR9863b.2 differ by only one SNP at the 9th nt position (Fig. 1A), however, they account for ∼60–80% difference in Mla1 target protein accumulation upon coexpression in N. benthamiana (Fig. 2A, 2E; S6 Figure). Similarly, one SNP variation at 2nd nt position in miR9863c and miR9863b.1 can fully dictate the regulation specificity on Mla1 (Fig. 1A; Fig. 2B, 2C and 2E). In this context, it is worth noting that the NLR-targeting miR482/2118 superfamily contains at least 9 SNPs and 31 isoforms across diverse plant species [38], [40]. Since many isoforms of the miR482/2118 family are species or genera specific, it is possible that the SNP variations within members of this superfamily have evolved to allow specific isoforms to regulate species-specific NLR genes and other targets as well [40].
A single or two adjacent SNPs in Mla alleles can dictate the regulation specificity of the miRNA members on Mla, thus, group I Mla but not group II or III Mla are targets of miR9863 members (Figs. 3 and 4; S7 Figure). The above findings are supported not only by miRNA expression in the heterologous N. benthamiana system, but also by miRNA silencing in barley using BMSV-VIGS coupled with STTM technology (Figs. 4 and 5). Moreover, single cell transient expression assays in barley have demonstrated that overexpression of MIR9863a or MIR9863b specifically attenuated Mla1 - but not Mla10-mediated disease resistance (Fig. 8A); similarly, miR9863a expression in N. benthamiana blocked Mla1 - rather than Mla10-triggered cell death (Fig. 8C and 8D). Therefore, miR9863 variants paired with their respective Mla alleles supports the notion that SNP variations in the miRNA and the target binding site are important in determining regulation specificity as well as in phenotypic variations in plants. We believe that this is particularly crucial for the control of the large NLR gene family, to allow for balancing the fitness cost and effectively coping with the fast evolving pathogen isolates.
Evolution of Mla alleles with respect to miR9863 regulation
Although the Mla locus encodes highly related alleles apart from other dissimilar RGH families [48], [77], [96], only a subset of these alleles are regulated by three out of four miR9863 family members presented here. We postulate that specificity between Mla and miR9863 is determined by SNP variation(s) in members among miR9863 family as well as in the binding site of target Mla alleles (see above). Nonetheless, the evolutionary and functional relevance of Mla-miRNA regulation remains unclear. To shade light on this aspect, we utilized the cDNA sequences encoding full-length MLA, CC, NB-ARC or LRR domain to respectively construct a phylogeny tree of the 29 Mla alleles (S11 Figure). When each individual Mla is mapped in accordance with subgroup identity based on SNPs in the target site, i.e., group I, II and III (see Fig. 3A), we observed more similar architecture for the phylogeny tree derived from full-length or NB-ARC cDNA sequence than to the others (S11 Figure, A and C). Most interestingly, in the NB-ARC phylogeny tree, Mla alleles of group II and III comprise one more recent and distinct branch whereas group I Mla constitute a more complex pattern, indicating group II and III Mla are evolutionarily closer to each other than to group I Mla with regard to regulation by miR9863. Indeed, we show that this control only applies to group I Mla alleles. This is reminiscent of the proposed parallel evolutionary pathways for the Mla family, i.e., one branch contains Mla1 (from group I) and the other branch contains Mla6 and Mla13 (from group II and III, respectively) [59]. Thus, we speculate that the evolution of the Mla family might be influenced by the adaptation of miR9863. In light of this, natural SNP variations might be fixed within the miR9863 binding site of group II and III Mla, which result in the deregulation of the miRNAs and subsequent formation of a Mla clade distinctive from group I Mla members.
Additional evidence to support evolutionary pathways differentiating group I Mla alleles from group II and III comes from the differential Rar1-dependency of Mla alleles. RAR1 is believed to act as a cochaperone and assist NB-LRR proteins of low accumulation level to reach threshold steady-state level for effective immunity [60], [61], [97], [98]. For example, MLA6 accumulates only one fourth the level of MLA1, and thus depends on RAR1 to maintain a threshold level to mediate disease resistance [61]. Interestingly enough, while Mla1 from group I is Rar1-independent, Mla alleles from group II or III, such as Mla6, Mla9, Mla12, Mla13, Mla22 and Mla23, excepting Mla7, are all Rar1-dependent [59], [99] (Fig. 3; S10 Figure). One would hypothesize that, in barley and barley powdery mildew interactions, Mla-miR9863 adaptations are likely fixed for Mla alleles whose expression level and protein accumulation are high, for example those of group I; whereas for Mla natural variants, like members of group II and III that are unstable or with lower accumulation level, the miR9863 regulation might be unnecessary and thus released. Nevertheless, how the evolution of Mla branches is coupled with miR9863 regulation as well as the cochaperone RAR1 requires future investigation.
Implications from using the N. benthamiana expression system in dissecting miR9863 regulation of Mla alleles
In the present study, we leveraged the heterologous N. benthamiana expression system to investigate the regulation of barley miR9863 on its barley NLR targets. This system allows the testing of individual components in the absence of unknown confounding factors in the barley host. Indeed, despite the apparent absence of the miR9863 family in the dicotyledonous N. benthamiana and Arabidopsis (S3 Figure, and discussion above), transient expression of barley and wheat miR9863 progenitors or artificial miRNA precursors in N. benthamiana can generate respective mature miRNAs of the correct size (Fig. 2D; S2B Figure), suggesting that a conserved small RNA biogenesis machinery exists in both monocots and dicots. This is further corroborated by the observation of direct coexpression of 22-nt miR9863 members and Mla1 in N. benthamiana which enhanced the production of 21-nt phasiRNAs, and that many of these miR9863 members were identified in sRNA libraries derived from Mla1-containing barley isogenic line (Fig. 6).
VIGS of NbAGO1 in N. benthamiana resulted in the loss of miR9863a or miR9863b control of Mla1. This suggests that AGO1 may be functionally conserved in N. benthamiana and in barley (S9 Figure). This is consistent with previous observation that plant AGO1 has the capacity to bind 22-nt miRNAs and trigger the production of secondary siRNAs [25]. It would be interesting in future experiments to test whether in barley 22-nt miR9863 members are sorted into AGO1-containing complex where they direct Mla transcript cleavage.
Lastly, previous studies found that barley MLA1-triggered immunity against Bgh fungal isolate is fully retained in Arabidopsis and that MLA-triggered cell-death signaling is likely conserved in Arabidopsis and N. benthamiana [56]–[58]. Similarly, recent studies show that expression of some wheat Pm alleles, conferring resistance against B. graminis pathogens, also triggers cell-death responses in N. benthamiana [100], [101]. Interestingly, another study also demonstrates that interfamily transfer of the dual TIR-type NLR genes, RPS4/RRS1, from Arabidopsis into other Brassicaceae plants and Solanaceae species, confers broad-spectrum or isolate-specific disease resistance to fungal or bacterial pathogens [102]. These examples suggest that some plant NLR receptors may engage evolutionarily conserved downstream signaling components for triggering immune responses. Here, coexpression of miR9863a with Mla1 or Mla10 in N. benthamiana attenuates Mla1 - but not Mla10-triggered cell-death responses (Fig. 8), together with above-mentioned retained miR9863 specificity on Mla alleles in N. benthamiana, might imply a conserved post-transcriptional regulatory machinery acting on heterologously expressed barley Mla NLR genes.
Materials and Methods
Plant and fungal materials
Barley (Hordeum vulgare L.), bread wheat (Triticum aestivum L.) and Brachypodium distachyon plants were grown in a growth chamber under a 16 hrs/8 hrs, 20°C/18°C day/night cycle, respectively, with 70% relative humidity; Rice (O. sativa) was maintained in a growth chamber under a 16 hrs/8 hrs, 28°C/26°C day/night cycle, respectively, with 90% relative humidity; Maize (Z. mays) was grown in a chamber at 22°C under a 16 hrs/8 hrs day/night cycle under 70% relative humidity; N. benthamiana and A. thaliana were grown in greenhouse at 24±1°C with a 16 hr light period.
Blumeria graminis f. sp. hordei (Bgh) isolates K1 (AVRa1, vira6, vira10, vira12) and A6 (AVRa6, AVRa10, AVRa12, vira1) were maintained on barley cultivar ‘I10’ (containing Mla12) and ‘P01’ (containing Mla1), respectively, and kept at 70% relative humidity, and a 16 hrs/8 hrs, 20°C/18°C day/night cycle.
Small RNA isolation and deep sequencing
Seven-day-old barley leaves of barley isogenic line P01 were inoculated with Bgh A6 and K1 for 22 hrs and total RNAs were isolated using TRIzol solution (Invitrogen 15596-026) according to the manufacturer's instructions. Small RNAs of 18–30 nt were excised and isolated from 5 to 10 µg total RNAs electrophoresed on 15% polyacrylamide denaturing gel, and then were ligated with 5′ and 3′ adapters. The ligated small RNAs were used as templates for cDNA synthesis followed by PCR amplification. The obtained libraries were sequenced using the Solexa sequencing platform (BGI, Beijing).
MIRNA precursor identification, secondary structure prediction, and cloning
The mature miRNAs sequences were used in a BLASTn search against the barley genome sequencing database (http://webblast.ipk-gatersleben.de/barley/) and bread wheat expressed sequence tag (EST) database (http://www.ncbi.nlm.nih.gov/). The secondary structure of flanking sequence around perfectly matched site was predicted using the RNA-folding program Mfold [103].
Primers for cloning of MIR9863 precursors were designed according to the flanking sequences of the hairpin structures. Primers (J19/J20) for hvu-MIR9863a cloning were designed according to the sequence of tae-MIR9863a. hvu-MIR9863b was PCR-amplified using barley ‘Morex’ genomic DNA as template (primers, J15/J16); tae-MIR9863 cluster containing tae-MIR9863a and tae-MIR9863b was amplified using bread wheat ‘Chancellor’ genomic DNA as template and primer J05/J06; tae-MIR9863c was amplified using bread wheat ‘Chancellor’ genomic DNA as template and primer J07/J08. All primer sequences are shown in supplemental S2 Table.
Validation of miRNA cleavage site by 5′ RACE assay
RLM-RACE kit (TaKaRa, Code D315) was used for 5′ RACE according to the manufacturer's instruction. Total RNAs were isolated from 7-day-old leaves of barley line P01 infected with Bgh K1 spores, and mRNAs were enriched from 100 µg of total RNAs using the PolyATtract mRNA isolation kit (Promega, Z5210). The RNA Oligo adaptor was ligated to mRNAs without calf intestinal phosphatase treatment. For the first round PCR, the 5′ RACE Outer Primer J01 together with Mla1 gene specific outer primer J03 were used. Nested PCR amplification was performed using the 5′ RACE Inner Primer J02 and Mla1 specific inner primer J04.
Plasmid constructions
Plasmids constructed in this study are based on several expression vectors previously described and listed in supplemental S3 Table.
CTAPi-GW-3HA was used as the transient expression vector for Mla genes in N. benthamiana, and derived plasmids were constructed using Gateway technology (Invitrogen) following the instructions of the manufacturer.
For construction of miRNAs overexpression constructs used in N. benthamiana expression assays, indicated MIRNAs precursor sequences were PCR amplified, double digested by KpnI/HindIII, and ligated into 35S-pKANNIBAL vector. Precursor aMIR9863a, aMIR9863b.1 and aMIR9863b.2 were constructed by replacing the miR173/miR173* duplex in ath-MIR173 with amiR9863a/amiR9863a*, amiR9863b.1/amiR9863b.1* or amiR9863b.2/amiR9863b.2* (S5 Figure) using overlapping primers J35/J36/J37, J31/J32/J33 and J36/J37/J38, respectively.
pTRV2-derivatives used in TRV-VIGS analysis were constructed based on pTRV2-LIC by ligation-independent cloning (LIC) as described previously [104]. J44/J45, J46/J47, J48/J49 and J50/J51 were used for amplifying anti-sense fragment from cDNA of N. benthamiana for silencing NbAGO1-1, NbAGO1-2, NbAGO4-1 and NbAGO4-2, respectively. pTRV2-LIC vector was digested with PstI and ligated with PCR fragments.
For BSMV mediated silencing of miR9863 members in barley, STTM-miR9863 and STTM-EV were first designed and amplified by using overlapping primer groups J52/J53/J54 and J55/J56, according to Fig. 5A and Yan and associates [80]. Then PCR products were cloned into pCaBS-γbLIC vector through LIC strategy as described above.
For single-cell transient overexpression of miR9863 members, the tae-MIR9863a, tae-MIR9863c or hvu-MIR9863b precursors with attB sites were cloned into pUbi-GATE vector by Gateway technology described above. Similarly, pUbi-GW-mYFP vector was used for expression of Mla1 or Mla10 cDNA.
One-step site-directed mutagenesis
Point mutations for different Mla cDNAs or MIRNA precursors were introduced using one-step site-directed mutagenesis as described previously with minor modifications [105]. The entry vectors harboring wild-type Mla allele cDNA or 35S-pKANNIBAL vectors containing indicated wild-type MIRNA precursor sequences were used as templates for PCR reaction to introduce mutations.
Real-time or semi-quantitative RT-PCR analysis
Total RNAs were extracted from plant materials using TRIzol solution, and treated with RNase-free DNase I (TaKaRa). About 2 µg of total RNA and M-MLV Reverse Transcriptase (Promega) were further used for reverse transcription. For coding genes reverse transcription, first-strand cDNA was synthesized using Oligo (dT)18. For small RNA reverse transcription, specifically designed stem-loop reverse transcription primers were used, and followed the procedures described by Chen and colleagues [106]. Primer J71, J72, J75 and J76 were used for miR9863a/b.2, miR9863c/b.1, phasiRNAI and phasiRNAII, respectively; primer J81 was used for U6 (U6 stands for U6 spliceosomal RNA), and obtained cDNA was diluted 10 times and used for further analysis. Real-time qPCR was performed using StepOne real-time system (Applied Biosystems) and GoTaq qPCR Master Mix (Promega, A6001); for the determination of three Mla1 amplicons (Fig. 7), primer pairs J65/J66, J67/J68 and J69/J70 were used; Primer J73, J74, J77 and J78 were respectively used with J79 to quantify the level of miR9863a/b.2, miR9863c/b.1, phasiRNAI and phasiRNAII; Primer pair J80/J81 and J82/J83 were used for the detection of U6 and Actin. Semi-quantitative RT-PCR was performed as described previously [56]. Primer pair J61/J62, J63/J64 were designed to determine the silencing efficiency of NbAGO1 and NbAGO4, respectively.
RNA gel-blot analysis
RNA gel blot analysis was performed as described previously with minor modifications [107]. Total RNAs of 10 to 15 µg were separated on a 15% polyacrylamide denaturing gel by electrophoresis and cross-linking was performed as described previously [108]. The complementary sequences corresponding to miRNAs were used as probes after labeled with [γ-32P]ATP using T4 polynucleotide kinase (New England Biolabs). Probe J84, J85, J86 and J88 were used to detect the mature miR9863a, miR9863b.1, miR9863b.2 and miR156a signal, respectively; probe J87 was used for Mla1 derived phasiRNAs in S8 Figure according to Chen and colleagues [44].
Agrobacterium-mediated transient gene expression and protein analysis
Agrobacterium-mediated transient expression in N. benthamiana was performed as described previously [56], [109]. For single expression, Agrobacterium suspensions expressing gene of interest were infiltrated into 4 - to 5-week old N. benthamiana leaves. For co-expressions, Agrobacterium suspensions expressing MIRNA precursor were first infiltrated, and 24 hrs later, Agrobacterium suspensions expressing gene candidate was infiltrated into the same position. Samples were collected from the infiltrated sites at 36 hrs post infiltration. Total soluble proteins were extracted using 180 µl of 2× Laemmli buffer [110] from 60 mg leaf samples, and detected by immunoblotting as described previously [56]. Rat anti-HA antibody (1∶5000; Roche, 11867423001) and anti-rat IgG conjugated with horseradish peroxidase (HRP) (1∶10000; Sigma, A5795) were used for HA-tagged proteins detection. The levels of β-actin were determined using an anti-β-actin antibody (1∶1000; CWBIO, CW0264) coupled with anti-mouse IgG conjugated with HRP (1∶75000; Sigma, A9044).
Single-cell transient gene expression assay
Single-cell transient gene expression assay using biolistic delivery of plasmid DNA into barley epidermal cells was performed as previously described [77]. For MIRNAs overexpression, the β-glucuronidase (GUS) reporter gene was mixed with respective plasmids (molar ratio 1∶1) before coating of gold particles. Barley leaf epidermal cells were transformed with the biolistic particle delivery system (Bio-Rad, Model PDS-1000/He), and incubated 4 hrs before inoculation of Bgh spores. To identify transformed cells, bombarded leaves were fixed in solution at 48 hrs after Bgh infection and further stained for GUS activity. For MIRNAs and Mla co-expression, the GUS reporter and MIR9863b plasmids were co-coated with Mla1-mYFP or Mla10-mYFP plasmids (molar ratio 1∶1∶1).
Trypan blue staining
Trypan blue staining was described previously [56]. Briefly, N. benthamiana leaves were boiled for 5 min in staining solution, and were then de-stained in 2.5 g ml-1 chloral hydrate in distilled water for at least 3 days.
TRV and BSMV virus-induced gene silencing assay
TRV-based virus induced gene silencing assay was performed as described [56], [111]. Briefly, pTRV1 and pTRV2-derived constructs (pTRV2: EV, pTRV2: NbPDSas, pTRV2: NbAGO1-1as, pTRV2: NbAGO1-2as, pTRV2: NbAGO4-1as, pTRV2: NbAGO4-2as) were transformed into A. tumefaciens strain GV3101. Cell suspensions containing pTRV1 and pTRV2-derived constructs were collected and mixed at 1∶1 ratio and infiltrated into the third to fifth leaf of three-week-old N. benthamiana plants. Three weeks after infiltration, the upper newly expanded leaves were selected for further analysis.
For BSMV-mediated STTM-VIGS assay of miR9863 members, constructs of pCaBS-α, pCaBS-β, and pCaBS-γbLIC derivatives (pCaBS-γSTTM-EV and pCaBS-γSTTM-miR9863) were transformed into the A. tumefaciens strain EHA105, respectively. The Agrobacterium suspensions of OD600 = 0.8 were mixed at 1∶1∶1 ratio and infiltrated in N. benthamiana leaves. The N. benthamiana sap was extracted from leaves with BSMV symptom at about 12 days post infiltration, and inoculated to the first two emerging leaves of barley leaves. About 15 days later, the newly grown upper barley leaves with virus symptom were collected for further analysis.
Accession numbers
Sequence data for genes used in this article can be found under GenBank accession numbers AY009939 (HvuMla1), GU245938 (HvuMla2), GU245939 (HvuMla3), AJ302292 (HvuMla6), AY266444 (HvuMla7), GU245940 (HvuMla8), GU245941 (HvuMla9), AY266445 (HvuMla10), AY196347 (HvuMla12), AF523683 (HvuMla13-1), GU245942 (HvuMla16-1), GU245943 (HvuMla18-1), GU245944 (HvuMla18-2), GU245945 (HvuMla19-1), GU245946 (HvuMla22), GU245947 (HvuMla23), GU245948 (HvuMla25-1), GU245949 (HvuMla27-1), GU245950 (HvuMla27-2), GU245951 (HvuMla28), GU245952 (HvuMla30), GU245953 (HvuMla31-1), GU245954 (HvuMla32), GU245955 (HvuMla34), GU245956 (HvuMla35), GU245957 (HvuMla36-1), GU245958 (HvuMla37-1), GU245959 (HvuMla38-1), GU245960 (HvuMla39-1), KJ619975 (hvu-MIR9863a), AK364228 (hvu-MIR9863b), CK193889 (tae-MIR9863a, tae-MIR9863b, tae-MIR9863a/b cluster), DR736484 (tae-MIR9863c), DQ321488 (NbAGO1-1), DQ321489 (NbAGO1-2), DQ321490 (NbAGO4-1), DQ321491 (NbAGO4-2), ACZ65507 (HchMla1), ADX06722 (TmoMla1), AGP75918 (TmoSr35), KF031291 (AetSr33), ACG63536 (TduLr10), AAG42168 (TaeYr10), AAY21626 (TaePm3a), AAQ96158 (TaePm3b), ABB78077 (TaePm3c) and AAY21627 (TaePm3d).
Supporting Information
Zdroje
1. JonesJD, DanglJL (2006) The plant immune system. Nature 444 : 323–329.
2. ZipfelC (2009) Early molecular events in PAMP-triggered immunity. Curr Opin Plant Biol 12 : 414–420.
3. DoddsPN, RathjenJP (2010) Plant immunity: towards an integrated view of plant–pathogen interactions. Nat Rev Genet 11 : 539–548.
4. MaekawaT, KuferTA, Schulze-LefertP (2011) NLR functions in plant and animal immune systems: so far and yet so close. Nat Immunol 12 : 817–826.
5. TsudaK, SatoM, StoddardT, GlazebrookJ, KatagiriF (2009) Network properties of robust immunity in plants. PLoS Genet 5: e1000772.
6. MeyersBC, KaushikS, NandetyRS (2005) Evolving disease resistance genes. Curr Opin Plant Biol 8 : 129–134.
7. TianD, TrawMB, ChenJQ, KreitmanM, BergelsonJ (2003) Fitness costs of R-gene-mediated resistance in Arabidopsis thaliana. Nature 423 : 74–77.
8. BombliesK, LempeJ, EppleP, WarthmannN, LanzC, et al. (2007) Autoimmune response as a mechanism for a dobzhansky-muller-type incompatibility syndrome in plants. PLoS Biol 5: e236.
9. Jones-RhoadesMW, BartelDP, BartelB (2006) MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol 57 : 19–53.
10. MalloryA, VaucheretH (2006) Functions of microRNAs and related small RNAs in plants. Nat Genet 38: S31–S36.
11. DingSW (2010) RNA-based antiviral immunity. Nat Rev Immunol 10 : 632–644.
12. Katiyar-AgarwalS, JinH (2010) Role of small RNAs in host-microbe interactions. Annu Rev Phytopathol 48 : 225–246.
13. PumplinN, VoinnetO (2013) RNA silencing suppression by plant pathogens: defence, counter-defence and counter-counter-defence. Nat Rev Micro 11 : 745–760.
14. NavarroL, DunoyerP, JayF, ArnoldB, DharmasiriN, et al. (2006) A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 312 : 436–439.
15. NavarroL, JayF, NomuraK, HeSY, VoinnetO (2008) Suppression of the microRNA pathway by bacterial effector proteins. Science 321 : 964–967.
16. ZhangX, ZhaoH, GaoS, WangW-C, Katiyar-AgarwalS, et al. (2011) Arabidopsis argonaute 2 regulates innate immunity via miRNA393*-mediated silencing of a golgi-localized SNARE gene, MEMB12. Mol Cell 42 : 356–366.
17. XuW, MengY, WiseRP (2014) Mla - and Rom1-mediated control of microRNA398 and chloroplast copper/zinc superoxide dismutase regulates cell death in response to the barley powdery mildew fungus. New Phytol 201 : 1396–1412.
18. Katiyar-AgarwalS, GaoS, Vivian-SmithA, JinH (2007) A novel class of bacteria-induced small RNAs in Arabidopsis. Genes Dev 21 : 3123–3134.
19. Katiyar-AgarwalS, MorganR, DahlbeckD, BorsaniO, VillegasAJr, et al. (2006) A pathogen-inducible endogenous siRNA in plant immunity. Proc Natl Acad Sci U S A 103 : 18002–18007.
20. EllendorffU, FradinEF, de JongeR, ThommaBPHJ (2009) RNA silencing is required for Arabidopsis defence against Verticillium wilt disease. J Exp Bot 60 : 591–602.
21. QiaoY, LiuL, XiongQ, FloresC, WongJ, et al. (2013) Oomycete pathogens encode RNA silencing suppressors. Nat Genet 45 : 330–333.
22. WeibergA, WangM, LinF-M, ZhaoH, ZhangZ, et al. (2013) Fungal small RNAs suppress plant immunity by hijacking host RNA interference pathways. Science 342 : 118–123.
23. BaulcombeD (2004) RNA silencing in plants. Nature 431 : 356–363.
24. RogersK, ChenX (2013) Biogenesis, turnover, and mode of action of plant microRNAs. Plant Cell 25 : 2383–2399.
25. MalloryA, VaucheretH (2010) Form, function, and regulation of ARGONAUTE proteins. Plant Cell 22 : 3879–3889.
26. VoinnetO (2009) Origin, biogenesis, and activity of plant microRNAs. Cell 136 : 669–687.
27. TangG, ReinhartBJ, BartelDP, ZamorePD (2003) A biochemical framework for RNA silencing in plants. Genes Dev 17 : 49–63.
28. BaumbergerN, BaulcombeDC (2005) Arabidopsis ARGONAUTE1 is an RNA slicer that selectively recruits microRNAs and short interfering RNAs. Proc Natl Acad Sci U S A 102 : 11928–11933.
29. LlaveC, XieZ, KasschauKD, CarringtonJC (2002) Cleavage of scarecrow-like mRNA targets directed by a class of Arabidopsis miRNA. Science 297 : 2053–2056.
30. AukermanM, SakaiH (2003) Regulation of flowering time and floral organ identity by a microRNA and its APETALA2-like target genes. Plant Cell 15 : 2730–2741.
31. ChenX (2004) A microRNA as a translational repressor of APETALA2 in Arabidopsis flower development. Science 303 : 2022–2025.
32. BrodersenP, Sakvarelidze-AchardL, Bruun-RasmussenM, DunoyerP, YamamotoYY, et al. (2008) Widespread translational inhibition by plant miRNAs and siRNAs. Science 320 : 1185–1190.
33. DugasD, BartelB (2008) Sucrose induction of Arabidopsis miR398 represses two Cu/Zn superoxide dismutases. Plant Mol Biol 67 : 403–417.
34. LanetE, DelannoyE, SormaniR, FlorisM, BrodersenP, et al. (2009) Biochemical evidence for translational repression by Arabidopsis microRNAs. Plant Cell 21 : 1762–1768.
35. BeauclairL, YuA, BouchéN (2010) microRNA-directed cleavage and translational repression of the copper chaperone for superoxide dismutase mRNA in Arabidopsis. Plant J 62 : 454–462.
36. GandikotaM, BirkenbihlR, HohmannS, CardonG, SaedlerH, et al. (2007) The miRNA156/157 recognition element in the 3' UTR of the Arabidopsis SBP box gene SPL3 prevents early flowering by translational inhibition in seedlings. Plant J 49 : 683–693.
37. LiS, LiuL, ZhuangX, YuY, LiuX, et al. (2013) MicroRNAs inhibit the translation of target mRNAs on the endoplasmic reticulum in Arabidopsis. Cell 153 : 562–574.
38. ZhaiJ, JeongDH, De PaoliE, ParkS, RosenBD, et al. (2011) MicroRNAs as master regulators of the plant NB-LRR defense gene family via the production of phased, trans-acting siRNAs. Genes Dev 25 : 2540–2553.
39. LiF, PignattaD, BendixC, BrunkardJO, CohnMM, et al. (2012) MicroRNA regulation of plant innate immune receptors. Proc Natl Acad Sci U S A 109 : 1790–1795.
40. ShivaprasadPV, ChenHM, PatelK, BondDM, SantosBA, et al. (2012) A microRNA superfamily regulates nucleotide binding site-leucine-rich repeats and other mRNAs. Plant Cell 24 : 859–874.
41. BoccaraM, SarazinA, ThiebeauldO, JayF, VoinnetO, et al. (2014) The Arabidopsis miR472-RDR6 silencing pathway modulates PAMP - and effector-triggered immunity through the post-transcriptional control of disease resistance genes. PLoS Pathog 10: e1003883.
42. KällmanT, ChenJ, GyllenstrandN, LagercrantzU (2013) A significant fraction of 21-nucleotide small RNA originates from phased degradation of resistance genes in several perennial species. Plant Physiol 162 : 741–754.
43. FeiQ, XiaR, MeyersBC (2013) Phased, secondary, small interfering RNAs in posttranscriptional regulatory networks. Plant Cell 25 : 2400–2415.
44. ChenHM, ChenLT, PatelK, LiYH, BaulcombeDC, et al. (2010) 22-Nucleotide RNAs trigger secondary siRNA biogenesis in plants. Proc Natl Acad Sci U S A 107 : 15269–15274.
45. CuperusJT, CarbonellA, FahlgrenN, Garcia-RuizH, BurkeRT, et al. (2010) Unique functionality of 22-nt miRNAs in triggering RDR6-dependent siRNA biogenesis from target transcripts in Arabidopsis. Nat Struct Mol Biol 17 : 997–1003.
46. ZhaiJ, ZhaoY, SimonSA, HuangS, PetschK, et al. (2013) Plant microRNAs display differential 3′ truncation and tailing modifications that are ARGONAUTE1 dependent and conserved across species. Plant Cell 25 : 2417–2428.
47. ManavellaPA, KoenigD, WeigelD (2012) Plant secondary siRNA production determined by microRNA-duplex structure. Proc Natl Acad Sci U S A 109 : 2461–2466.
48. SeeholzerS, TsuchimatsuT, JordanT, BieriS, PajonkS, et al. (2010) Diversity at the Mla powdery mildew resistance locus from cultivated barley reveals sites of positive selection. Mol Plant Microbe Interact 23 : 497–509.
49. ShenQH, Schulze-LefertP (2007) Rumble in the nuclear jungle: compartmentalization, trafficking, and nuclear action of plant immune receptors. EMBO J 26 : 4293–4301.
50. ChangC, ZhangL, ShenQH (2013) Partitioning, repressing and derepressing: dynamic regulations in MLA immune receptor triggered defense signaling. Front Plant Sci 4 : 396.
51. CaldoRA, NettletonD, WiseRP (2004) Interaction-dependent gene expression in Mla-specified response to barley powdery mildew. Plant Cell 16 : 2514–2528.
52. ShenQH, SaijoY, MauchS, BiskupC, BieriS, et al. (2007) Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science 315 : 1098–1103.
53. MengY, WiseRP (2012) HvWRKY10, HvWRKY19, and HvWRKY28 regulate Mla-triggered immunity and basal defense to barley powdery mildew. Mol Plant Microbe Interact 25 : 1492–1505.
54. ChangC, YuD, JiaoJ, JingS, Schulze-LefertP, et al. (2013) Barley MLA immune receptors directly interfere with antagonistically acting transcription factors to initiate disease resistance signaling. Plant Cell 25 : 1158–1173.
55. MoscouMJ, LauterN, CaldoRA, NettletonD, WiseRP (2011) Quantitative and temporal definition of the Mla transcriptional regulon during barley–powdery mildew interactions. Mol Plant Microbe Interact 24 : 694–705.
56. BaiS, LiuJ, ChangC, ZhangL, MaekawaT, et al. (2012) Structure-function analysis of barley NLR immune receptor MLA10 reveals its cell compartment specific activity in cell death and disease resistance. PLoS Pathog 8: e1002752.
57. MaekawaT, KracherB, VernaldiS, Ver Loren van ThemaatE, Schulze-LefertP (2012) Conservation of NLR-triggered immunity across plant lineages. Proc Natl Acad Sci U S A 109 : 20119–20123.
58. MaekawaT, ChengW, SpiridonLN, TollerA, LukasikE, et al. (2011) Coiled-coil domain-dependent homodimerization of intracellular barley immune receptors defines a minimal functional module for triggering cell death. Cell Host Microbe 9 : 187–199.
59. HaltermanDA, WiseRP (2004) A single-amino acid substitution in the sixth leucine-rich repeat of barley MLA6 and MLA13 alleviates dependence on RAR1 for disease resistance signaling. Plant J 38 : 215–226.
60. ShirasuK (2009) The HSP90-SGT1 chaperone complex for NLR immune sensors. Annu Rev Plant Biol 60 : 139–164.
61. BieriS, MauchS, ShenQH, PeartJ, DevotoA, et al. (2004) RAR1 positively controls steady state levels of barley MLA resistance proteins and enables sufficient MLA6 accumulation for effective resistance. Plant Cell 16 : 3480–3495.
62. KølsterP, MunkL, StølenO, LøhdeJ (1986) Near-Isogenic Barley Lines with Genes for Resistance to Powdery Mildew. Crop Sci 26 : 903–907.
63. Mahalingam G, Meyers B (2010) Computational methods for comparative analysis of plant small RNAs. In: Meyers BC, Green PJ, editors. Plant MicroRNAs: Humana Press. pp. 163–181.
64. SchreiberA, ShiB-J, HuangC-Y, LangridgeP, BaumannU (2011) Discovery of barley miRNAs through deep sequencing of short reads. BMC Genomics 12 : 129–149.
65. HackenbergM, ShiBJ, GustafsonP, LangridgeP (2012) A transgenic transcription factor (TaDREB3) in barley affects the expression of microRNAs and other small non-coding RNAs. PLoS One 7: e42030.
66. WeiB, CaiT, ZhangR, LiA, HuoN, et al. (2009) Novel microRNAs uncovered by deep sequencing of small RNA transcriptomes in bread wheat (Triticum aestivum L.) and Brachypodium distachyon (L.) Beauv. Funct Int Genomics 9 : 499–511.
67. KozomaraA, Griffiths-JonesS (2011) miRBase: integrating microRNA annotation and deep-sequencing data. Nucleic Acids Res 39: D152–D157.
68. MeyersBC, AxtellMJ, BartelB, BartelDP, BaulcombeD, et al. (2008) Criteria for annotation of plant microRNAs. Plant Cell 20 : 3186–3190.
69. PatersonAH, BowersJE, BruggmannR, DubchakI, GrimwoodJ, et al. (2009) The Sorghum bicolor genome and the diversification of grasses. Nature 457 : 551–556.
70. BertoliniE, VerelstW, HornerDS, GianfranceschiL, PiccoloV, et al. (2013) Addressing the Role of microRNAs in Reprogramming Leaf Growth during Drought Stress in Brachypodium distachyon. Molecular Plant 6 : 423–443.
71. JeongD-H, SchmidtS, RymarquisL, ParkS, GanssmannM, et al. (2013) Parallel analysis of RNA ends enhances global investigation of microRNAs and target RNAs of Brachypodium distachyon. Genome Biology 14: R145.
72. JeongDH, ThatcherSR, BrownRS, ZhaiJ, ParkS, et al. (2013) Comprehensive investigation of microRNAs enhanced by analysis of sequence variants, expression patterns, ARGONAUTE loading, and target cleavage. Plant Physiol 162 : 1225–1245.
73. EhrenreichIM, PuruggananMD (2008) Sequence variation of MicroRNAs and their binding sites in Arabidopsis. Plant Physiol 146 : 1974–1982.
74. SchwabR, OssowskiS, RiesterM, WarthmannN, WeigelD (2006) Highly specific gene silencing by artificial microRNAs in Arabidopsis. Plant Cell 18 : 1121–1133.
75. HaltermanD, ZhouF, WeiF, WiseRP, Schulze-LefertP (2001) The MLA6 coiled-coil, NBS-LRR protein confers AvrMla6-dependent resistance specificity to Blumeria graminis f. sp. hordei in barley and wheat. Plant J 25 : 335–348.
76. HaltermanDA, WeiF, WiseRP (2003) Powdery mildew-induced Mla mRNAs are alternatively spliced and contain multiple upstream open reading frames. Plant Physiol 131 : 558–567.
77. ShenQH, ZhouF, BieriS, HaizelT, ShirasuK, et al. (2003) Recognition specificity and RAR1/SGT1 dependence in barley Mla disease resistance genes to the powdery mildew fungus. Plant Cell 15 : 732–744.
78. ZhouF, KurthJ, WeiF, ElliottC, ValeG, et al. (2001) Cell-autonomous expression of barley Mla1 confers race-specific resistance to the powdery mildew fungus via a Rar1-independent signaling pathway. Plant Cell 13 : 337–350.
79. YuanC, LiC, YanL, JacksonAO, LiuZ, et al. (2011) A high throughput barley stripe mosaic virus vector for virus induced gene silencing in monocots and dicots. PLoS One 6: e26468.
80. YanJ, GuY, JiaX, KangW, PanS, et al. (2012) Effective small RNA destruction by the expression of a short tandem target mimic in Arabidopsis. Plant Cell 24 : 415–427.
81. MiS, CaiT, HuY, ChenY, HodgesE, et al. (2008) Sorting of small RNAs into Arabidopsis argonaute complexes is directed by the 5' terminal nucleotide. Cell 133 : 116–127.
82. YeR, WangW, IkiT, LiuC, WuY, et al. (2012) Cytoplasmic assembly and selective nuclear import of arabidopsis ARGONAUTE4/siRNA complexes. Mol Cell 46 : 859–870.
83. JonesL, KeiningT, EamensA, VaistijFE (2006) Virus-induced gene silencing of argonaute genes in Nicotiana benthamiana demonstrates that extensive systemic silencing requires Argonaute1-like and Argonaute4-like genes. Plant Physiol 141 : 598–606.
84. LiJF, ChungHS, NiuY, BushJ, McCormackM, et al. (2013) Comprehensive protein-based artificial microRNA screens for effective gene silencing in plants. Plant Cell 25 : 1507–1522.
85. PanstrugaR (2004) A golden shot: how ballistic single cell transformation boosts the molecular analysis of cereal–mildew interactions. Mol Plant Pathol 5 : 141–148.
86. CaldoRA, NettletonD, PengJ, WiseRP (2006) Stage-specific suppression of basal defense discriminates barley plants containing fast - and delayed-acting Mla powdery mildew resistance alleles. Mol Plant Microbe Interact 19 : 939–947.
87. MaC, LuY, BaiS, ZhangW, DuanX, et al. (2014) Cloning and Characterization of miRNAs and Their Targets, Including a Novel miRNA-Targeted NBS–LRR Protein Class Gene in Apple (Golden Delicious). Mol Plant 7 : 218–230.
88. LvS, NieX, WangL, DuX, BiradarSS, et al. (2012) Identification and characterization of microRNAs from barley (Hordeum vulgare L.) by high-throughput sequencing. Int J Mol Sci 13 : 2973–2984.
89. PeriyannanS, MooreJ, AyliffeM, BansalU, WangX, et al. (2013) The gene Sr33, an ortholog of barley Mla genes, encodes resistance to wheat stem rust race Ug99. Science 341 : 786–788.
90. JordanT, SeeholzerS, SchwizerS, TollerA, SomssichIE, et al. (2011) The wheat Mla homologue TmMla1 exhibits an evolutionarily conserved function against powdery mildew in both wheat and barley. Plant J 65 : 610–621.
91. GuoX, GuiY, WangY, ZhuQ-H, HelliwellC, et al. (2008) Selection and mutation on microRNA target sequences during rice evolution. BMC Genomics 9 : 454.
92. ChenK, RajewskyN (2006) Natural selection on human microRNA binding sites inferred from SNP data. Nat Genet 38 : 1452–1456.
93. SaundersMA, LiangH, LiWH (2007) Human polymorphism at microRNAs and microRNA target sites. Proc Natl Acad Sci U S A 104 : 3300–3305.
94. WangY, ShenD, BoS, ChenH, ZhengJ, et al. (2010) Sequence variation and selection of small RNAs in domesticated rice. BMC Evol Biol 10 : 119.
95. TodescoM, BalasubramanianS, CaoJ, OttF, SureshkumarS, et al. (2012) Natural variation in biogenesis efficiency of individual Arabidopsis thaliana microRNAs. Curr Biol 22 : 166–170.
96. WeiF, WingRA, WiseRP (2002) Genome dynamics and evolution of the Mla (powdery mildew) resistance locus in barley. Plant Cell 14 : 1903–1917.
97. ShirasuK, Schulze-LefertP (2000) Regulators of cell death in disease resistance. Plant Mol Biol 44 : 371–385.
98. HoltBF, BelkhadirY, DanglJL (2005) Antagonistic control of disease resistance protein stability in the plant immune system. Science 309 : 929–932.
99. Schulze-LefertP, VogelJ (2000) Closing the ranks to attack by powdery mildew. Trends Plant Sci 5 : 343–348.
100. Stirnweis D, Milani SD, Brunner S, Herren G, Buchmann G, et al. (2014) Suppression among alleles encoding nucleotide-binding–leucine-rich repeat resistance proteins interferes with resistance in F1 hybrid and allele-pyramided wheat plants. Plant J: doi:10.1111/tpj.12592. [Epub ahead of print].
101. Hurni S, Brunner S, Stirnweis D, Herren G, Peditto D, et al. (2014) The powdery mildew resistance gene Pm8 derived from rye is suppressed by its wheat ortholog Pm3. Plant J: doi:10.1111/tpj.12592. [Epub ahead of print].
102. NarusakaM, KuboY, HatakeyamaK, ImamuraJ, EzuraH, et al. (2013) Interfamily transfer of dual NB-LRR genes confers resistance to multiple pathogens. PLoS One 8: e55954.
103. ZukerM (2003) Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 31 : 3406–3415.
104. DongY, Burch-SmithTM, LiuY, MamillapalliP, Dinesh-KumarSP (2007) A ligation-independent cloning tobacco rattle virus vector for high-throughput virus-induced gene silencing identifies roles for NbMADS4-1 and -2 in floral development. Plant Physiol 145 : 1161–1170.
105. ZhengL, BaumannU, ReymondJL (2004) An efficient one-step site-directed and site-saturation mutagenesis protocol. Nucleic Acids Res 32: e115.
106. ChenC, RidzonDA, BroomerAJ, ZhouZ, LeeDH, et al. (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33: e179.
107. LiuB, LiP, LiX, LiuC, CaoS, et al. (2005) Loss of function of OsDCL1 affects microRNA accumulation and causes developmental defects in rice. Plant Physiol 139 : 296–305.
108. PallGS, HamiltonAJ (2008) Improved northern blot method for enhanced detection of small RNA. Nat Protoc 3 : 1077–1084.
109. Van OoijenG, MayrG, KasiemMM, AlbrechtM, CornelissenBJ, et al. (2008) Structure-function analysis of the NB-ARC domain of plant disease resistance proteins. J Exp Bot 59 : 1383–1397.
110. LaemmliUK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227 : 680–685.
111. LiuY, SchiffM, MaratheR, Dinesh-KumarSP (2002) Tobacco Rar1, EDS1 and NPR1/NIM1 like genes are required for N-mediated resistance to tobacco mosaic virus. Plant J 30 : 415–429.
Štítky
Genetika Reprodukčná medicína
Článek Large-scale Metabolomic Profiling Identifies Novel Biomarkers for Incident Coronary Heart DiseaseČlánek Notch Signaling Mediates the Age-Associated Decrease in Adhesion of Germline Stem Cells to the NicheČlánek Phosphorylation of Mitochondrial Polyubiquitin by PINK1 Promotes Parkin Mitochondrial TetheringČlánek Natural Variation Is Associated With Genome-Wide Methylation Changes and Temperature SeasonalityČlánek Overlapping and Non-overlapping Functions of Condensins I and II in Neural Stem Cell DivisionsČlánek Unisexual Reproduction Drives Meiotic Recombination and Phenotypic and Karyotypic Plasticity inČlánek Tetraspanin (TSP-17) Protects Dopaminergic Neurons against 6-OHDA-Induced Neurodegeneration inČlánek ABA-Mediated ROS in Mitochondria Regulate Root Meristem Activity by Controlling Expression inČlánek Mutations in Global Regulators Lead to Metabolic Selection during Adaptation to Complex EnvironmentsČlánek The Evolution of Sex Ratio Distorter Suppression Affects a 25 cM Genomic Region in the Butterfly
Článok vyšiel v časopisePLOS Genetics
Najčítanejšie tento týždeň
2014 Číslo 12- Gynekologové a odborníci na reprodukční medicínu se sejdou na prvním virtuálním summitu
- Je „freeze-all“ pro všechny? Odborníci na fertilitu diskutovali na virtuálním summitu
-
Všetky články tohto čísla
- Stratification by Smoking Status Reveals an Association of Genotype with Body Mass Index in Never Smokers
- Genome Wide Meta-analysis Highlights the Role of Genetic Variation in in the Regulation of Circulating Serum Chemerin
- Occupancy of Mitochondrial Single-Stranded DNA Binding Protein Supports the Strand Displacement Mode of DNA Replication
- Distinct Genealogies for Plasmids and Chromosome
- Large-scale Metabolomic Profiling Identifies Novel Biomarkers for Incident Coronary Heart Disease
- Non-coding RNAs Prevent the Binding of the MSL-complex to Heterochromatic Regions
- Plasmid Flux in ST131 Sublineages, Analyzed by Plasmid Constellation Network (PLACNET), a New Method for Plasmid Reconstruction from Whole Genome Sequences
- Epigenome-Guided Analysis of the Transcriptome of Plaque Macrophages during Atherosclerosis Regression Reveals Activation of the Wnt Signaling Pathway
- The Inventiveness of Nature: An Interview with Werner Arber
- Mediation Analysis Demonstrates That -eQTLs Are Often Explained by -Mediation: A Genome-Wide Analysis among 1,800 South Asians
- Generation of Antigenic Diversity in by Structured Rearrangement of Genes During Mitosis
- A Massively Parallel Pipeline to Clone DNA Variants and Examine Molecular Phenotypes of Human Disease Mutations
- Genetic Analysis of the Cardiac Methylome at Single Nucleotide Resolution in a Model of Human Cardiovascular Disease
- Genetic Analysis of Circadian Responses to Low Frequency Electromagnetic Fields in
- The Dissection of Meiotic Chromosome Movement in Mice Using an Electroporation Technique
- Altered Chromatin Occupancy of Master Regulators Underlies Evolutionary Divergence in the Transcriptional Landscape of Erythroid Differentiation
- Syd/JIP3 and JNK Signaling Are Required for Myonuclear Positioning and Muscle Function
- Notch Signaling Mediates the Age-Associated Decrease in Adhesion of Germline Stem Cells to the Niche
- Mutation of Leads to Blurred Tonotopic Organization of Central Auditory Circuits in Mice
- The IKAROS Interaction with a Complex Including Chromatin Remodeling and Transcription Elongation Activities Is Required for Hematopoiesis
- RAN-Binding Protein 9 is Involved in Alternative Splicing and is Critical for Male Germ Cell Development and Male Fertility
- Enhanced Longevity by Ibuprofen, Conserved in Multiple Species, Occurs in Yeast through Inhibition of Tryptophan Import
- Phosphorylation of Mitochondrial Polyubiquitin by PINK1 Promotes Parkin Mitochondrial Tethering
- Recurrent Loss of Specific Introns during Angiosperm Evolution
- Natural Variation Is Associated With Genome-Wide Methylation Changes and Temperature Seasonality
- SEEDSTICK is a Master Regulator of Development and Metabolism in the Arabidopsis Seed Coat
- Overlapping and Non-overlapping Functions of Condensins I and II in Neural Stem Cell Divisions
- Unisexual Reproduction Drives Meiotic Recombination and Phenotypic and Karyotypic Plasticity in
- Tetraspanin (TSP-17) Protects Dopaminergic Neurons against 6-OHDA-Induced Neurodegeneration in
- ABA-Mediated ROS in Mitochondria Regulate Root Meristem Activity by Controlling Expression in
- Mutations in Global Regulators Lead to Metabolic Selection during Adaptation to Complex Environments
- Global Analysis of Photosynthesis Transcriptional Regulatory Networks
- Mucolipin Co-deficiency Causes Accelerated Endolysosomal Vacuolation of Enterocytes and Failure-to-Thrive from Birth to Weaning
- Controlling Pre-leukemic Thymocyte Self-Renewal
- How Malaria Parasites Avoid Running Out of Ammo
- Echoes of the Past: Hereditarianism and
- Deep Reads: Strands in the History of Molecular Genetics
- Keep on Laying Eggs Mama, RNAi My Reproductive Aging Blues Away
- Analysis of a Plant Complex Resistance Gene Locus Underlying Immune-Related Hybrid Incompatibility and Its Occurrence in Nature
- Epistatic Adaptive Evolution of Human Color Vision
- Increased and Imbalanced dNTP Pools Symmetrically Promote Both Leading and Lagging Strand Replication Infidelity
- Genetic Basis of Haloperidol Resistance in Is Complex and Dose Dependent
- Genome-Wide Analysis of DNA Methylation Dynamics during Early Human Development
- Interaction between Conjugative and Retrotransposable Elements in Horizontal Gene Transfer
- The Evolution of Sex Ratio Distorter Suppression Affects a 25 cM Genomic Region in the Butterfly
- is Required for Adult Maintenance of Dopaminergic Neurons in the Ventral Substantia Nigra
- PRL1, an RNA-Binding Protein, Positively Regulates the Accumulation of miRNAs and siRNAs in Arabidopsis
- Genetic Control of Contagious Asexuality in the Pea Aphid
- Early Mesozoic Coexistence of Amniotes and Hepadnaviridae
- Local and Systemic Regulation of Plant Root System Architecture and Symbiotic Nodulation by a Receptor-Like Kinase
- Gene Pathways That Delay Reproductive Senescence
- The Evolution of Fungal Metabolic Pathways
- Maf1 Is a Novel Target of PTEN and PI3K Signaling That Negatively Regulates Oncogenesis and Lipid Metabolism
- Formation of Linear Amplicons with Inverted Duplications in Requires the MRE11 Nuclease
- Identification of Rare Causal Variants in Sequence-Based Studies: Methods and Applications to , a Gene Involved in Cohen Syndrome and Autism
- Rrp12 and the Exportin Crm1 Participate in Late Assembly Events in the Nucleolus during 40S Ribosomal Subunit Biogenesis
- The Mutations in the ATP-Binding Groove of the Rad3/XPD Helicase Lead to -Cockayne Syndrome-Like Phenotypes
- Topoisomerase I Plays a Critical Role in Suppressing Genome Instability at a Highly Transcribed G-Quadruplex-Forming Sequence
- A Cbx8-Containing Polycomb Complex Facilitates the Transition to Gene Activation during ES Cell Differentiation
- Transcriptional Frameshifting Rescues Type VI Secretion by the Production of Two Length Variants from the Prematurely Interrupted Gene
- Association Mapping across Numerous Traits Reveals Patterns of Functional Variation in Maize
- Genome-Wide Analysis of -Regulated and Phased Small RNAs Underscores the Importance of the ta-siRNA Pathway to Maize Development
- Dissemination of Cephalosporin Resistance Genes between Strains from Farm Animals and Humans by Specific Plasmid Lineages
- The Tau Tubulin Kinases TTBK1/2 Promote Accumulation of Pathological TDP-43
- Germline Signals Deploy NHR-49 to Modulate Fatty-Acid β-Oxidation and Desaturation in Somatic Tissues of
- Microevolution of in Macrophages Restores Filamentation in a Nonfilamentous Mutant
- Vangl2-Regulated Polarisation of Second Heart Field-Derived Cells Is Required for Outflow Tract Lengthening during Cardiac Development
- Chondrocytes Transdifferentiate into Osteoblasts in Endochondral Bone during Development, Postnatal Growth and Fracture Healing in Mice
- A ABC Transporter Regulates Lifespan
- RA and FGF Signalling Are Required in the Zebrafish Otic Vesicle to Pattern and Maintain Ventral Otic Identities
- , and Reprogram Thymocytes into Self-Renewing Cells
- The miR9863 Family Regulates Distinct Alleles in Barley to Attenuate NLR Receptor-Triggered Disease Resistance and Cell-Death Signaling
- Detection of Pleiotropy through a Phenome-Wide Association Study (PheWAS) of Epidemiologic Data as Part of the Environmental Architecture for Genes Linked to Environment (EAGLE) Study
- Extensive Copy-Number Variation of Young Genes across Stickleback Populations
- The and Genetic Modules Interact to Regulate Ciliogenesis and Ciliary Microtubule Patterning in
- Analysis of the Genome, Transcriptome and Secretome Provides Insight into Its Pioneer Colonization Strategies of Wood
- PLOS Genetics
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- Tetraspanin (TSP-17) Protects Dopaminergic Neurons against 6-OHDA-Induced Neurodegeneration in
- Maf1 Is a Novel Target of PTEN and PI3K Signaling That Negatively Regulates Oncogenesis and Lipid Metabolism
- The IKAROS Interaction with a Complex Including Chromatin Remodeling and Transcription Elongation Activities Is Required for Hematopoiesis
- Echoes of the Past: Hereditarianism and
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



