-
Články
- Časopisy
- Kurzy
- Témy
- Kongresy
- Videa
- Podcasty
MMS Exposure Promotes Increased MtDNA Mutagenesis in the Presence of Replication-Defective Disease-Associated DNA Polymerase γ Variants
Thousands of mitochondrial DNA (mtDNA) per cell are necessary to maintain energy required for cellular survival in humans. Interfering with the mtDNA polymerase can result in mitochondrial diseases and mitochondrial toxicity. Therefore, it is important to explore new genetic and environmental mechanisms that alter the effectiveness and accuracy of mtDNA replication. This genetic study uses the budding yeast to demonstrate that heterozygous strains harboring disease-associated mutations in the mtDNA polymerase gene in the presence of a wild type copy of the mtDNA polymerase are associated with increased mtDNA point mutagenesis in the presence of methane methylsulfonate, a known base damaging agent. Further observations suggest that the inability of disease-associated variants to replicate mtDNA resulted in increased vulnerability to irreparable base damage that was likely to result in mutations when replicated. Also, this study showed that trace amounts of the environmental contaminant cadmium chloride impairs mtDNA replication but eliminates damage-induced mutagenesis in the remaining functional mitochondria. This interplay between disease-associated variant and wild type polymerase offers new insights on possible disease variation and implicates novel environmental consequences for compound heterozygous patients.
Published in the journal: MMS Exposure Promotes Increased MtDNA Mutagenesis in the Presence of Replication-Defective Disease-Associated DNA Polymerase γ Variants. PLoS Genet 10(10): e32767. doi:10.1371/journal.pgen.1004748
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1004748Summary
Thousands of mitochondrial DNA (mtDNA) per cell are necessary to maintain energy required for cellular survival in humans. Interfering with the mtDNA polymerase can result in mitochondrial diseases and mitochondrial toxicity. Therefore, it is important to explore new genetic and environmental mechanisms that alter the effectiveness and accuracy of mtDNA replication. This genetic study uses the budding yeast to demonstrate that heterozygous strains harboring disease-associated mutations in the mtDNA polymerase gene in the presence of a wild type copy of the mtDNA polymerase are associated with increased mtDNA point mutagenesis in the presence of methane methylsulfonate, a known base damaging agent. Further observations suggest that the inability of disease-associated variants to replicate mtDNA resulted in increased vulnerability to irreparable base damage that was likely to result in mutations when replicated. Also, this study showed that trace amounts of the environmental contaminant cadmium chloride impairs mtDNA replication but eliminates damage-induced mutagenesis in the remaining functional mitochondria. This interplay between disease-associated variant and wild type polymerase offers new insights on possible disease variation and implicates novel environmental consequences for compound heterozygous patients.
Introduction
Mitochondrial DNA (mtDNA) maintenance is necessary for the majority of ATP production in eukaryotic cells. The inability to properly replicate mtDNA potentially impacts human health in several ways. The premature aging phenotype of POLG exonuclease deficient mice indicates that increased mtDNA mutagenesis can be detrimental [1]–3. Also, mutations in genes encoding the mitochondrial replisome, including DNA polymerase γ (pol γ, encoded by POLG), contribute to mitochondrial diseases characterized by mtDNA depletion, deletions, or point mutations [4]–[14]. Additionally, environmental changes can modify mitochondrial biology and potentially impact health. Chain-terminating nucleotide analogs used in anti-viral therapy impair mtDNA replication and can result in mitochondrial toxicity [15]. Antioxidants and exercise have been shown in model systems to improve mitochondrial function and suppress the premature aging phenotype associated with increased point mutations and deletions [3], [14]. Therefore, environmental changes can be important for mitochondrial function, and the mechanisms that cause mtDNA mutations warrant further study.
Currently hundreds of POLG mutations have been identified in patients with mitochondrial disease such as Alpers syndrome, progressive external ophthalmoplegia, and ataxia-neuropathy syndrome (mutations listed in http://tools.niehs.nih.gov/polg/) [16]. Pol γ-related mitochondrial diseases display a wide variety of severities. For instance, Alpers syndrome manifests in infants and young children, and these patients rarely live through their first decade of life [17]. Alternatively, patients with progressive external ophthalmoplegia (PEO) and sensory ataxia neuropathy, dysarthria, and ophthalmoparesis (SANDO) often are asymptomatic until>20 years of age [4], [18].
The catalytic subunit of pol γ contains DNA polymerase, 3′-5′ exonuclease, and 5′ dRP lyase activities, with known discrete polymerase and exonuclease domains [19]–[22]. Among the POLG mutations associated with mitochondrial disease, many have been characterized biochemically and shown to disrupt polymerase activity [5]–[10], [13], [14], [23]–[27]. POLG polymerase variants H932Y, R943H, and Y955C alter dNTP-interacting side chains and are associated with less than 1% polymerase activity [26]. Polymerase variants G848S, T851A, R852C, and R853Q also reduce polymerase activity to <1% of wild type activity; in addition, G848S also exhibits a DNA-binding defect [24]. Although mutagenic effects of point mutations that disrupt exonuclease activity have been well established, disease-associated mutations in the human exonuclease domain surprisingly do not disrupt the exonuclease activity in Mip1 [9], [10]. These disease associated exonuclease mutations have not been studied in the human enzyme.
S. cerevisiae has been useful to characterize Pol γ functionality with mutations that alter amino acids within conserved stretches between human POLG and yeast MIP1, most of which are in the polymerase domain [28], [29]. Mitochondrial functionality and mtDNA point mutagenesis have been determined in various mutants using assays that measure frequency of petite colony formation (ie, lacking mitochondrial function either with [rho−] or without mtDNA [rho0]) and erythromycin resistance, respectively [30]. For instance, mutations that alter the catalytic aspartates (eg, Asp171 and Asp 230 in yeast) in the exonuclease domain are associated with 1440-fold and 160-fold increases in point mutagenesis [31]–[33] and deletions between direct repeats in mice, respectively [32], [33]; the corresponding increases in yeast are 2000-fold for point mutagenesis [31] and 90-fold for deletions between direct repeats [12], [33]. These increases in mtDNA mutagenesis establish Asp171 and Asp 230 as critical domains for “proofreading” against misinsertions. Surprisingly, disease-associated mutations in the exonuclease domain are associated with only modest increases in mutant frequency, suggesting that exonuclease activity is functionally sufficient to correct misinsertions [9], [10]. Many of the disease-associated mutations to Mip1 eliminated the ability to replicate mtDNA and were associated with petite colony formation, including human variants R807C, R807P, R853W, N864S, G923D, H932Y, K947R, G1076V, R1096C, S1104C, and V1106I and Alpers-associated mutations G848S, T851A, R853Q, D930N, A957P, P1073A, and R1096H [9]. However, several strains that contain variants, including R853Q and Q308H (R656Q and Q264H in yeast) coexpressed with wild type MIP1 to maintain mtDNA showed significant increases in mutagenesis, and no mechanism has been described for this increase [9].
Environmental agents have also been shown to affect mitochondrial DNA replication both positively and negatively. The presence of antioxidants such as MitoQ and dihydrolipoic acid have been shown to improve mitochondrial function in mutants with disease-associated polymerase domain mutations by salvaging reactive oxygen species [14]. These results suggest an increase of oxidative damage in mtDNA in model systems with defective pol γ, a hypothesis supported by the increased levels of 8-oxo-dG in the mtDNA of a transgenic mouse model that overexpressed the Y955C mutant variant in cardiac tissue [34]. Methyl methanesulfonate (MMS) is an alkylating agent that is associated with increases in mtDNA base damage [35]. Interestingly, in embryonic fibroblasts, 2 mM MMS was associated with persistent mtDNA damage but not with loss of mtDNA or mitochondrial function [36]. In yeast, repair of alkylation damage of mtDNA by MMS involves Apn1 nuclease [37], and Ntg1 [38]. Also, chronic exposure to trace amounts of the known human carcinogen, cadmium chloride, resulted in loss of mitochondrial function [39]. Because cadmium also results in extreme nuclear hypermutability [39], the possibility that cadmium alters mtDNA replication warrants further study.
Base excision repair is active in yeast and human mitochondria and protects cells against alkylation damage [40]. However, lesions on single-stranded DNA are not substrates for base excision repair because there is no complementary strand with which it can reanneal and are therefore highly mutagenic [41]–[43]. The proposed model of asymmetrical mtDNA replication of human mtDNA suggests that single-stranded mtDNA is exposed even in optimal conditions and could be more vulnerable under conditions of decreased replication efficiency [44]. To test whether reduction in mtDNA replication efficiency could leave the cell vulnerable to mutagenic base damage, mtDNA mutagenesis in previously characterized disease-associated mutants were tested in the presence of MMS.
Results
Chronic MMS exposure increases mtDNA point mutagenesis in mip1 mutants
Mutations in mip1 that result in changes in conserved amino acids previously have been shown to cause defective mtDNA replication and, in some cases, increased mtDNA mutagenesis when coexpressed with wild type MIP1 [9]. These experiments were performed in haploid heteroallelic strains with intact chromosomal wild type MIP1 and one of 31 mutant mip1 alleles on a centromeric plasmid with the endogenous promoter. This study interrogated some of the heteroallelic strains and newly created diploid heterozygotes to determine mtDNA point mutagenesis of the gene encoding the 16S ribosomal subunit that confers resistance to erythromycin and the fraction of cells unable to grow on glycerol which requires mitochondrial function.
To test whether the presence of the catalytically defective mutant variant increases the vulnerability of mtDNA to base damage and mutagenic replication by the wild type polymerase, heteroallelic S. cerevisiae strains expressing either Q264H, R656W, R853H, or wild type MIP1 on a centromeric plasmid and chromosomal wild type MIP1 were grown in the presence of sublethal concentrations (3 mM) of MMS (see Table 1 for list of all genotypes). None of these mutant proteins are capable of maintaining mtDNA without the presence of wild type Mip1 [9]. MMS exposure caused a modest 2-fold increase in mutagenesis in the wild type control as compared to no exposure to MMS, whereas a greater increase in mtDNA mutagenesis—17-fold, 11-fold, and 6-fold—was observed in strains expressing Q264H, R656W, and R853H, respectively (Figure 1 and Table 1). Absolute mtDNA mutant frequencies after MMS exposures were associated with 30-fold, 18-fold, and 7-fold increases in strains with Q264H, R656W, and R853H mutant variants, respectively, compared with that of the wild type strain. The control strain with wild type MIP1 on both the centromeric plasmid and the chromosome was associated with only 2.7-fold increase in MMS-induce mutagenesis compared with no MMS exposure.
Fig. 1. Methyl methanesulfonate exposure increases mtDNA point mutagenesis in heteroallelic strains encoding Mip1 variants. 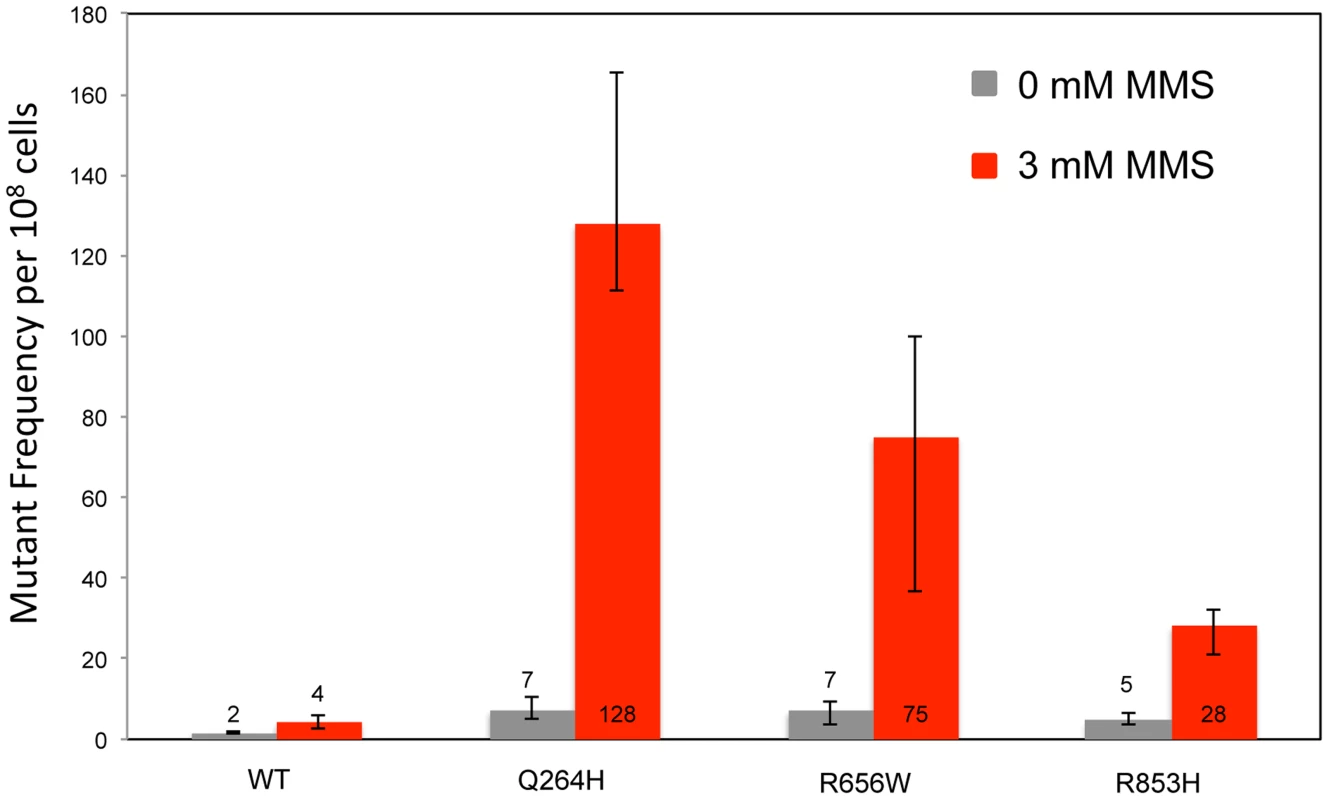
Median frequency (±95% CI) of erythromycin-resistant mutants per 108 rho+ cells of haploid strains with wild type MIP1 encoded in the chromosome and a centromeric plasmid encoding MIP1 or mip1 with various disease-associated mutations in the presence (3 mM) or absence (0 mM) of MMS. Mutant frequencies were determined from 40 (for R656W and R853H) and 80 (for MIP1 and Q264H) independent cultures from at least 2 separate experiments. CI, confidence interval; MMS, methyl methanesulfonate; mtDNA, mitochondrial DNA. Tab. 1. List of Pol γ mutations analyzed in this study and summary of results. 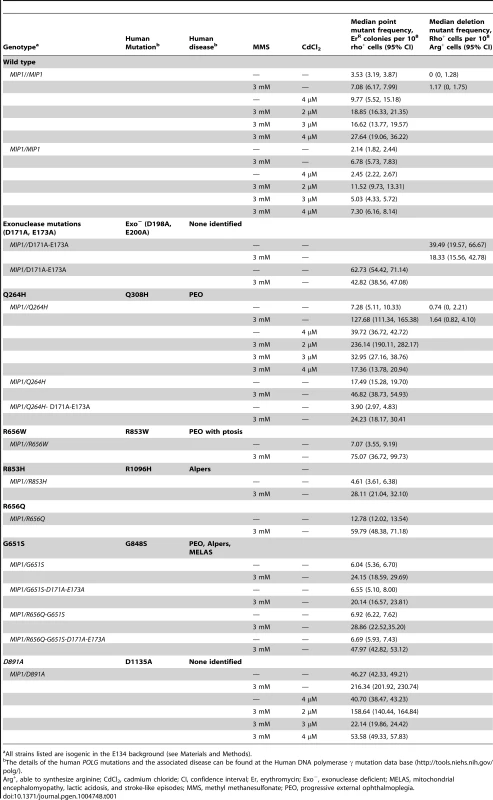
All strains listed are isogenic in the E134 background (see Materials and Methods). To avoid the possibility of multicopy expression of the Mip1 variant from the plasmid, heterozygotes were created with mutations that encode Mip1 with defective exonuclease activity [31] or amino acid variants Q264H, R656Q, G651S, or D891A (Table 1). D891A is not associated with mitochondrial disease, but the conserved aspartate in human POLG (Asp1135) is essential for binding catalytic Mg2+ in the active site [19]. Alanine substitution of this equivalent residue in other human DNA polymerases has been shown to disrupt binding of the catalytic Mg2+, eliminating DNA polymerase activity but not binding to DNA binding [45]. With the exception of Q264H, biochemical characterizations have been reported for the remaining mutant variants [24]. In agreement with previous observations [9], disruption of Mip1 exonuclease activity increased mutagenesis; however, there was no additional increase in MMS-induced mutagenesis (Figure 2 and Table 1). Compared with wild type, Q264H, R656Q, and D891A heterozygotes were associated with 7-fold, 8-fold, and 19-fold increases in absolute MMS-induce mutant frequency. In fact, the resulting increases in mutant frequency were approximately equal to or greater than that of the exonuclease defective variant (Figure 2). Strikingly, MMS exposure of the D891A variant resulted in an approximately 3-fold increase in mutant frequency compared with that of the exonuclease deficient variant. These results demonstrate that catalytically inactive or less active polymerases, whether generated through a site-directed mutation or a disease-associated mutation, participate in a mechanism that causes MMS-induced mutations.
Fig. 2. Mutations that disrupted Mip1 polymerase activity caused increased mutant frequency after MMS exposure. 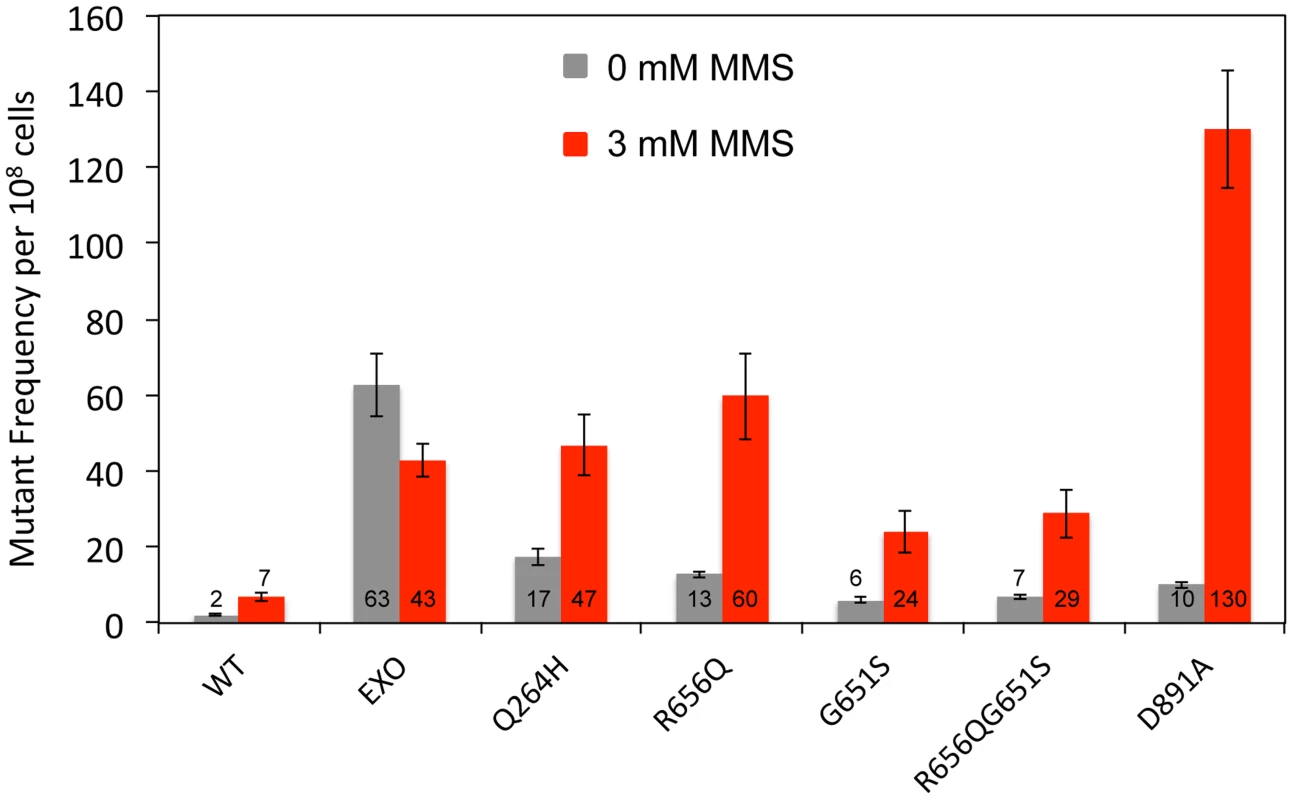
Median frequency (±95% CI) of erythromycin-resistant mutants per 108 rho+ cells of heterozygous diploid strains containing both MIP1 and mip1 with disease-associated mutation, exonuclease disrupting (EXO), or polymerase disrupting mutations in the presence (3 mM) or absence (0 mM) of MMS. Mutant frequencies were determined from at least 20 different independent cultures. CI, confidence interval; MMS, methyl methanesulfonate. Mip1 variants R656Q and G651S are homologous to human disease variant R853Q and G848S, respectively, which both exhibit ≤1% catalytic activity [24]. However, G848S uniquely displayed an approximately 5-fold reduction in DNA binding [24]. Interestingly, Mip1 G651S variant resulted in fewer MMS-induced mutations compared with the other polymerase variants, suggesting that DNA binding may be important for the mechanism. To test this hypothesis, a R656Q/G651S double mutant was created, and MMS-induced mutagenesis was compared with each single mutant. Mutagenesis after exposure to MMS in the double mutant was indistinguishable from the G651S variant and lower than in a single R656Q mutant (Figure 2), suggesting that DNA binding is an important component of the R656Q mutator effect.
Mutant Mip1 variants do not replicate mtDNA
To test whether Mip1 mutant variants participate in the bulk of mtDNA replication, mutagenesis was measured in heterozygous diploids that had one wild type MIP1 allele and one allele containing mutations that disrupt exonuclease activity and encode the Q264H and G651S variants in cis. Unlike exonuclease-deficient Mip1 without other mutations, eliminating exonuclease activity did not increase mutagenesis in either the Q264H or G651S variant, with or without MMS exposure (Figure 3 and Table 1). In fact, mutation frequency unexpectedly decreased when the exo− and Q264H were in cis. These results suggest minimal contributions of Q264H and G651S mutant variants to mtDNA replication.
Fig. 3. Q264H and G651S Mip1 variants were not significantly involved in mtDNA replication. 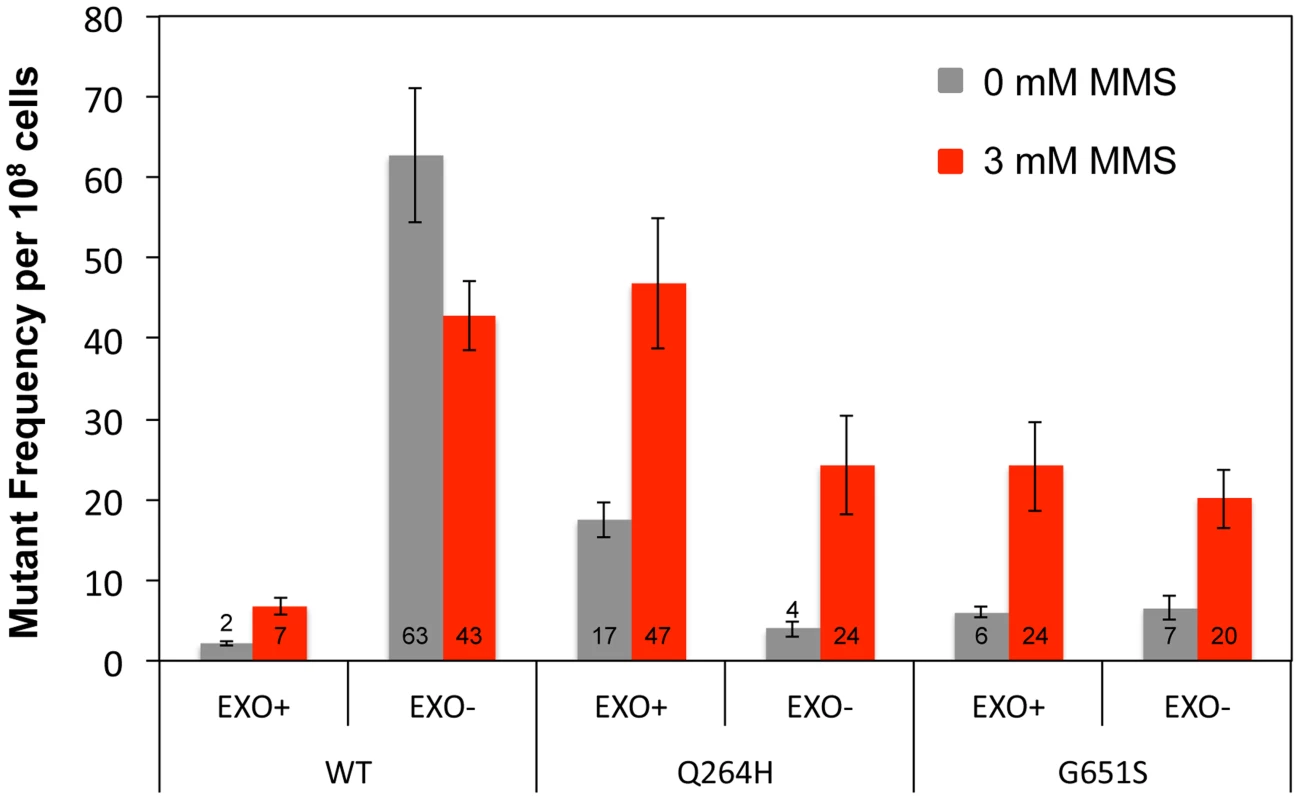
Median frequency (±95% CI) of erythromycin-resistant mutants per 108 rho+ cells of heterozygous diploid strains containing both a wild type MIP1 allele and a mip1 allele with disease-associated mutation with or without exonuclease disrupting mutations in cis in the presence (3 mM) or absence (0 mM) of MMS. Mutant frequencies were determined from at least 20 different independent cultures. CI, confidence interval, MMS, methyl methanesulfonate; mtDNA, mitochondrial DNA. MMS exposure is associated with increased C:G→G:C transversions
Resistance to erythromycin is conferred by one mutation at any of the following nucleotides: 1950 (G to T or G to A), 1951 (A to T, A to G, or A to C), 1952 (A to T or A to G), 3993 (C to G), or an insertion of G between nucleotide 1949 and 1950 of the 21S rRNA gene (Gen Bank accession number L36885) [31], [46], [47]. To determine if there was a change in the spectrum of mutations associated with MMS exposure, PCR fragments containing nucleotides 1797–1995 and 3895–4107 of the 16S ribosomal subunit gene from erythromycin resistant mutants were sequenced. Only one mutant was taken from each original culture to ensure that each mutation represented a separate event. In the absence of MMS, this and prior studies [46]–[48] demonstrate that A:T→G:C and A:T→T:A were the most frequent mutations (Figure 4 and Table S1). In this and previous studies, C to G mutations were not detected in wild type strains and were detected in only 5% of Δrev1 strains [46]. Interestingly, we found that exposure to MMS was associated with a significant change in mutational spectrum, wherein the most common mutation was C:G→G:C transversions in both the wild type strain (40%) and Q264H heteroallelic strain (45%). G:C→A:T was the only mutation other than C:G mutation detected, but it was only detected in the wild type strain and very low levels (7%). These results suggest that cytosine or guanine is especially sensitive to methylation by MMS, leading mostly to misincorporated cytosines or guanines, similar to previous studies [43].
Fig. 4. Methyl methanesulfonate exposure resulted in C:G→G:C transversions in mtDNA. 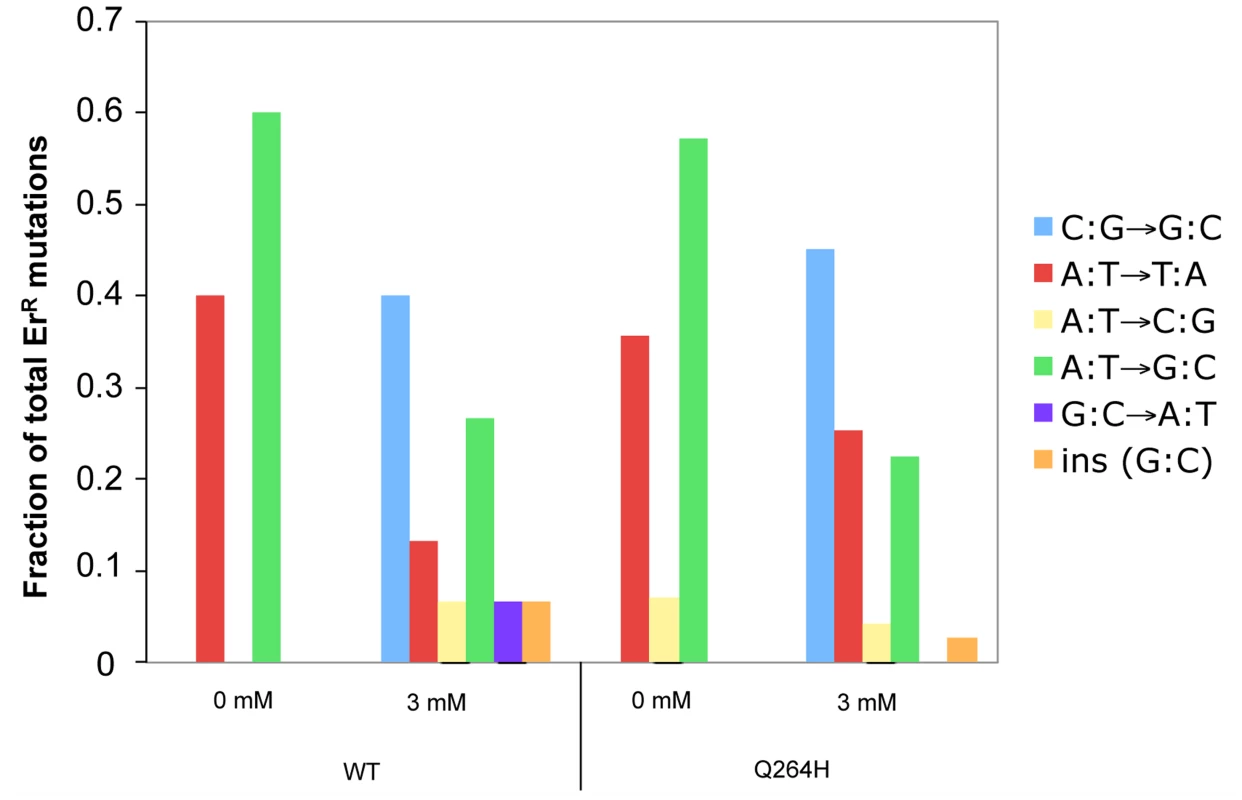
Fractions of different base substitutions in heterozygous strains with a wild type (WT) MIP1 allele and a mutant mip1 encoding the Q264H variant in the presence (3 mM) or absence (0 mM) of MMS. The following number of mutants was sequenced for each group: Wt (0 mM MMS), 5; Wt (3 mM MMS), 15; Q264H (0 mM MMS), 28; Q264H (0 mM MMS), 71. P-values for the C to G mutations were calculated using a one-tailed Fisher's Exact test. The p-value for the 0.46 fraction of G to C mutations with the Q264H and 3 mM MMS as compared to no MMS was <0.0001. The p-value of the 0.40 fraction of G to C mutations for the WT comparing 3 mM MMS with no MMS was 0.17 and <0.0001 using our WT no MMS data and the WT data from Kalifa and Sia [46], respectively. MMS, methyl methanesulfonate; mtDNA, mitochondrial DNA; Wt, wild type. MMS exposure does not increase MtDNA mutations by reducing exonuclease activity
Increased mutagenesis can arise by disrupting exonuclease activity and/or increasing the frequency of nucleotide misincorporation events. Previous work demonstrated that mitochondrial deletions between direct repeats of 21 nucleotides were rare events that were suppressed by exonuclease activity [12]. To test whether MMS promotes deletion formation, haploid deletion reporter strains that were heteroallelic for wild type, exonuclease-deficient, or Q264H variants of Mip1 were used to measure frequency of deletions between 21 nucleotide direct repeats that flank an ARG8 insertion in the mitochondrial genome. Frequency of deletions between direct repeats was increased (40-fold) in the strain with the exonuclease-deficient Mip1 variant but was not significantly different in the strain with the Q264H variant compared with wild type (Figure 5 and Table 1). MMS had no significant effect on deletion formation in any of the three strains, suggesting that the effect of MMS is specific to point mutations.
Fig. 5. Methyl methanesulfonate exposure has no significant effect on deletion mutations between 21mer repeats. 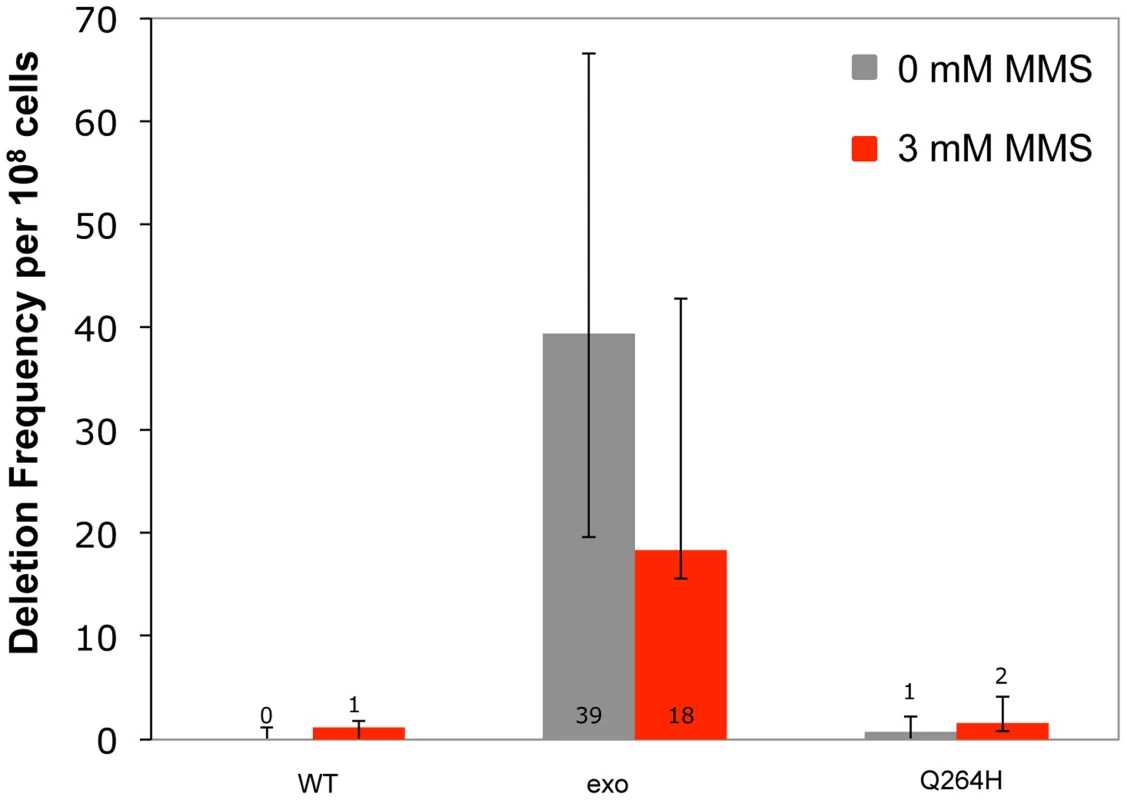
Median frequency (±95% CI) of deletions between 96-mer direct repeats per 108 Arg+ cells of heteroallelic strains containing a centromeric plasmid encoding MIP1 or mip1 with an exonuclease-disrupting mutation or a mutation encoding the Q264H variant in the presence (3 mM) or absence (0 mM) of methyl methanesulfonate. Mutant frequencies were determined from at least 20 different independent cultures. CI, confidence interval; MMS, methyl methanesulfonate. Trace amounts of cadmium chloride reduces mtDNA content and suppresses mtDNA mutagenesis
Mutations associated with MMS exposure occur in strains with mutants that affect mtDNA replication, suggesting that the mechanism requires suboptimal mtDNA replication. Therefore, it is possible that an environmental agent that reduces mtDNA replication may also be associated with MMS-induced mutagenesis in wild type cells. This was tested by treating wild type and mutant mip1 strains with CdCl2. Exposure to 3 µM CdCl2 was associated with increased petite formation frequency of about 30% with stepwise increases at 4 and 5 µM of about 60% and 80%, respectively (Figure 6) in both wild type and Q264H mutants. Exposure to 5 µM CdCl2 was associated with 3.6-fold reduction in mtDNA among rho+ cells, from 30.6±4.0 copies per cell without CdCl2 exposure to 8.5±0.9 copies per cell with 5 µM CdCl2, suggesting that trace amounts of CdCl2 are associated with mtDNA depletion.
Fig. 6. Cadmium chloride increases petite formation and decreases mtDNA replication. 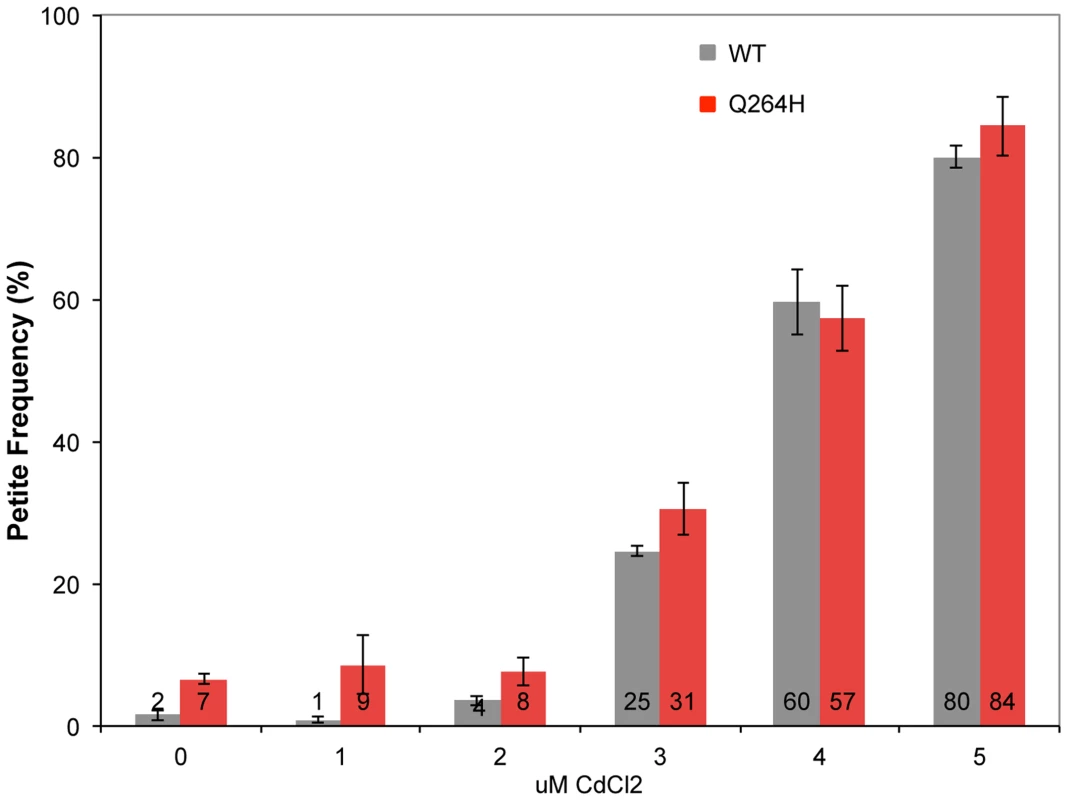
Petite colony formation of heteroallelic strains containing a centromeric plasmid encoding MIP1 or mip1 with the mutation encoding the Q264H variant after exposure to different concentrations of cadmium chloride. mtDNA, mitochondrial DNA; nDNA, nuclear DNA. Mitochondrial DNA mutagenesis was assayed in homozygous wild type diploid cells and Q264H heterozygotes to test if MMS-induced mtDNA mutagenesis occurs with CdCl2. Exposure to 4 µM CdCl2 had no effect on mtDNA mutagenesis in either the wild type or Q264H mutant strains (Figure 7). Therefore, the mutagenic effect of CdCl2 is specific to nuclear DNA [39]. Exposure to both MMS and CdCl2 resulted in no increase in mtDNA mutagenesis. However, the mutagenic effect of MMS observed in the heteroallelic Q264H strain was completely suppressed by 3 µM or 4 µM CdCl2. The lack of mtDNA mutagenicity and the suppression of the MMS-induced mtDNA mutagenesis by trace amounts of CdCl2 were recapitulated in heterozygotes expressing the D891A mutant variant (Table 1). Although reduced efficiency of mtDNA replication by a mutant variant is associated with MMS-induced mutagenesis, these results suggest that processes that reduce mtDNA replication suppress MMS mutagenesis.
Fig. 7. Exposure to CdCl2 suppressed the MMS-induced increase in MtDNA mutant frequency. 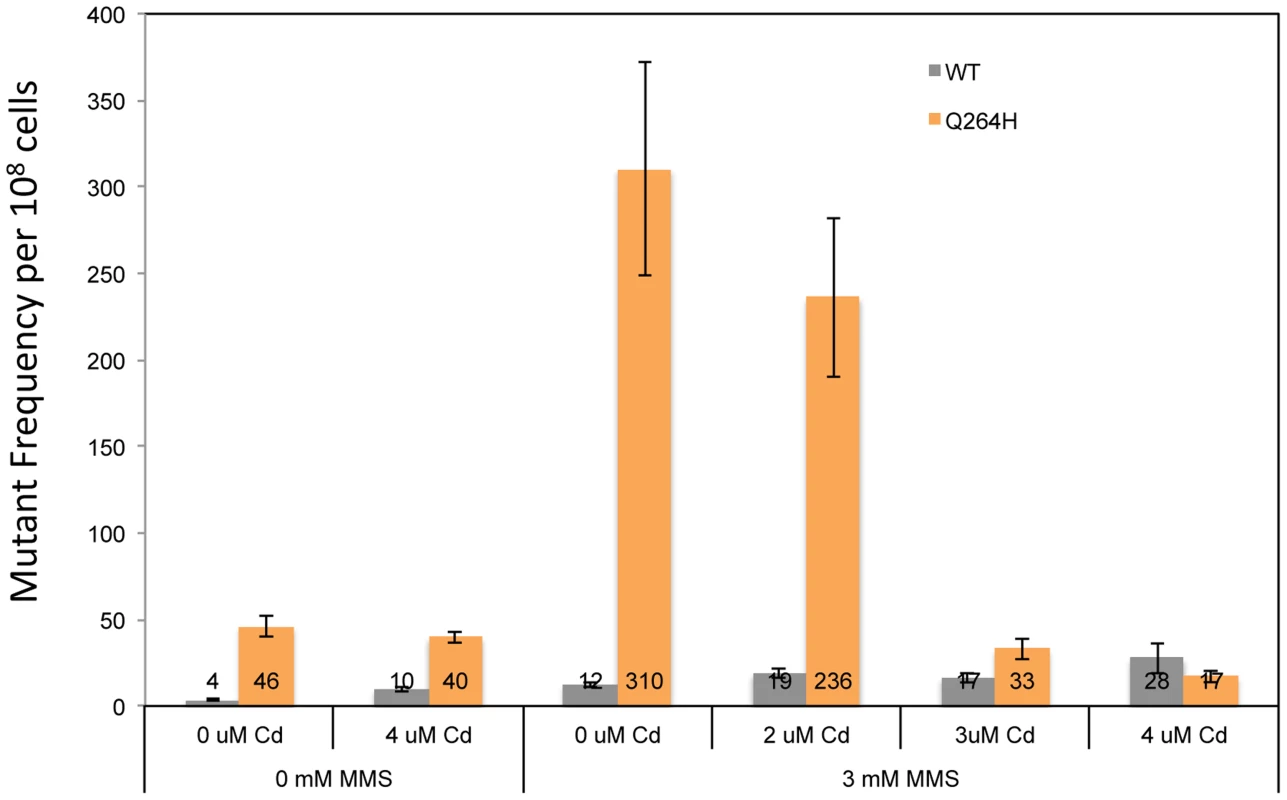
Median frequency (±95% CI) of erythromycin-resistant mutants per 108 rho+ cells of heterozygous diploid strains containing both a wild type MIP1 allele and a mip1 allele encoding the Q264H variant in the presence or absence of methyl methanesulfonate (0 or 3 mM) or CdCl2 (0, 2, 3, or 4 µM). Mutant frequencies were determined from at least 20 different independent cultures. CI, confidence interval; MMS, methyl methanesulfonate, mtDNA, mitochondrial DNA. Discussion
This study demonstrates a novel mechanism of MMS-induced mtDNA point mutagenesis, mostly C:G→G:C transversions, in heterozygous strains with a disease-associated mutation that disrupts polymerase activity. The frequency of the mutagenesis appeared to be modulated by the activity of the mutant variant in that the possible DNA binding defect observed in the human homologue to G651S reduced MMS-induced mutagenesis. This study showed that alleles with added exonuclease defect were not associated with increased mutagenesis (with or without MMS exposure), suggesting that the mutant variant replicated little or none of the mtDNA that remained and was propagated in the cell. Finally, chronic exposure to trace amounts (3–5 µM) of CdCl2 resulted in the inability to replicate mtDNA, which surprisingly did not increase but instead suppressed MMS-induced mutagenesis. These results are the first to support a mechanism to understand the interplay between polymerases in heterozygous cells and reveal a novel pathway for environmentally-induced mtDNA mutagenesis.
There are few known pathways that increase mtDNA mutations in yeast or humans. Mutations that disrupt the exonuclease activity of the mtDNA polymerase have been shown to cause increased mtDNA mutagenesis. One study used a reversion assay in yeast to identify several mitochondrial mutators including pos5, a gene that encodes an NADPH kinase [49]. Other genes associated with increased mtDNA mutagenesis—such as hap2, fen1, and ntg1—have been identified, but their effect on mtDNA mutagenesis has been modest or requiring long incubation times [49]–[51]. Even base damaging agents such as H2O2 and MMS are associated with modest increases in mtDNA but only in strains without crucial repair pathways [52].
Previously, disease-associated mutations were shown to increase mtDNA mutagenesis, but these mutations also led to the inability to maintain functional mitochondria because of mtDNA depletion [9]. The increase in mutant frequency in some strains was evident in this study in the unexposed controls (Figures 1 and 2). Therefore, it was difficult to ascertain how mutant polymerases that in some cases were suggested to have little or no activity could significantly increase mtDNA mutagenesis. Exposing the heteroallelic strains to sublethal doses of MMS resulted in up to 30-fold increases in mtDNA mutant frequency (Figure 1). Interestingly, repeating the experiment in heterozygous strains recapitulated the MMS-induced increase, albeit at a lower frequency (Figure 2). The heteroallelic strain contains the mutant mip1 and its endogenous promoter on a centromeric plasmid that has been previously shown to have 1–2 copies of the gene per cell [13]. It is possible that MMS-induced mutagenesis is sensitive to differences in the number of Mip1 mutant copies. The fact that the heteroallelic strains are haploid whereas the heterozygotes are diploid could suggest that increased copy number of other replication proteins may alter the MMS-induced mutagenesis phenotype, although there is probably no difference in the protein concentration relative to the genome copy number.
MMS-induced mutagenesis was shown in several disease-associated mutants and the polymerase defective mutant but not the exonuclease-deficient mutant. Although Q264H is an exonuclease domain disease-associated variant, it was previously shown to be detrimental to mtDNA replication. Mutations that result in the Mip1 R656Q and G651S variants are homologous to the mutations in the human pol γ thumb domain that were biochemically characterized to have <1% polymerase activity including a 5-fold reduction in DNA binding affinity in the G651S homologue [24]. Interestingly, G651S is associated with reduced mtDNA mutant frequency and suppression of R656Q when the two mutations are in cis. These results suggest that lower DNA binding affinity impedes the mechanism of MMS-induced mutagenesis. Gly651 is in a stretch of amino acids conserved between humans and yeast, making it likely that Gly651 is also involved in DNA binding. However, future studies will be necessary to show that G651S does not possess other characteristics (eg, decreased stability) that impair MMS-induced mutagenesis.
Biochemical evidence suggesting that some disease-associated mutant variants were impaired for mtDNA replication was further supported by the observation that the mutant variants by themselves could not maintain mtDNA [9]. However, it has been unclear to what extent these polymerases function in the cell. It is well known that one of the catalytic aspartates (Asp891 in Mip1) is necessary for mtDNA replication; therefore, D891A is unable to catalyze the polymerase reaction. Interestingly, the heterozygous strain with D891A was associated with the largest increase in MMS mutagenesis, approximately 3-fold more than the exonuclease-deficient strain. Because the generation of mutations requires DNA replication, this result indicates that the wild type polymerase, which is normally accurate, becomes more likely to incorporate the wrong nucleotide in the D891A strain upon MMS exposure. In the case of the disease-associated mutations, it is possible that the mutant variants contribute to the incorporation of the incorrect nucleotide. However, removal of exonuclease activity in cis with Q264H and G651S showed no increase in mutation frequency regardless of MMS exposure and even showed an unexpected reduction of mutagenesis in the Q264H strain. Considering that the mutant frequency in the Q264H/exo− strain was similar to the G651S mutant frequency, this study cannot discount the possibility that the combination of the two mutations may have similar characteristics to G651S (eg, lower DNA binding affinity). Regardless, these results suggest that the mutant polymerase is not contributing directly to the mutations that drive the mutant frequency. It is possible that the mutant variants replicate mtDNA molecules that may be selected against or are not propagated, possibly because of incomplete replication.
Resistance to erythromycin is associated with a limited mutational spectrum that rarely includes C:G→G:C transversions [46]–[48]. Interestingly, exposure to MMS dramatically changed the mutation spectrum such that 40–45% of the mutations were C:G→G:C transversions. These mutations could either arise from a cytosine incorporated opposite a cytosine or a guanine incorporated opposite a guanine. Previously, MMS exposure of artificially formed or random ssDNA in yeast was associated with increased frequency of all 3 kinds of cytosine substitutions, C→T, C→G and C→A [42], [43]. The mutation spectra and strand bias suggested that N3-methyl cytosine is the prominent mutagenic lesion caused by MMS lesion in the yeast nuclear ssDNA [42], [43]. Therefore, it is possible that C:G→G:C transversions that predominate after MMS exposure result from cytosine incorporation opposite of a methylated cytosine.
These combined results support a model (Figure 8) wherein disease-associated mutant polymerase variants bind to and temporarily stall mtDNA replication, an event that could result in ssDNA intermediates (eg, because of polymerase-helicase uncoupling). Although mtDNA base excision repair would normally repair most damaged bases in dsDNA, there are no known repair systems that act on ssDNA in yeast. In the absence of MMS, the endogenous sources of DNA damage (eg, oxidative damage) impart a small but significant amount of DNA damage, whereas MMS magnifies the damage. Either the mtDNA continues to be stalled and degraded (an event that would not yield a mutant colony) or polymerase switching might occur, allowing the wild type polymerase access to the replication fork. In some cases the polymerase would incorporate the incorrect nucleotide leading to the development of a mutation. This model suggests that the mutant polymerase would stably bind DNA but be unable to replicate mtDNA efficiently.
Fig. 8. MMS-induced mutagenesis in Mip1 variants may be associated with unrepaired ssDNA damage. 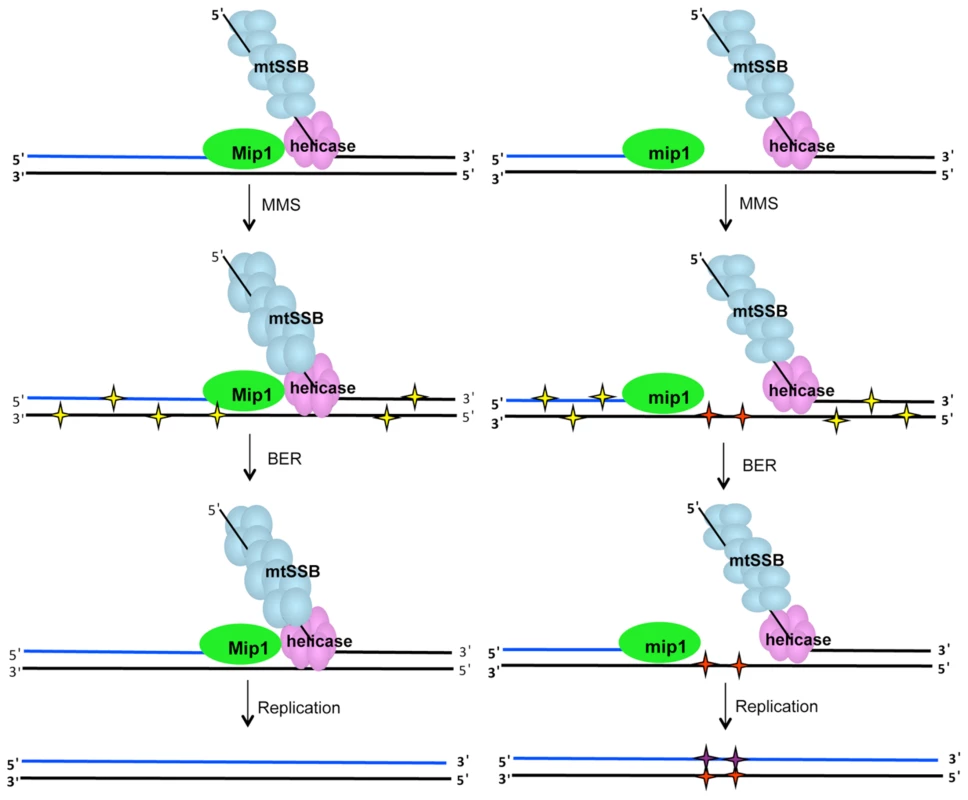
Under normal circumstances (left), single-stranded gaps at the replication fork are limited because of Mip1 processivity. However, reduced catalytic activity of the Mip1 disease variants (right scheme) results in the greater likelihood of stalling, which could produce long tracks of ssDNA if the helicase proceeds normally. When exposed to MMS, potentially mutagenic lesions in dsDNA (yellow stars) as well as N3-methylated cytosines in ssDNA (red stars) are produced. Although, BER is expected to correct the base damage in dsDNA, ssDNA lesions are not a BER substrate and are therefore unrepaired. Replication of the lesion is likely to result in incorporation of a cytosine opposite the N3-methylated cytosine to make the C:G→G:C mutation. BER, base excision repair; dsDNA, double-stranded DNA; MMS, methyl methanesulfonate; ssDNA, single-stranded DNA. Recently it was shown that the exonuclease domain suppresses mtDNA deletions between 21-mer direct repeats up to 160-fold [12]. One hypothesis could be that MMS also inhibits exonuclease activity which would lead to an increase in mutagenesis. This hypothesis was unlikely because MMS exhibited a modest effect on the diploid wild type strain. This study showed that MMS did not increase the frequency of mtDNA deletion mutants in wild type or Q264H background suggesting that the mechanism that caused MMS-induced point mutations is different than that of mtDNA deletion formation. Furthermore, MMS does not alter the exonuclease activity involved in suppression of mtDNA deletion formation.
A previous report showed that exposure to trace amounts of CdCl2 was associated not only with suppression of nuclear mismatch repair but also increase in petite colony formation frequency [39]. This study recapitulates this finding and further shows that CdCl2 does not significantly affect mtDNA mutant frequency. These results indicate that either there is no efficient mismatch repair system in yeast mtDNA or the amount of CdCl2 needed to observe a defect in mismatch repair is similar to the amount that is associated with a high frequency of dysfunctional mitochondrial. The only mismatch repair homologue associated with yeast mitochondria is Msh1, and it has been proposed to play a role in base excision repair [48]. This study also showed that mtDNA content was reduced in rho+ cells exposed to CdCl2 suggesting that cadmium negatively affects mtDNA replication or maintenance. We tested whether the effect of inhibiting mtDNA replication could mimic the effect of a disease-associated mutation by increasing MMS-induced mutagenesis. Unexpectedly, 3 and 4 µM CdCl2 did not promote MMS-induced mutagenesis in wild type cells. It is possible that CdCl2 does not stall replication but rather affects another replication-related process, such as replication initiation or termination. Even more unexpectedly, CdCl2 suppressed MMS-induced mutagenesis associated with Q264H. It should be noted that mutant frequency is determined among only rho+ cells so increased petite colony formation frequency from CdCl2 exposure does not explain the suppression of mutant frequency. One possible explanation is that the presence of CdCl2 selects against the maintenance of mtDNA molecules that are damaged or stalled either through some direct inhibition of the enzymes involved or as a result of a cellular response to mtDNA stress. Another possibility is that damaged mtDNA is more sensitive to CdCl2-induced inhibition of mtDNA replication, and the replication of these mtDNA molecules are not completed or maintained during the growth of the colony. Therefore, the various combinations of environmental exposures and genetics may be a useful tool to understand different pathways that occur in response to DNA damage.
This study shows a novel gene-environment interaction which greatly increases mtDNA mutagenesis and supports a polymerase-switching mechanism that has not been described in mtDNA replication. The interpretations of this study are limited in humans because there are key differences in yeast mtDNA maintenance compared with mammalian mtDNA (eg, lower mtDNA copy number and high frequency of recombination). Also, the study assumes that the mutant frequency (ie, mutants per culture) is indicative of the mutation frequency (mutations per mitochondrial division). True mutation frequencies would require information on mtDNA kinetics and mitochondrial dynamics that is currently unavailable. However, with the advent of high-throughput genome sequencing, similar studies in mtDNA mutagenesis will be possible in a model system that more closely mimics human mtDNA. Interestingly, this interaction involves heterozygotes containing mutations which are normally associated with disease [53]. It is interesting to consider that if a similar mechanism occurs in mammalian mtDNA, people who are heterozygous for a disease-associated mutation could be sensitive to environmental exposures that would impair mtDNA replication and promote symptoms involved in mitochondrial toxicity or disease.
Materials and Methods
Media and growth conditions
S. cerevisiae strains were grown at 30°C in YP (yeast extract 1%, peptone 2%) with 2% glucose or glycerol as carbon sources or synthetic complete media. Escherichia coli strains were grown in standard LB media at 37°C. When appropriate, gentamicin and ampicillin were added to YPD (0.2 mg/ml) and LB (0.1 mg/ml), respectively.
Plasmid constructions
The plasmid, pFL39, which contains MIP1 on a centromeric plasmid was previously described [31]. Site-directed mutagenesis of plasmid-encoded MIP1 was performed using the QuikChange Site-Directed Mutagenesis Kit (Invitrogen) as described previously. The construction of plasmids containing ACT1, COX1, and COX2 fragments, used for mtDNA quantitation was previously described.
Strain constructions
All S. cerevisiae strains were derived from E134 (MATα ade5-1 his7-2 lys2-A14 leu2-3,112 ura3-52 trp1-289) [54] and its MATa isogenic strain, YH747. Heteroallelic mip1 strains were made by transforming pFL39-MIP1 or a mutant derivative into E134 by selecting for TRP. Strains that measure deletions between direct repeats were created by transforming TRP1-containing plasmids PFL39 [12] containing wild type MIP1 or mip1 encoding an exonuclease-deficient mutant variant (mip1-exo [Het]; JSY114) into trp1::G418 NPY75 (JSY77) [12]. Chromosomal mip1 mutations were created in haploid E134 with wild type MIP1 using a PCR-based delitto perfetto method as previously described. All strains were checked phenotypically for the absence of the CORE casette used in delitto perfetto (ie, screening for Ura− and gentamicin sensitive cells) and were sequenced to confirm the presence of the mutation and the absence of undesired nearby mutations. The mip1 haploid mutant strains were mated with the isogenic E134 with mating type a.
Point and deletion mutant frequency measurements
Resistance to erythromycin is conferred by one of several missense mutations in the 21S rRNA gene in mitochondrial DNA [46], [47], [55]. Yeast strains were replica plated onto YPD plates with or without 1–5 mM MMS or 1–5 µM CdCl2 and grown at 30C for 2 days. In most cases 3 mM MMS was the highest exposure that allowed growth and was used for the experiments. After plating on CdCl2, increased chromosomal mutagenesis was confirmed qualitatively by the presence of colonies on media lacking lysine. From these plates, 20–40 independent colonies from each strain were used to inoculate into 4 ml of synthetic media without tryptophan (heteroallelic strains) or YPD (heterozygous strains), and these cultures were incubated to saturation (for 2 days) at 30°C. The cells were plated on YPEG (1.7% ethanol, 2% glycerol) with 4 g/L erythromycin. A small aliquot of 5–10 cultures were used to titer the number of rho+ cells by plating 10−5 dilutions on YPG. Erythromycin resistant colonies were counted after 6 days of incubation at 30°C. The mutant frequency was the median number of erythromycin colonies per 108 rho+ cells plated. To determine the spectrum of erythromycin-resistant mutations, DNA was extracted from one mutant per culture and used for as a template in a PCR reaction to amplify two regions, approximately 200 nucleotides flanking nucleotides 1950 (using 5′-GAGGTCCCGCATGAATGACG and 5′-CGATCTATCTAATTACAGTAAAGC) and 3993 (using 5′-CTATGTTTGCCACCTCGATGTC and 5′-CAATAGATACACCATGGGTTGATTC). The resulting amplified DNA was the template for the sequence reactions.
To measure deletions between direct repeats, all strains were replica plated onto YPD with or without MMS exposure as described above. Independent cultures were grown from at least 20 colonies at 30°C in YP (yeast extract 1%, peptone 2%) with 2% glucose and adenine for 2 days. Appropriate dilutions of samples from the saturated cultures were plated on synthetic complete media lacking arginine to determine total number of cells with mtDNA. The cultures were plated onto YP with 2% glycerol and deletion mutants were counted after 4 days. The mutant frequency was determined as the median number of mutant colonies per 108 Arg+ cells. In all mutagenesis experiments, 95% confidence levels were determined using the method of the median.
Petite frequency measurements
Petite frequencies are the frequency of rho− cells (petites) in the total population. Rho−cells are devoid of mitochondrial functions but are not necessarily devoid of mtDNA (rho0). To determine petite frequency in heteroallelic strains, at least 12 fresh transformants per strain were diluted in water and between 200–1000 colonies were plated onto YPD. For monoallelic strains, rho+ cells derived from several tetrad dissections were single colony purified on YPD and then assayed for petite frequency. Cells were incubated at 30°C for 2 days. Rho+ cells were identified either by the accumulation of red pigment as a result of mutations in the ade biosynthetic pathway or by the inability to grow on YPG. Using either or both method, at least 300 colonies per plate were counted, and petite colonies were identified. Frequencies were determined for each plate, and the median number of the frequencies was calculated. 95% confidence levels were determined by using the method of the median (52). For monoallelic strains determined to be 100% petite, no rho+ cells could be isolated from cells derived from 10 different haploid spores.
Mitochondrial DNA quantitation
Mitochondrial DNA copy number was quantified relative to nuclear DNA copy number using real time PCR. Primers and probes designed to specifically amplify within the mitochondrial-encoded COX1 gene and the nuclear-encoded ACT1 gene. Real time PCR reactions using Taqman Universal PCR Master Mix (Applied Biosystems) were performed at 40 cycles of 95 for 30 sec and 50 degrees for 30 sec. Known concentrations of plasmid molecules containing COX1and ACT1 were quantified as a positive control [9] and real time PCR was performed on 7 different dilutions to determine a logarithmic equation of a curve (R2 values>0.98) that represents numbers of molecules as a function of the critical threshold of every reaction. Every reaction was done in triplicate, and three replicates were tested for each experimental condition. Data represent the average ratio (± SEM) of the number of COX1 molecules to the number of ACT1 molecules.
Supporting Information
Zdroje
1. TrifunovicA, WredenbergA, FalkenbergM, SpelbrinkJN, RovioAT, et al. (2004) Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429 : 417–423.
2. KujothGC, HionaA, PughTD, SomeyaS, PanzerK, et al. (2005) Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science 309 : 481–484.
3. SafdarA, BourgeoisJM, OgbornDI, LittleJP, HettingaBP, et al. (2011) Endurance exercise rescues progeroid aging and induces systemic mitochondrial rejuvenation in mtDNA mutator mice. Proc Natl Acad Sci U S A 108 : 4135–4140.
4. Van GoethemG, DermautB, LofgrenA, MartinJJ, Van BroeckhovenC (2001) Mutation of POLG is associated with progressive external ophthalmoplegia characterized by mtDNA deletions. Nat Genet 28 : 211–212.
5. ChanSSL, LongleyMJ, CopelandWC (2005) The common A467T mutation in the human mitochondrial DNA polymerase (POLG) compromises catalytic efficiency and interaction with the accessory subunit. J Biol Chem 280 : 31341–31346.
6. ChanSSL, LongleyMJ, CopelandWC (2006) Modulation of the W748S mutation in DNA polymerase {gamma} by the E1143G polymorphism in mitochondrial disorders. Hum Mol Genet 15 : 3473–3483.
7. ChanSSL, LongleyMJ, NaviauxRK, CopelandWC (2005) Mono-allelic POLG expression resulting from nonsense-mediated decay and alternative splicing in a patient with Alpers syndrome. DNA Repair 4 : 1381–1389.
8. StuartGR, SantosJH, StrandMK, Van HoutenB, CopelandWC (2006) Mitochondrial DNA defects in S. cerevisiae with mutations in DNA polymerase gamma associated with Progressive External Ophthalmolplegia. Hum Mol Genet 15 : 363–374.
9. StumpfJD, BaileyCM, SpellD, StillwagonM, AndersonKS, et al. (2010) mip1 Containing mutations associated with mitochondrial disease causes mutagenesis and depletion of mtDNA in Saccharomyces cerevisiae. Hum Mol Genet 19 : 2123–2133.
10. SzczepanowskaK, FouryF (2010) A cluster of pathogenic mutations in the 3′-5′ exonuclease domain of DNA polymerase gamma defines a novel module coupling DNA synthesis and degradation. Hum Mol Genet 19 : 3516–3529.
11. LongleyMJ, ClarkS, Yu Wai ManC, HudsonG, DurhamSE, et al. (2006) Mutant POLG2 Disrupts DNA Polymerase gamma Subunits and Causes Progressive External Ophthalmoplegia. Am J Hum Genet 78 : 1026–1034.
12. StumpfJD, CopelandWC (2013) The exonuclease activity of the yeast mitochondrial DNA polymerase gamma suppresses mitochondrial DNA deletions between short direct repeats in Saccharomyces cerevisiae. Genetics 194 : 519–522.
13. BaruffiniE, FerreroI, FouryF (2007) Mitochondrial DNA defects in Saccharomyces cerevisiae caused by functional interactions between DNA polymerase gamma mutations associated with disease in human. Biochim Biophys Acta 1772 : 1225–1235.
14. BaruffiniE, LodiT, DallabonaC, PuglisiA, ZevianiM, et al. (2006) Genetic and chemical rescue of the Saccharomyces cerevisiae phenotype induced by mitochondrial DNA polymerase mutations associated with progressive external ophthalmoplegia in humans. Hum Mol Genet 15 : 2846–2855.
15. KakudaTN (2000) Pharmacology of nucleoside and nucleotide reverse transcriptase inhibitor-induced mitochondrial toxicity. Clin Ther 22 : 685–708.
16. StumpfJD, SanetoRP, CopelandWC (2013) Clinical and molecular features of POLG-related mitochondrial disease. Cold Spring Harb Perspect Biol 5: a011395.
17. BlackwoodW, BuxtonPH, CumingsJN, RobertsonDJ, TuckerSM (1963) Diffuse cerebral degeneration in infancy (Alpers' disease). Arch Dis Child 38 : 193–204.
18. GagoMF, RosasMJ, GuimaraesJ, FerreiraM, VilarinhoL, et al. (2006) SANDO: Two novel mutations in POLG1 gene. Neuromuscul Disord 16 : 507–509.
19. LeeYS, KennedyWD, YinYW (2009) Structural insight into processive human mitochondrial DNA synthesis and disease-related polymerase mutations. Cell 139 : 312–324.
20. LongleyMJ, PrasadR, SrivastavaDK, WilsonSH, CopelandWC (1998) Identification of 5′-deoxyribose phosphate lyase activity in human DNA polymerase gamma and its role in mitochondrial base excision repair in vitro. Proc Natl Acad Sci U S A 95 : 12244–12248.
21. LongleyMJ, RoppPA, LimSE, CopelandWC (1998) Characterization of the native and recombinant catalytic subunit of human DNA polymerase gamma: identification of residues critical for exonuclease activity and dideoxynucleotide sensitivity. Biochemistry 37 : 10529–10539.
22. JohnsonAA, JohnsonKA (2001) Exonuclease proofreading by human mitochondrial dna polymerase. J Biol Chem 276 : 38097–38107.
23. ChanSS, NaviauxRK, BasingerAA, CasasKA, CopelandWC (2009) De novo mutation in POLG leads to haplotype insufficiency and Alpers syndrome. Mitochondrion 9 : 340–345.
24. KasiviswanathanR, LongleyMJ, ChanSS, CopelandWC (2009) Disease mutations in the human mitochondrial DNA polymerase thumb subdomain impart severe defects in MtDNA replication. J Biol Chem 284 : 19501–19510.
25. PonamarevMV, LongleyMJ, NguyenD, KunkelTA, CopelandWC (2002) Active Site Mutation in DNA Polymerase gamma Associated with Progressive External Ophthalmoplegia Causes Error-prone DNA Synthesis. J Biol Chem 277 : 15225–15228.
26. GraziewiczMA, LongleyMJ, BienstockRJ, ZevianiM, CopelandWC (2004) Structure-function defects of human mitochondrial DNA polymerase in autosomal dominant progressive external ophthalmoplegia. Nat Struct Mol Biol 11 : 770–776.
27. GraziewiczMA, BienstockRJ, CopelandWC (2007) The DNA polymerase {gamma} Y955C disease variant associated with PEO and parkinsonism mediates the incorporation and translesion synthesis opposite 7,8-dihydro-8-oxo-2′-deoxyguanosine. Hum Mol Genet 16 : 2729–2739.
28. FouryF (1989) Cloning and sequencing of the nuclear gene MIP1 encoding the catalytic subunit of the yeast mitochondrial DNA polymerase. J Biol Chem 264 : 20552–20560.
29. RoppPA, CopelandWC (1996) Cloning and characterization of the human mitochondrial DNA polymerase, DNA polymerase gamma. Genomics 36 : 449–458.
30. BaruffiniE, FerreroI, FouryF (2010) In vivo analysis of mtDNA replication defects in yeast. Methods 51 : 426–436.
31. FouryF, VanderstraetenS (1992) Yeast mitochondrial DNA mutators with deficient proofreading exonucleolytic activity. Embo J 11 : 2717–2726.
32. VermulstM, BielasJH, KujothGC, LadigesWC, RabinovitchPS, et al. (2007) Mitochondrial point mutations do not limit the natural lifespan of mice. Nat Genet 39 : 540–543.
33. VermulstM, WanagatJ, KujothGC, BielasJH, RabinovitchPS, et al. (2008) DNA deletions and clonal mutations drive premature aging in mitochondrial mutator mice. Nat Genet 40 : 392–394.
34. LewisW, DayBJ, KohlerJJ, HosseiniSH, ChanSSL, et al. (2007) MtDNA depletion, oxidative stress, cardiomyopathy, and death from transgenic cardiac targeted human mutant polymerase gamma. Lab Invest 87 : 326–335.
35. PirselM, BohrVA (1993) Methyl methanesulfonate adduct formation and repair in the DHFR gene and in mitochondrial DNA in hamster cells. Carcinogenesis 14 : 2105–2108.
36. FurdaAM, MarrangoniAM, LokshinA, Van HoutenB (2012) Oxidants and not alkylating agents induce rapid mtDNA loss and mitochondrial dysfunction. DNA Repair (Amst) 11 : 684–692.
37. Acevedo-TorresK, Fonseca-WilliamsS, Ayala-TorresS, Torres-RamosCA (2009) Requirement of the Saccharomyces cerevisiae APN1 gene for the repair of mitochondrial DNA alkylation damage. Environ Mol Mutagen 50 : 317–327.
38. YouHJ, SwansonRL, HarringtonC, CorbettAH, Jinks-RobertsonS, et al. (1999) Saccharomyces cerevisiae Ntg1p and Ntg2p: broad specificity N-glycosylases for the repair of oxidative DNA damage in the nucleus and mitochondria. Biochemistry 38 : 11298–11306.
39. JinYH, ClarkAB, SlebosRJ, Al-RefaiH, TaylorJA, et al. (2003) Cadmium is a mutagen that acts by inhibiting mismatch repair. Nat Genet 34 : 326–329.
40. BogenhagenDF, PinzKG, Perez-JannottiRM (2001) Enzymology of mitochondrial base excision repair. Prog Nucleic Acid Res Mol Biol 68 : 257–271.
41. YangY, SterlingJ, StoriciF, ResnickMA, GordeninDA (2008) Hypermutability of damaged single-strand DNA formed at double-strand breaks and uncapped telomeres in yeast Saccharomyces cerevisiae. PLoS Genet 4: e1000264.
42. RobertsSA, SterlingJ, ThompsonC, HarrisS, MavD, et al. (2012) Clustered mutations in yeast and in human cancers can arise from damaged long single-strand DNA regions. Mol Cell 46 : 424–435.
43. YangY, GordeninDA, ResnickMA (2010) A single-strand specific lesion drives MMS-induced hyper-mutability at a double-strand break in yeast. DNA Repair (Amst) 9 : 914–921.
44. ClaytonDA (1982) Replication of animal mitochondrial DNA. Cell 28 : 693–705.
45. CopelandWC, WangTS (1993) Mutational analysis of the human DNA polymerase alpha. The most conserved region in alpha-like DNA polymerases is involved in metal - specific catalysis. J Biol Chem 268 : 11028–11040.
46. KalifaL, SiaEA (2007) Analysis of Rev1p and Pol zeta in mitochondrial mutagenesis suggests an alternative pathway of damage tolerance. DNA Repair (Amst) 6 : 1732–1739.
47. VanderstraetenS, Van den BruleS, HuJ, FouryF (1998) The role of 3′-5′ exonucleolytic proofreading and mismatch repair in yeast mitochondrial DNA error avoidance. J Biol Chem 273 : 23690–23697.
48. PogorzalaL, MookerjeeS, SiaEA (2009) Evidence that msh1p plays multiple roles in mitochondrial base excision repair. Genetics 182 : 699–709.
49. StrandMK, StuartGR, LongleyMJ, GraziewiczMA, DominickOC, et al. (2003) POS5 gene of Saccharomyces cerevisiae encodes a mitochondrial NADH kinase required for stability of mitochondrial DNA. Eukaryot Cell 2 : 809–820.
50. KalifaL, BeutnerG, PhadnisN, SheuSS, SiaEA (2009) Evidence for a role of FEN1 in maintaining mitochondrial DNA integrity. DNA Repair (Amst) 8 : 1242–1249.
51. PhadnisN, MehtaR, MeednuN, SiaEA (2006) Ntg1p, the base excision repair protein, generates mutagenic intermediates in yeast mitochondrial DNA. DNA Repair (Amst) 5 : 829–839.
52. DoudicanNA, SongB, ShadelGS, DoetschPW (2005) Oxidative DNA damage causes mitochondrial genomic instability in Saccharomyces cerevisiae. Mol Cell Biol 25 : 5196–5204.
53. StumpfJD, CopelandWC (2011) Mitochondrial DNA replication and disease: insights from DNA polymerase gamma mutations. Cell Mol Life Sci 68 : 219–233.
54. TranHT, KeenJD, KrickerM, ResnickMA, GordeninDA (1997) Hypermutability of homonucleotide runs in mismatch repair and DNA polymerase proofreading yeast mutants. Mol Cell Biol 17 : 2859–2865.
55. SorF, FukuharaH (1984) Erythromycin and spiramycin resistance mutations of yeast mitochondria: nature of the rib2 locus in the large ribosomal RNA gene. Nucleic Acids Res 12 : 8313–8318.
Štítky
Genetika Reprodukčná medicína
Článek Oligoasthenoteratozoospermia and Infertility in Mice Deficient for miR-34b/c and miR-449 LociČlánek The Kinesin AtPSS1 Promotes Synapsis and is Required for Proper Crossover Distribution in MeiosisČlánek Payoffs, Not Tradeoffs, in the Adaptation of a Virus to Ostensibly Conflicting Selective PressuresČlánek Examination of Prokaryotic Multipartite Genome Evolution through Experimental Genome ReductionČlánek BMP-FGF Signaling Axis Mediates Wnt-Induced Epidermal Stratification in Developing Mammalian SkinČlánek Role of STN1 and DNA Polymerase α in Telomere Stability and Genome-Wide Replication in ArabidopsisČlánek RNA-Processing Protein TDP-43 Regulates FOXO-Dependent Protein Quality Control in Stress ResponseČlánek Integrating Functional Data to Prioritize Causal Variants in Statistical Fine-Mapping StudiesČlánek Salt-Induced Stabilization of EIN3/EIL1 Confers Salinity Tolerance by Deterring ROS Accumulation inČlánek Ethylene-Induced Inhibition of Root Growth Requires Abscisic Acid Function in Rice ( L.) SeedlingsČlánek Metabolic Respiration Induces AMPK- and Ire1p-Dependent Activation of the p38-Type HOG MAPK PathwayČlánek Signature Gene Expression Reveals Novel Clues to the Molecular Mechanisms of Dimorphic Transition inČlánek A Mouse Model Uncovers LKB1 as an UVB-Induced DNA Damage Sensor Mediating CDKN1A (p21) DegradationČlánek Dominant Sequences of Human Major Histocompatibility Complex Conserved Extended Haplotypes from to
Článok vyšiel v časopisePLOS Genetics
Najčítanejšie tento týždeň
2014 Číslo 10- Gynekologové a odborníci na reprodukční medicínu se sejdou na prvním virtuálním summitu
- Je „freeze-all“ pro všechny? Odborníci na fertilitu diskutovali na virtuálním summitu
-
Všetky články tohto čísla
- An Deletion Is Highly Associated with a Juvenile-Onset Inherited Polyneuropathy in Leonberger and Saint Bernard Dogs
- Licensing of Yeast Centrosome Duplication Requires Phosphoregulation of Sfi1
- Oligoasthenoteratozoospermia and Infertility in Mice Deficient for miR-34b/c and miR-449 Loci
- Basement Membrane and Cell Integrity of Self-Tissues in Maintaining Immunological Tolerance
- The Kinesin AtPSS1 Promotes Synapsis and is Required for Proper Crossover Distribution in Meiosis
- Germline Mutations in Are Associated with Familial Gastric Cancer
- POT1a and Components of CST Engage Telomerase and Regulate Its Activity in
- Controlling Meiotic Recombinational Repair – Specifying the Roles of ZMMs, Sgs1 and Mus81/Mms4 in Crossover Formation
- Payoffs, Not Tradeoffs, in the Adaptation of a Virus to Ostensibly Conflicting Selective Pressures
- FHIT Suppresses Epithelial-Mesenchymal Transition (EMT) and Metastasis in Lung Cancer through Modulation of MicroRNAs
- Genome-Wide Mapping of Yeast RNA Polymerase II Termination
- Examination of Prokaryotic Multipartite Genome Evolution through Experimental Genome Reduction
- White Cells Facilitate Opposite- and Same-Sex Mating of Opaque Cells in
- BMP-FGF Signaling Axis Mediates Wnt-Induced Epidermal Stratification in Developing Mammalian Skin
- Genome-Wide Association Study of CSF Levels of 59 Alzheimer's Disease Candidate Proteins: Significant Associations with Proteins Involved in Amyloid Processing and Inflammation
- COE Loss-of-Function Analysis Reveals a Genetic Program Underlying Maintenance and Regeneration of the Nervous System in Planarians
- Fat-Dachsous Signaling Coordinates Cartilage Differentiation and Polarity during Craniofacial Development
- Identification of Genes Important for Cutaneous Function Revealed by a Large Scale Reverse Genetic Screen in the Mouse
- Sensors at Centrosomes Reveal Determinants of Local Separase Activity
- Genes Integrate and Hedgehog Pathways in the Second Heart Field for Cardiac Septation
- Systematic Dissection of Coding Exons at Single Nucleotide Resolution Supports an Additional Role in Cell-Specific Transcriptional Regulation
- Recovery from an Acute Infection in Requires the GATA Transcription Factor ELT-2
- HIPPO Pathway Members Restrict SOX2 to the Inner Cell Mass Where It Promotes ICM Fates in the Mouse Blastocyst
- Role of and in Development of Abdominal Epithelia Breaks Posterior Prevalence Rule
- The Formation of Endoderm-Derived Taste Sensory Organs Requires a -Dependent Expansion of Embryonic Taste Bud Progenitor Cells
- Role of STN1 and DNA Polymerase α in Telomere Stability and Genome-Wide Replication in Arabidopsis
- Keratin 76 Is Required for Tight Junction Function and Maintenance of the Skin Barrier
- Encodes the Catalytic Subunit of N Alpha-Acetyltransferase that Regulates Development, Metabolism and Adult Lifespan
- Disruption of SUMO-Specific Protease 2 Induces Mitochondria Mediated Neurodegeneration
- Caudal Regulates the Spatiotemporal Dynamics of Pair-Rule Waves in
- It's All in Your Mind: Determining Germ Cell Fate by Neuronal IRE-1 in
- A Conserved Role for Homologs in Protecting Dopaminergic Neurons from Oxidative Stress
- The Master Activator of IncA/C Conjugative Plasmids Stimulates Genomic Islands and Multidrug Resistance Dissemination
- An AGEF-1/Arf GTPase/AP-1 Ensemble Antagonizes LET-23 EGFR Basolateral Localization and Signaling during Vulva Induction
- The Proteomic Landscape of the Suprachiasmatic Nucleus Clock Reveals Large-Scale Coordination of Key Biological Processes
- RNA-Processing Protein TDP-43 Regulates FOXO-Dependent Protein Quality Control in Stress Response
- A Complex Genetic Switch Involving Overlapping Divergent Promoters and DNA Looping Regulates Expression of Conjugation Genes of a Gram-positive Plasmid
- ZTF-8 Interacts with the 9-1-1 Complex and Is Required for DNA Damage Response and Double-Strand Break Repair in the Germline
- Integrating Functional Data to Prioritize Causal Variants in Statistical Fine-Mapping Studies
- Tpz1-Ccq1 and Tpz1-Poz1 Interactions within Fission Yeast Shelterin Modulate Ccq1 Thr93 Phosphorylation and Telomerase Recruitment
- Salt-Induced Stabilization of EIN3/EIL1 Confers Salinity Tolerance by Deterring ROS Accumulation in
- Telomeric (s) in spp. Encode Mediator Subunits That Regulate Distinct Virulence Traits
- Ethylene-Induced Inhibition of Root Growth Requires Abscisic Acid Function in Rice ( L.) Seedlings
- Ancient Expansion of the Hox Cluster in Lepidoptera Generated Four Homeobox Genes Implicated in Extra-Embryonic Tissue Formation
- Mechanism of Suppression of Chromosomal Instability by DNA Polymerase POLQ
- A Mutation in the Mouse Gene Leads to Impaired Hedgehog Signaling
- Keeping mtDNA in Shape between Generations
- Targeted Exon Capture and Sequencing in Sporadic Amyotrophic Lateral Sclerosis
- TIF-IA-Dependent Regulation of Ribosome Synthesis in Muscle Is Required to Maintain Systemic Insulin Signaling and Larval Growth
- At Short Telomeres Tel1 Directs Early Replication and Phosphorylates Rif1
- Evidence of a Bacterial Receptor for Lysozyme: Binding of Lysozyme to the Anti-σ Factor RsiV Controls Activation of the ECF σ Factor σ
- Hsp40s Specify Functions of Hsp104 and Hsp90 Protein Chaperone Machines
- Feeding State, Insulin and NPR-1 Modulate Chemoreceptor Gene Expression via Integration of Sensory and Circuit Inputs
- Functional Interaction between Ribosomal Protein L6 and RbgA during Ribosome Assembly
- Multiple Regulatory Systems Coordinate DNA Replication with Cell Growth in
- Fast Evolution from Precast Bricks: Genomics of Young Freshwater Populations of Threespine Stickleback
- Mmp1 Processing of the PDF Neuropeptide Regulates Circadian Structural Plasticity of Pacemaker Neurons
- The Nuclear Immune Receptor Is Required for -Dependent Constitutive Defense Activation in
- Genetic Modifiers of Neurofibromatosis Type 1-Associated Café-au-Lait Macule Count Identified Using Multi-platform Analysis
- Juvenile Hormone-Receptor Complex Acts on and to Promote Polyploidy and Vitellogenesis in the Migratory Locust
- Uncovering Enhancer Functions Using the α-Globin Locus
- The Analysis of Mutant Alleles of Different Strength Reveals Multiple Functions of Topoisomerase 2 in Regulation of Chromosome Structure
- Metabolic Respiration Induces AMPK- and Ire1p-Dependent Activation of the p38-Type HOG MAPK Pathway
- The Specification and Global Reprogramming of Histone Epigenetic Marks during Gamete Formation and Early Embryo Development in
- The DAF-16 FOXO Transcription Factor Regulates to Modulate Stress Resistance in , Linking Insulin/IGF-1 Signaling to Protein N-Terminal Acetylation
- Genetic Influences on Translation in Yeast
- Analysis of Mutants Defective in the Cdk8 Module of Mediator Reveal Links between Metabolism and Biofilm Formation
- Ribosomal Readthrough at a Short UGA Stop Codon Context Triggers Dual Localization of Metabolic Enzymes in Fungi and Animals
- Gene Duplication Restores the Viability of Δ and Δ Mutants
- Selection on a Variant Associated with Improved Viral Clearance Drives Local, Adaptive Pseudogenization of Interferon Lambda 4 ()
- Break-Induced Replication Requires DNA Damage-Induced Phosphorylation of Pif1 and Leads to Telomere Lengthening
- Dynamic Partnership between TFIIH, PGC-1α and SIRT1 Is Impaired in Trichothiodystrophy
- Signature Gene Expression Reveals Novel Clues to the Molecular Mechanisms of Dimorphic Transition in
- Mutations in Moderate or Severe Intellectual Disability
- Multifaceted Genome Control by Set1 Dependent and Independent of H3K4 Methylation and the Set1C/COMPASS Complex
- A Role for Taiman in Insect Metamorphosis
- The Small RNA Rli27 Regulates a Cell Wall Protein inside Eukaryotic Cells by Targeting a Long 5′-UTR Variant
- MMS Exposure Promotes Increased MtDNA Mutagenesis in the Presence of Replication-Defective Disease-Associated DNA Polymerase γ Variants
- Coexistence and Within-Host Evolution of Diversified Lineages of Hypermutable in Long-term Cystic Fibrosis Infections
- Comprehensive Mapping of the Flagellar Regulatory Network
- Topoisomerase II Is Required for the Proper Separation of Heterochromatic Regions during Female Meiosis
- A Splice Mutation in the Gene Causes High Glycogen Content and Low Meat Quality in Pig Skeletal Muscle
- KDM5 Interacts with Foxo to Modulate Cellular Levels of Oxidative Stress
- H2B Mono-ubiquitylation Facilitates Fork Stalling and Recovery during Replication Stress by Coordinating Rad53 Activation and Chromatin Assembly
- Copy Number Variation in the Horse Genome
- Unifying Genetic Canalization, Genetic Constraint, and Genotype-by-Environment Interaction: QTL by Genomic Background by Environment Interaction of Flowering Time in
- Spinster Homolog 2 () Deficiency Causes Early Onset Progressive Hearing Loss
- Genome-Wide Discovery of Drug-Dependent Human Liver Regulatory Elements
- Developmentally-Regulated Excision of the SPβ Prophage Reconstitutes a Gene Required for Spore Envelope Maturation in
- Protein Phosphatase 4 Promotes Chromosome Pairing and Synapsis, and Contributes to Maintaining Crossover Competence with Increasing Age
- The bHLH-PAS Transcription Factor Dysfusion Regulates Tarsal Joint Formation in Response to Notch Activity during Leg Development
- A Mouse Model Uncovers LKB1 as an UVB-Induced DNA Damage Sensor Mediating CDKN1A (p21) Degradation
- Notch3 Interactome Analysis Identified WWP2 as a Negative Regulator of Notch3 Signaling in Ovarian Cancer
- An Integrated Cell Purification and Genomics Strategy Reveals Multiple Regulators of Pancreas Development
- Dominant Sequences of Human Major Histocompatibility Complex Conserved Extended Haplotypes from to
- The Vesicle Protein SAM-4 Regulates the Processivity of Synaptic Vesicle Transport
- A Gain-of-Function Mutation in Impeded Bone Development through Increasing Expression in DA2B Mice
- Nephronophthisis-Associated Regulates Cell Cycle Progression, Apoptosis and Epithelial-to-Mesenchymal Transition
- Beclin 1 Is Required for Neuron Viability and Regulates Endosome Pathways via the UVRAG-VPS34 Complex
- The Not5 Subunit of the Ccr4-Not Complex Connects Transcription and Translation
- Abnormal Dosage of Ultraconserved Elements Is Highly Disfavored in Healthy Cells but Not Cancer Cells
- Genome-Wide Distribution of RNA-DNA Hybrids Identifies RNase H Targets in tRNA Genes, Retrotransposons and Mitochondria
- The Chromosomal Association of the Smc5/6 Complex Depends on Cohesion and Predicts the Level of Sister Chromatid Entanglement
- Cell-Autonomous Progeroid Changes in Conditional Mouse Models for Repair Endonuclease XPG Deficiency
- PLOS Genetics
- Archív čísel
- Aktuálne číslo
- Informácie o časopise
Najčítanejšie v tomto čísle- The Master Activator of IncA/C Conjugative Plasmids Stimulates Genomic Islands and Multidrug Resistance Dissemination
- A Splice Mutation in the Gene Causes High Glycogen Content and Low Meat Quality in Pig Skeletal Muscle
- Keratin 76 Is Required for Tight Junction Function and Maintenance of the Skin Barrier
- A Role for Taiman in Insect Metamorphosis
Prihlásenie#ADS_BOTTOM_SCRIPTS#Zabudnuté hesloZadajte e-mailovú adresu, s ktorou ste vytvárali účet. Budú Vám na ňu zasielané informácie k nastaveniu nového hesla.
- Časopisy



